Yi Xin
Rethinking the Design Space of Reinforcement Learning for Diffusion Models: On the Importance of Likelihood Estimation Beyond Loss Design
Feb 04, 2026Abstract:Reinforcement learning has been widely applied to diffusion and flow models for visual tasks such as text-to-image generation. However, these tasks remain challenging because diffusion models have intractable likelihoods, which creates a barrier for directly applying popular policy-gradient type methods. Existing approaches primarily focus on crafting new objectives built on already heavily engineered LLM objectives, using ad hoc estimators for likelihood, without a thorough investigation into how such estimation affects overall algorithmic performance. In this work, we provide a systematic analysis of the RL design space by disentangling three factors: i) policy-gradient objectives, ii) likelihood estimators, and iii) rollout sampling schemes. We show that adopting an evidence lower bound (ELBO) based model likelihood estimator, computed only from the final generated sample, is the dominant factor enabling effective, efficient, and stable RL optimization, outweighing the impact of the specific policy-gradient loss functional. We validate our findings across multiple reward benchmarks using SD 3.5 Medium, and observe consistent trends across all tasks. Our method improves the GenEval score from 0.24 to 0.95 in 90 GPU hours, which is $4.6\times$ more efficient than FlowGRPO and $2\times$ more efficient than the SOTA method DiffusionNFT without reward hacking.
Modality Gap-Driven Subspace Alignment Training Paradigm For Multimodal Large Language Models
Feb 02, 2026Abstract:Despite the success of multimodal contrastive learning in aligning visual and linguistic representations, a persistent geometric anomaly, the Modality Gap, remains: embeddings of distinct modalities expressing identical semantics occupy systematically offset regions. Prior approaches to bridge this gap are largely limited by oversimplified isotropic assumptions, hindering their application in large-scale scenarios. In this paper, we address these limitations by precisely characterizing the geometric shape of the modality gap and leveraging it for efficient model scaling. First, we propose the Fixed-frame Modality Gap Theory, which decomposes the modality gap within a frozen reference frame into stable biases and anisotropic residuals. Guided by this precise modeling, we introduce ReAlign, a training-free modality alignment strategy. Utilizing statistics from massive unpaired data, ReAlign aligns text representation into the image representation distribution via a three-step process comprising Anchor, Trace, and Centroid Alignment, thereby explicitly rectifying geometric misalignment. Building on ReAlign, we propose ReVision, a scalable training paradigm for Multimodal Large Language Models (MLLMs). ReVision integrates ReAlign into the pretraining stage, enabling the model to learn the distribution of visual representations from unpaired text before visual instruction tuning, without the need for large-scale, high-quality image-text pairs. Our framework demonstrates that statistically aligned unpaired data can effectively substitute for expensive image-text pairs, offering a robust path for the efficient scaling of MLLMs.
Prism: Efficient Test-Time Scaling via Hierarchical Search and Self-Verification for Discrete Diffusion Language Models
Feb 02, 2026Abstract:Inference-time compute has re-emerged as a practical way to improve LLM reasoning. Most test-time scaling (TTS) algorithms rely on autoregressive decoding, which is ill-suited to discrete diffusion language models (dLLMs) due to their parallel decoding over the entire sequence. As a result, developing effective and efficient TTS methods to unlock dLLMs' full generative potential remains an underexplored challenge. To address this, we propose Prism (Pruning, Remasking, and Integrated Self-verification Method), an efficient TTS framework for dLLMs that (i) performs Hierarchical Trajectory Search (HTS) which dynamically prunes and reallocates compute in an early-to-mid denoising window, (ii) introduces Local branching with partial remasking to explore diverse implementations while preserving high-confidence tokens, and (iii) replaces external verifiers with Self-Verified Feedback (SVF) obtained via self-evaluation prompts on intermediate completions. Across four mathematical reasoning and code generation benchmarks on three dLLMs, including LLaDA 8B Instruct, Dream 7B Instruct, and LLaDA 2.0-mini, our Prism achieves a favorable performance-efficiency trade-off, matching best-of-N performance with substantially fewer function evaluations (NFE). The code is released at https://github.com/viiika/Prism.
UniPercept: Towards Unified Perceptual-Level Image Understanding across Aesthetics, Quality, Structure, and Texture
Dec 25, 2025



Abstract:Multimodal large language models (MLLMs) have achieved remarkable progress in visual understanding tasks such as visual grounding, segmentation, and captioning. However, their ability to perceive perceptual-level image features remains limited. In this work, we present UniPercept-Bench, a unified framework for perceptual-level image understanding across three key domains: Aesthetics, Quality, Structure and Texture. We establish a hierarchical definition system and construct large-scale datasets to evaluate perceptual-level image understanding. Based on this foundation, we develop a strong baseline UniPercept trained via Domain-Adaptive Pre-Training and Task-Aligned RL, enabling robust generalization across both Visual Rating (VR) and Visual Question Answering (VQA) tasks. UniPercept outperforms existing MLLMs on perceptual-level image understanding and can serve as a plug-and-play reward model for text-to-image generation. This work defines Perceptual-Level Image Understanding in the era of MLLMs and, through the introduction of a comprehensive benchmark together with a strong baseline, provides a solid foundation for advancing perceptual-level multimodal image understanding.
dMLLM-TTS: Self-Verified and Efficient Test-Time Scaling for Diffusion Multi-Modal Large Language Models
Dec 22, 2025



Abstract:Diffusion Multi-modal Large Language Models (dMLLMs) have recently emerged as a novel architecture unifying image generation and understanding. However, developing effective and efficient Test-Time Scaling (TTS) methods to unlock their full generative potential remains an underexplored challenge. To address this, we propose dMLLM-TTS, a novel framework operating on two complementary scaling axes: (1) trajectory exploration scaling to enhance the diversity of generated hypotheses, and (2) iterative refinement scaling for stable generation. Conventional TTS approaches typically perform linear search across these two dimensions, incurring substantial computational costs of O(NT) and requiring an external verifier for best-of-N selection. To overcome these limitations, we propose two innovations. First, we design an efficient hierarchical search algorithm with O(N+T) complexity that adaptively expands and prunes sampling trajectories. Second, we introduce a self-verified feedback mechanism that leverages the dMLLMs' intrinsic image understanding capabilities to assess text-image alignment, eliminating the need for external verifier. Extensive experiments on the GenEval benchmark across three representative dMLLMs (e.g., Lumina-DiMOO, MMaDA, Muddit) show that our framework substantially improves generation quality while achieving up to 6x greater efficiency than linear search. Project page: https://github.com/Alpha-VLLM/Lumina-DiMOO.
From Masks to Worlds: A Hitchhiker's Guide to World Models
Oct 23, 2025Abstract:This is not a typical survey of world models; it is a guide for those who want to build worlds. We do not aim to catalog every paper that has ever mentioned a ``world model". Instead, we follow one clear road: from early masked models that unified representation learning across modalities, to unified architectures that share a single paradigm, then to interactive generative models that close the action-perception loop, and finally to memory-augmented systems that sustain consistent worlds over time. We bypass loosely related branches to focus on the core: the generative heart, the interactive loop, and the memory system. We show that this is the most promising path towards true world models.
LayerT2V: Interactive Multi-Object Trajectory Layering for Video Generation
Aug 06, 2025Abstract:Controlling object motion trajectories in Text-to-Video (T2V) generation is a challenging and relatively under-explored area, particularly in scenarios involving multiple moving objects. Most community models and datasets in the T2V domain are designed for single-object motion, limiting the performance of current generative models in multi-object tasks. Additionally, existing motion control methods in T2V either lack support for multi-object motion scenes or experience severe performance degradation when object trajectories intersect, primarily due to the semantic conflicts in colliding regions. To address these limitations, we introduce LayerT2V, the first approach for generating video by compositing background and foreground objects layer by layer. This layered generation enables flexible integration of multiple independent elements within a video, positioning each element on a distinct "layer" and thus facilitating coherent multi-object synthesis while enhancing control over the generation process. Extensive experiments demonstrate the superiority of LayerT2V in generating complex multi-object scenarios, showcasing 1.4x and 4.5x improvements in mIoU and AP50 metrics over state-of-the-art (SOTA) methods. Project page and code are available at https://kr-panghu.github.io/LayerT2V/ .
TR-PTS: Task-Relevant Parameter and Token Selection for Efficient Tuning
Jul 30, 2025Abstract:Large pre-trained models achieve remarkable performance in vision tasks but are impractical for fine-tuning due to high computational and storage costs. Parameter-Efficient Fine-Tuning (PEFT) methods mitigate this issue by updating only a subset of parameters; however, most existing approaches are task-agnostic, failing to fully exploit task-specific adaptations, which leads to suboptimal efficiency and performance. To address this limitation, we propose Task-Relevant Parameter and Token Selection (TR-PTS), a task-driven framework that enhances both computational efficiency and accuracy. Specifically, we introduce Task-Relevant Parameter Selection, which utilizes the Fisher Information Matrix (FIM) to identify and fine-tune only the most informative parameters in a layer-wise manner, while keeping the remaining parameters frozen. Simultaneously, Task-Relevant Token Selection dynamically preserves the most informative tokens and merges redundant ones, reducing computational overhead. By jointly optimizing parameters and tokens, TR-PTS enables the model to concentrate on task-discriminative information. We evaluate TR-PTS on benchmark, including FGVC and VTAB-1k, where it achieves state-of-the-art performance, surpassing full fine-tuning by 3.40% and 10.35%, respectively. The code are available at https://github.com/synbol/TR-PTS.
Low-Cost Test-Time Adaptation for Robust Video Editing
Jul 29, 2025



Abstract:Video editing is a critical component of content creation that transforms raw footage into coherent works aligned with specific visual and narrative objectives. Existing approaches face two major challenges: temporal inconsistencies due to failure in capturing complex motion patterns, and overfitting to simple prompts arising from limitations in UNet backbone architectures. While learning-based methods can enhance editing quality, they typically demand substantial computational resources and are constrained by the scarcity of high-quality annotated data. In this paper, we present Vid-TTA, a lightweight test-time adaptation framework that personalizes optimization for each test video during inference through self-supervised auxiliary tasks. Our approach incorporates a motion-aware frame reconstruction mechanism that identifies and preserves crucial movement regions, alongside a prompt perturbation and reconstruction strategy that strengthens model robustness to diverse textual descriptions. These innovations are orchestrated by a meta-learning driven dynamic loss balancing mechanism that adaptively adjusts the optimization process based on video characteristics. Extensive experiments demonstrate that Vid-TTA significantly improves video temporal consistency and mitigates prompt overfitting while maintaining low computational overhead, offering a plug-and-play performance boost for existing video editing models.
Lumina-mGPT 2.0: Stand-Alone AutoRegressive Image Modeling
Jul 23, 2025
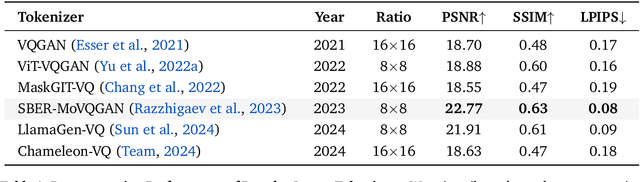
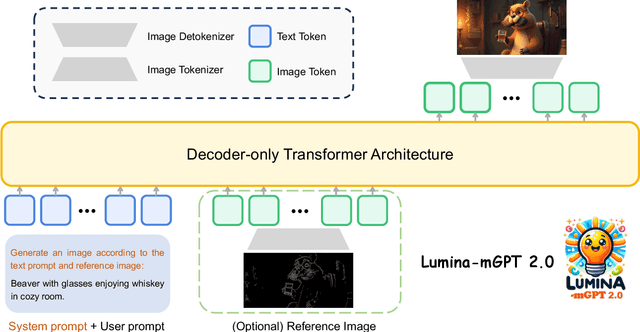
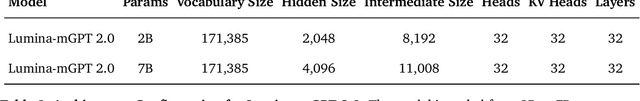
Abstract:We present Lumina-mGPT 2.0, a stand-alone, decoder-only autoregressive model that revisits and revitalizes the autoregressive paradigm for high-quality image generation and beyond. Unlike existing approaches that rely on pretrained components or hybrid architectures, Lumina-mGPT 2.0 is trained entirely from scratch, enabling unrestricted architectural design and licensing freedom. It achieves generation quality on par with state-of-the-art diffusion models such as DALL-E 3 and SANA, while preserving the inherent flexibility and compositionality of autoregressive modeling. Our unified tokenization scheme allows the model to seamlessly handle a wide spectrum of tasks-including subject-driven generation, image editing, controllable synthesis, and dense prediction-within a single generative framework. To further boost usability, we incorporate efficient decoding strategies like inference-time scaling and speculative Jacobi sampling to improve quality and speed, respectively. Extensive evaluations on standard text-to-image benchmarks (e.g., GenEval, DPG) demonstrate that Lumina-mGPT 2.0 not only matches but in some cases surpasses diffusion-based models. Moreover, we confirm its multi-task capabilities on the Graph200K benchmark, with the native Lumina-mGPT 2.0 performing exceptionally well. These results position Lumina-mGPT 2.0 as a strong, flexible foundation model for unified multimodal generation. We have released our training details, code, and models at https://github.com/Alpha-VLLM/Lumina-mGPT-2.0.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge