Ling Shao
Terminus Group, Beijing, China
Dynamic Dual-Attentive Aggregation Learning for Visible-Infrared Person Re-Identification
Jul 18, 2020
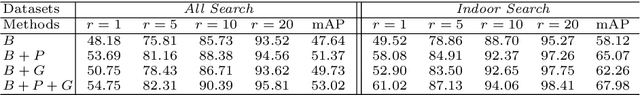
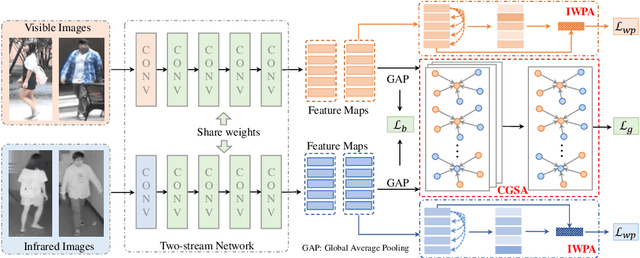
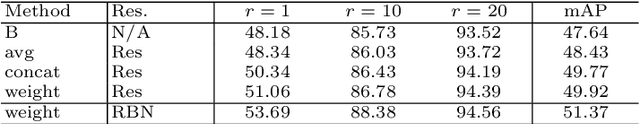
Abstract:Visible-infrared person re-identification (VI-ReID) is a challenging cross-modality pedestrian retrieval problem. Due to the large intra-class variations and cross-modality discrepancy with large amount of sample noise, it is difficult to learn discriminative part features. Existing VI-ReID methods instead tend to learn global representations, which have limited discriminability and weak robustness to noisy images. In this paper, we propose a novel dynamic dual-attentive aggregation (DDAG) learning method by mining both intra-modality part-level and cross-modality graph-level contextual cues for VI-ReID. We propose an intra-modality weighted-part attention module to extract discriminative part-aggregated features, by imposing the domain knowledge on the part relationship mining. To enhance robustness against noisy samples, we introduce cross-modality graph structured attention to reinforce the representation with the contextual relations across the two modalities. We also develop a parameter-free dynamic dual aggregation learning strategy to adaptively integrate the two components in a progressive joint training manner. Extensive experiments demonstrate that DDAG outperforms the state-of-the-art methods under various settings.
Learning to Learn with Variational Information Bottleneck for Domain Generalization
Jul 15, 2020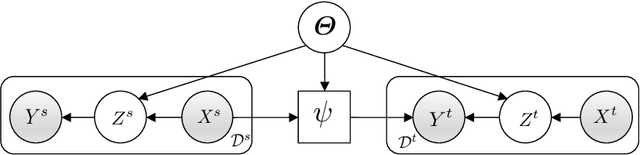
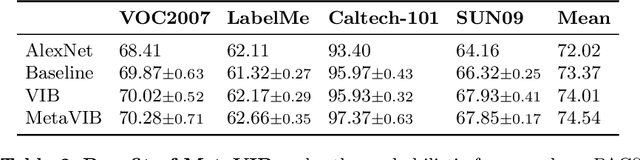

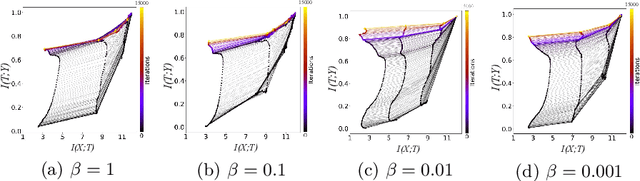
Abstract:Domain generalization models learn to generalize to previously unseen domains, but suffer from prediction uncertainty and domain shift. In this paper, we address both problems. We introduce a probabilistic meta-learning model for domain generalization, in which classifier parameters shared across domains are modeled as distributions. This enables better handling of prediction uncertainty on unseen domains. To deal with domain shift, we learn domain-invariant representations by the proposed principle of meta variational information bottleneck, we call MetaVIB. MetaVIB is derived from novel variational bounds of mutual information, by leveraging the meta-learning setting of domain generalization. Through episodic training, MetaVIB learns to gradually narrow domain gaps to establish domain-invariant representations, while simultaneously maximizing prediction accuracy. We conduct experiments on three benchmarks for cross-domain visual recognition. Comprehensive ablation studies validate the benefits of MetaVIB for domain generalization. The comparison results demonstrate our method outperforms previous approaches consistently.
PraNet: Parallel Reverse Attention Network for Polyp Segmentation
Jul 03, 2020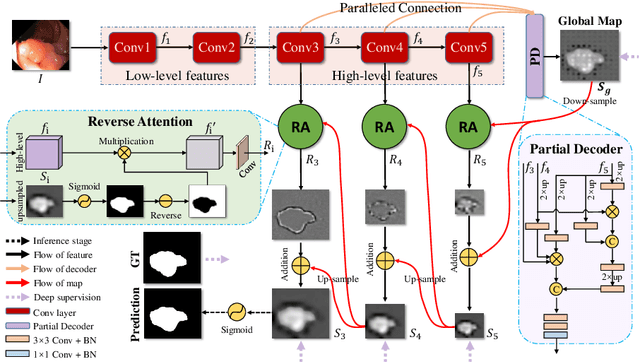
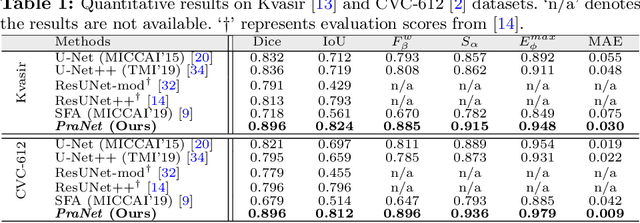
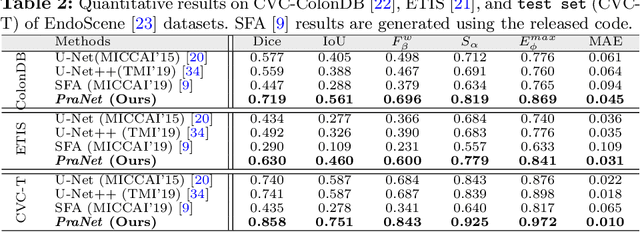

Abstract:Colonoscopy is an effective technique for detecting colorectal polyps, which are highly related to colorectal cancer. In clinical practice, segmenting polyps from colonoscopy images is of great importance since it provides valuable information for diagnosis and surgery. However, accurate polyp segmentation is a challenging task, for two major reasons: (i) the same type of polyps has a diversity of size, color and texture; and (ii) the boundary between a polyp and its surrounding mucosa is not sharp. To address these challenges, we propose a parallel reverse attention network (PraNet) for accurate polyp segmentation in colonoscopy images. Specifically, we first aggregate the features in high-level layers using a parallel partial decoder (PPD). Based on the combined feature, we then generate a global map as the initial guidance area for the following components. In addition, we mine the boundary cues using a reverse attention (RA) module, which is able to establish the relationship between areas and boundary cues. Thanks to the recurrent cooperation mechanism between areas and boundaries, our PraNet is capable of calibrating any misaligned predictions, improving the segmentation accuracy. Quantitative and qualitative evaluations on five challenging datasets across six metrics show that our PraNet improves the segmentation accuracy significantly, and presents a number of advantages in terms of generalizability, and real-time segmentation efficiency.
Surpassing Real-World Source Training Data: Random 3D Characters for Generalizable Person Re-Identification
Jun 23, 2020

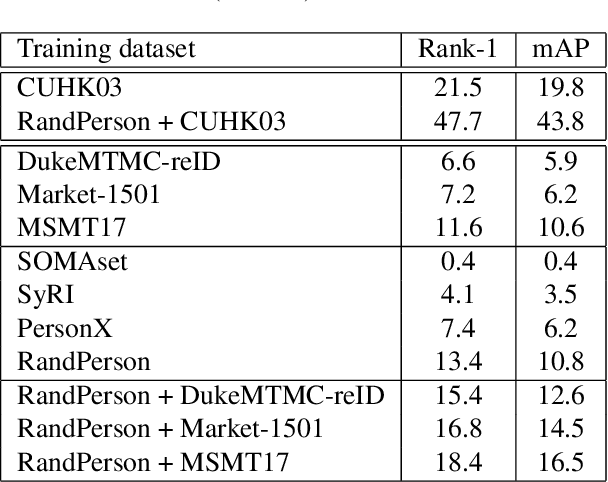
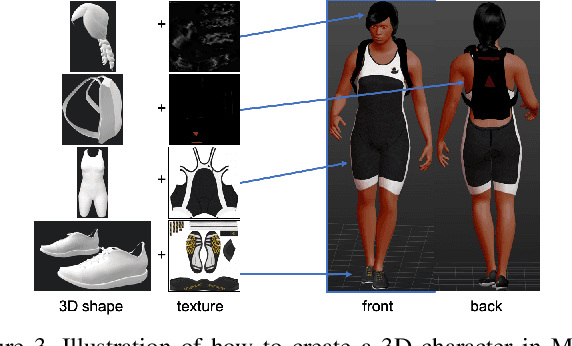
Abstract:Person re-identification has seen significant advancement in recent years. However, the ability of learned models to generalize to unknown target domains still remains limited. One possible reason for this is the lack of large-scale and diverse source training data, since manually labeling such a dataset is very expensive. To address this, we propose to automatically synthesize a large-scale person re-identification dataset following a set-up similar to real surveillance but with virtual environments, and then use the synthesized person images to train a generalizable person re-identification model. Specifically, we design a method to generate a large number of random UV texture maps and use them to create different 3D clothing models. Then, an automatic code is developed to randomly generate various different 3D characters with diverse clothes, races and attributes. Next, we simulate a number of different virtual environments using Unity3D, with customized camera networks similar to real surveillance systems, and import multiple 3D characters at the same time, with various movements and interactions along different paths through the camera networks. As a result, we obtain a virtual dataset, called RandPerson, with 1,756,759 person images of 8,000 identities. By training person re-identification models on these synthesized person images, we demonstrate, for the first time, that models trained on virtual data can generalize well to unseen target images, surpassing the models trained on various real-world datasets, including CUHK03, Market-1501, DukeMTMC-reID, and MSMT17. The RandPerson dataset will be released at https://github.com/VideoObjectSearch/RandPerson.
Pyramidal Convolution: Rethinking Convolutional Neural Networks for Visual Recognition
Jun 20, 2020
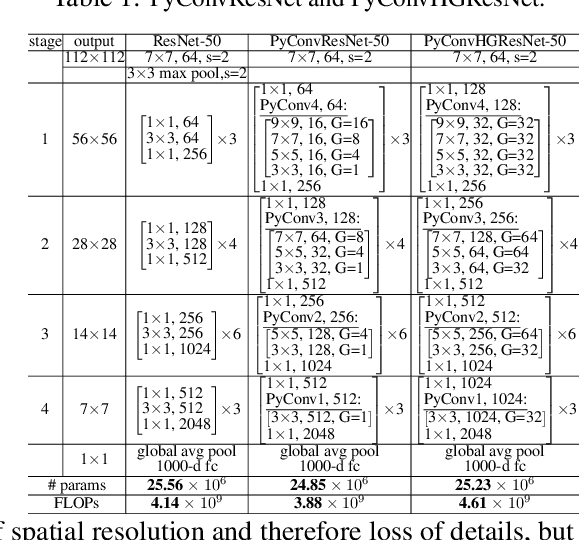
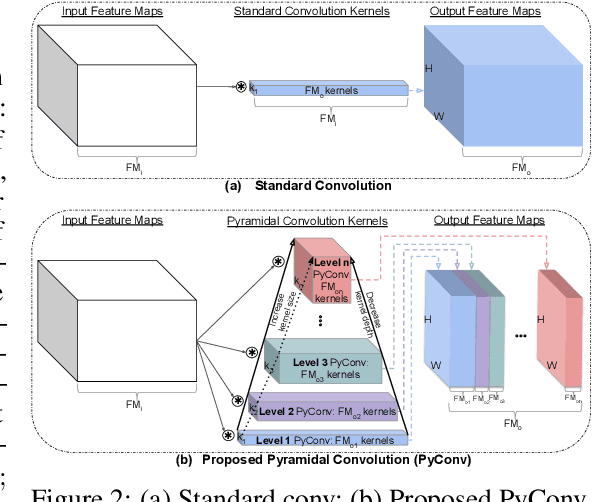

Abstract:This work introduces pyramidal convolution (PyConv), which is capable of processing the input at multiple filter scales. PyConv contains a pyramid of kernels, where each level involves different types of filters with varying size and depth, which are able to capture different levels of details in the scene. On top of these improved recognition capabilities, PyConv is also efficient and, with our formulation, it does not increase the computational cost and parameters compared to standard convolution. Moreover, it is very flexible and extensible, providing a large space of potential network architectures for different applications. PyConv has the potential to impact nearly every computer vision task and, in this work, we present different architectures based on PyConv for four main tasks on visual recognition: image classification, video action classification/recognition, object detection and semantic image segmentation/parsing. Our approach shows significant improvements over all these core tasks in comparison with the baselines. For instance, on image recognition, our 50-layers network outperforms in terms of recognition performance on ImageNet dataset its counterpart baseline ResNet with 152 layers, while having 2.39 times less parameters, 2.52 times lower computational complexity and more than 3 times less layers. On image segmentation, our novel framework sets a new state-of-the-art on the challenging ADE20K benchmark for scene parsing. Code is available at: https://github.com/iduta/pyconv
Learning to Learn Kernels with Variational Random Features
Jun 11, 2020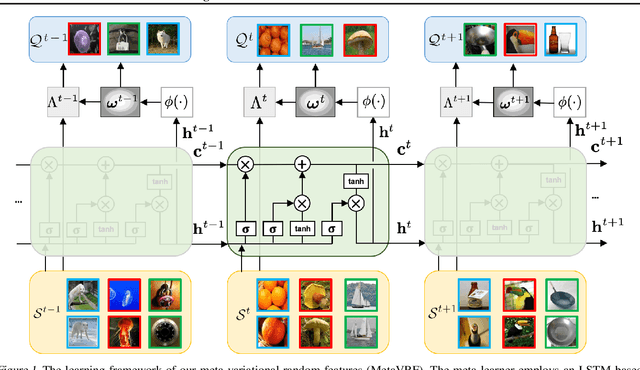
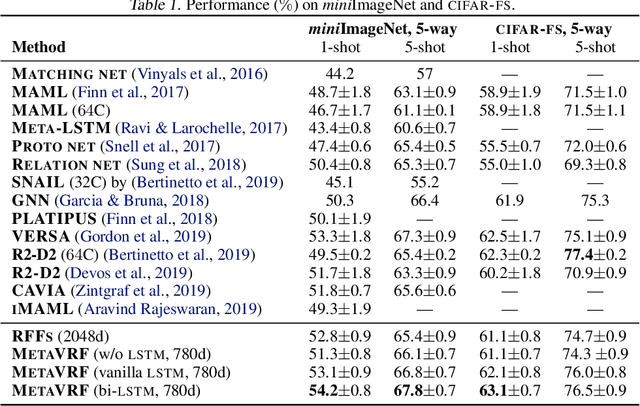
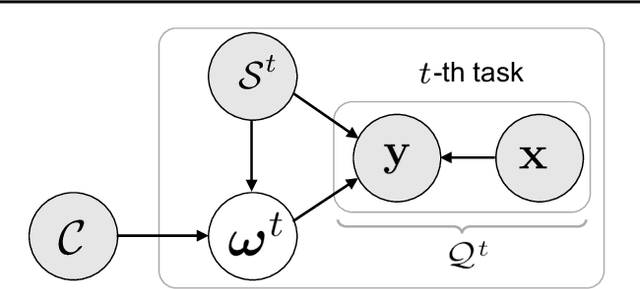
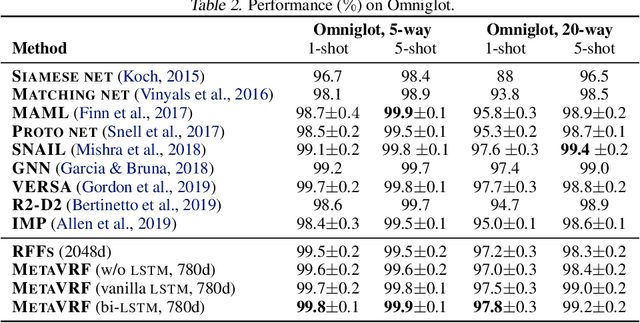
Abstract:In this work, we introduce kernels with random Fourier features in the meta-learning framework to leverage their strong few-shot learning ability. We propose meta variational random features (MetaVRF) to learn adaptive kernels for the base-learner, which is developed in a latent variable model by treating the random feature basis as the latent variable. We formulate the optimization of MetaVRF as a variational inference problem by deriving an evidence lower bound under the meta-learning framework. To incorporate shared knowledge from related tasks, we propose a context inference of the posterior, which is established by an LSTM architecture. The LSTM-based inference network can effectively integrate the context information of previous tasks with task-specific information, generating informative and adaptive features. The learned MetaVRF can produce kernels of high representational power with a relatively low spectral sampling rate and also enables fast adaptation to new tasks. Experimental results on a variety of few-shot regression and classification tasks demonstrate that MetaVRF delivers much better, or at least competitive, performance compared to existing meta-learning alternatives.
Attentive WaveBlock: Complementarity-enhanced Mutual Networks for Unsupervised Domain Adaptation in Person Re-identification
Jun 11, 2020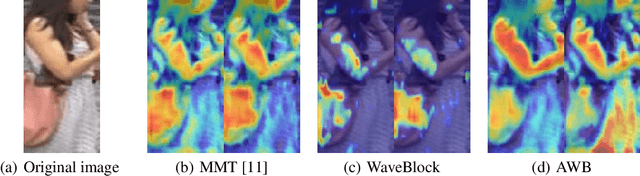
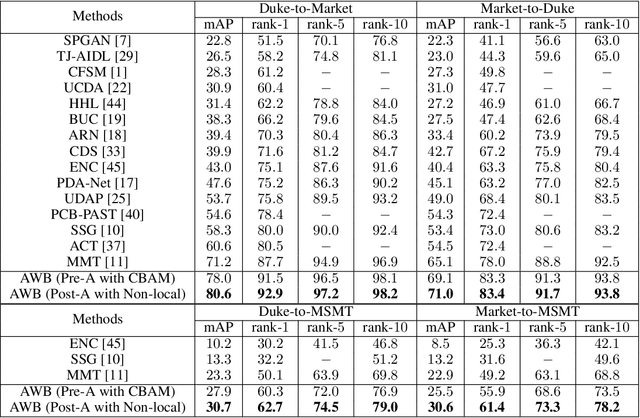
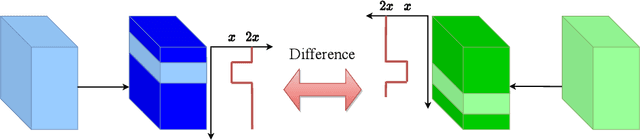
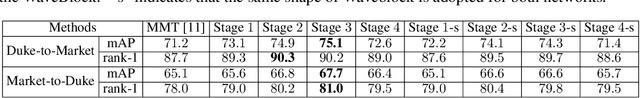
Abstract:Unsupervised domain adaptation (UDA) for person re-identification is challenging because of the huge gap between the source and target domain. A typical self-training method is to use pseudo-labels generated by clustering algorithms to iteratively optimize the model on the target domain. However, a drawback to this is that noisy pseudo-labels generally cause troubles in learning. To address this problem, a mutual learning method by dual networks has been developed to produce reliable soft labels. However, as the two neural networks gradually converge, their complementarity is weakened and they likely become biased towards the same kind of noise. In this paper, we propose a novel light-weight module, the Attentive WaveBlock (AWB), which can be integrated into the dual networks of mutual learning to enhance the complementarity and further depress noise in the pseudo-labels. Specifically, we first introduce a parameter-free module, the WaveBlock, which creates a difference between two networks by waving blocks of feature maps differently. Then, an attention mechanism is leveraged to enlarge the difference created and discover more complementary features. Furthermore, two kinds of combination strategies, i.e. pre-attention and post-attention, are explored. Experiments demonstrate that the proposed method achieves state-of-the-art performance with significant improvements of 9.4%, 5.9%, 7.4%, and 7.7% in mAP on Duke-to-Market, Market-to-Duke, Duke-to-MSMT, and Market-to-MSMT UDA tasks, respectively.
M2Net: Multi-modal Multi-channel Network for Overall Survival Time Prediction of Brain Tumor Patients
Jun 01, 2020
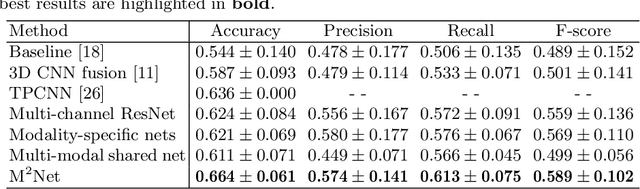
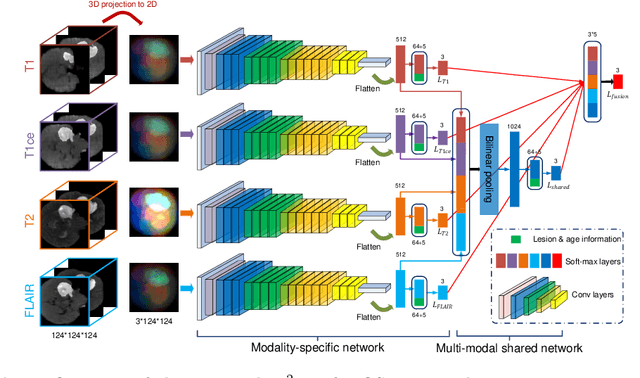
Abstract:Early and accurate prediction of overall survival (OS) time can help to obtain better treatment planning for brain tumor patients. Although many OS time prediction methods have been developed and obtain promising results, there are still several issues. First, conventional prediction methods rely on radiomic features at the local lesion area of a magnetic resonance (MR) volume, which may not represent the full image or model complex tumor patterns. Second, different types of scanners (i.e., multi-modal data) are sensitive to different brain regions, which makes it challenging to effectively exploit the complementary information across multiple modalities and also preserve the modality-specific properties. Third, existing methods focus on prediction models, ignoring complex data-to-label relationships. To address the above issues, we propose an end-to-end OS time prediction model; namely, Multi-modal Multi-channel Network (M2Net). Specifically, we first project the 3D MR volume onto 2D images in different directions, which reduces computational costs, while preserving important information and enabling pre-trained models to be transferred from other tasks. Then, we use a modality-specific network to extract implicit and high-level features from different MR scans. A multi-modal shared network is built to fuse these features using a bilinear pooling model, exploiting their correlations to provide complementary information. Finally, we integrate the outputs from each modality-specific network and the multi-modal shared network to generate the final prediction result. Experimental results demonstrate the superiority of our M2Net model over other methods.
Inf-Net: Automatic COVID-19 Lung Infection Segmentation from CT Images
May 21, 2020Abstract:Coronavirus Disease 2019 (COVID-19) spread globally in early 2020, causing the world to face an existential health crisis. Automated detection of lung infections from computed tomography (CT) images offers a great potential to augment the traditional healthcare strategy for tackling COVID-19. However, segmenting infected regions from CT slices faces several challenges, including high variation in infection characteristics, and low intensity contrast between infections and normal tissues. Further, collecting a large amount of data is impractical within a short time period, inhibiting the training of a deep model. To address these challenges, a novel COVID-19 Lung Infection Segmentation Deep Network (Inf-Net) is proposed to automatically identify infected regions from chest CT slices. In our Inf-Net, a parallel partial decoder is used to aggregate the high-level features and generate a global map. Then, the implicit reverse attention and explicit edge-attention are utilized to model the boundaries and enhance the representations. Moreover, to alleviate the shortage of labeled data, we present a semi-supervised segmentation framework based on a randomly selected propagation strategy, which only requires a few labeled images and leverages primarily unlabeled data. Our semi-supervised framework can improve the learning ability and achieve a higher performance. Extensive experiments on our COVID-SemiSeg and real CT volumes demonstrate that the proposed Inf-Net outperforms most cutting-edge segmentation models and advances the state-of-the-art performance.
Understanding and Correcting Low-quality Retinal Fundus Images for Clinical Analysis
May 12, 2020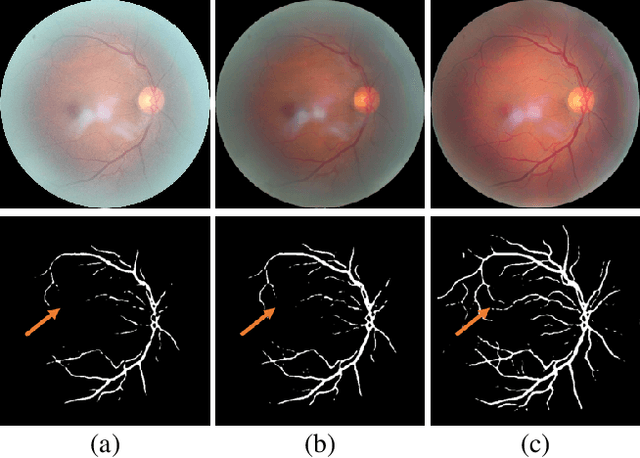
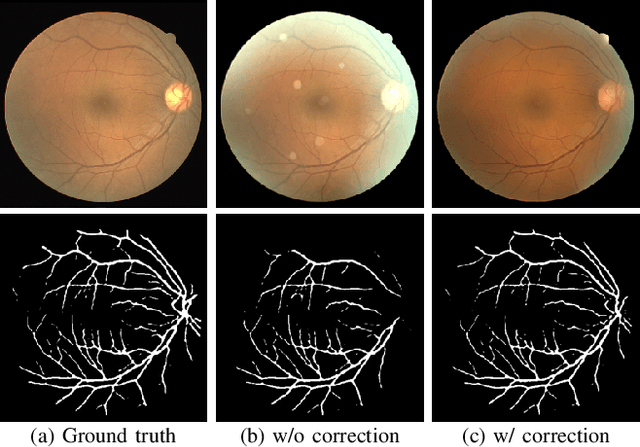
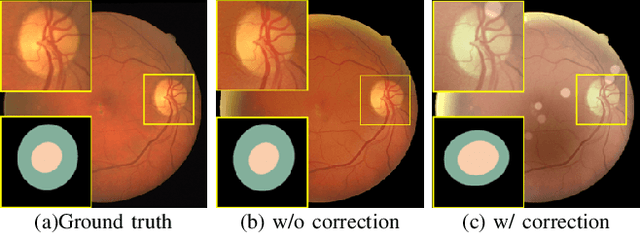
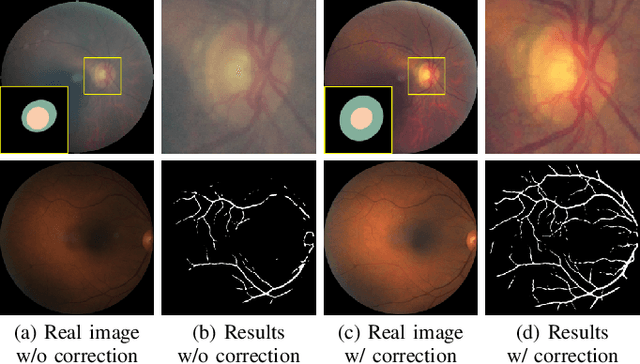
Abstract:Retinal fundus images are widely used for clinical screening and diagnosis of eye diseases. However, fundus images captured by operators with various levels of experiences have a large variation in quality. Low-quality fundus images increase the uncertainty in clinical observation and lead to a risk of misdiagnosis. Due to the special optical beam of fundus imaging and retinal structure, the natural image enhancement methods cannot be utilized directly. In this paper, we first analyze the ophthalmoscope imaging system and model the reliable degradation of major inferior-quality factors, including uneven illumination, blur, and artifacts. Then, based on the degradation model, a clinical-oriented fundus enhancement network~(cofe-Net)~is proposed to suppress the global degradation factors, and simultaneously preserve anatomical retinal structures and pathological characteristics for clinical observation and analysis. Experiments on both synthetic and real fundus images demonstrate that our algorithm effectively corrects low-quality fundus images without losing retinal details. Moreover, we also show that the fundus correction method can benefit medical image analysis applications, e.g, retinal vessel segmentation and optic disc/cup detection.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge