Lijun Wu
Lost in Tokenization: Context as the Key to Unlocking Biomolecular Understanding in Scientific LLMs
Oct 27, 2025Abstract:Scientific Large Language Models (Sci-LLMs) have emerged as a promising frontier for accelerating biological discovery. However, these models face a fundamental challenge when processing raw biomolecular sequences: the tokenization dilemma. Whether treating sequences as a specialized language, risking the loss of functional motif information, or as a separate modality, introducing formidable alignment challenges, current strategies fundamentally limit their reasoning capacity. We challenge this sequence-centric paradigm by positing that a more effective strategy is to provide Sci-LLMs with high-level structured context derived from established bioinformatics tools, thereby bypassing the need to interpret low-level noisy sequence data directly. Through a systematic comparison of leading Sci-LLMs on biological reasoning tasks, we tested three input modes: sequence-only, context-only, and a combination of both. Our findings are striking: the context-only approach consistently and substantially outperforms all other modes. Even more revealing, the inclusion of the raw sequence alongside its high-level context consistently degrades performance, indicating that raw sequences act as informational noise, even for models with specialized tokenization schemes. These results suggest that the primary strength of existing Sci-LLMs lies not in their nascent ability to interpret biomolecular syntax from scratch, but in their profound capacity for reasoning over structured, human-readable knowledge. Therefore, we argue for reframing Sci-LLMs not as sequence decoders, but as powerful reasoning engines over expert knowledge. This work lays the foundation for a new class of hybrid scientific AI agents, repositioning the developmental focus from direct sequence interpretation towards high-level knowledge synthesis. The code is available at github.com/opendatalab-raise-dev/CoKE.
MinerU2.5: A Decoupled Vision-Language Model for Efficient High-Resolution Document Parsing
Sep 26, 2025Abstract:We introduce MinerU2.5, a 1.2B-parameter document parsing vision-language model that achieves state-of-the-art recognition accuracy while maintaining exceptional computational efficiency. Our approach employs a coarse-to-fine, two-stage parsing strategy that decouples global layout analysis from local content recognition. In the first stage, the model performs efficient layout analysis on downsampled images to identify structural elements, circumventing the computational overhead of processing high-resolution inputs. In the second stage, guided by the global layout, it performs targeted content recognition on native-resolution crops extracted from the original image, preserving fine-grained details in dense text, complex formulas, and tables. To support this strategy, we developed a comprehensive data engine that generates diverse, large-scale training corpora for both pretraining and fine-tuning. Ultimately, MinerU2.5 demonstrates strong document parsing ability, achieving state-of-the-art performance on multiple benchmarks, surpassing both general-purpose and domain-specific models across various recognition tasks, while maintaining significantly lower computational overhead.
Middo: Model-Informed Dynamic Data Optimization for Enhanced LLM Fine-Tuning via Closed-Loop Learning
Aug 29, 2025



Abstract:Supervised Fine-Tuning (SFT) Large Language Models (LLM) fundamentally rely on high-quality training data. While data selection and data synthesis are two common strategies to improve data quality, existing approaches often face limitations in static dataset curation that fail to adapt to evolving model capabilities. In this paper, we introduce Middo, a self-evolving Model-informed dynamic data optimization framework that uses model-aware data selection and context-preserving data refinement. Unlike conventional one-off filtering/synthesis methods, our framework establishes a closed-loop optimization system: (1) A self-referential diagnostic module proactively identifies suboptimal samples through tri-axial model signals - loss patterns (complexity), embedding cluster dynamics (diversity), and self-alignment scores (quality); (2) An adaptive optimization engine then transforms suboptimal samples into pedagogically valuable training points while preserving semantic integrity; (3) This optimization process continuously evolves with model capability through dynamic learning principles. Experiments on multiple benchmarks demonstrate that our \method consistently enhances the quality of seed data and boosts LLM's performance with improving accuracy by 7.15% on average while maintaining the original dataset scale. This work establishes a new paradigm for sustainable LLM training through dynamic human-AI co-evolution of data and models. Our datasets, models, and code are coming soon.
A Survey of Scientific Large Language Models: From Data Foundations to Agent Frontiers
Aug 28, 2025



Abstract:Scientific Large Language Models (Sci-LLMs) are transforming how knowledge is represented, integrated, and applied in scientific research, yet their progress is shaped by the complex nature of scientific data. This survey presents a comprehensive, data-centric synthesis that reframes the development of Sci-LLMs as a co-evolution between models and their underlying data substrate. We formulate a unified taxonomy of scientific data and a hierarchical model of scientific knowledge, emphasizing the multimodal, cross-scale, and domain-specific challenges that differentiate scientific corpora from general natural language processing datasets. We systematically review recent Sci-LLMs, from general-purpose foundations to specialized models across diverse scientific disciplines, alongside an extensive analysis of over 270 pre-/post-training datasets, showing why Sci-LLMs pose distinct demands -- heterogeneous, multi-scale, uncertainty-laden corpora that require representations preserving domain invariance and enabling cross-modal reasoning. On evaluation, we examine over 190 benchmark datasets and trace a shift from static exams toward process- and discovery-oriented assessments with advanced evaluation protocols. These data-centric analyses highlight persistent issues in scientific data development and discuss emerging solutions involving semi-automated annotation pipelines and expert validation. Finally, we outline a paradigm shift toward closed-loop systems where autonomous agents based on Sci-LLMs actively experiment, validate, and contribute to a living, evolving knowledge base. Collectively, this work provides a roadmap for building trustworthy, continually evolving artificial intelligence (AI) systems that function as a true partner in accelerating scientific discovery.
InternVL3.5: Advancing Open-Source Multimodal Models in Versatility, Reasoning, and Efficiency
Aug 25, 2025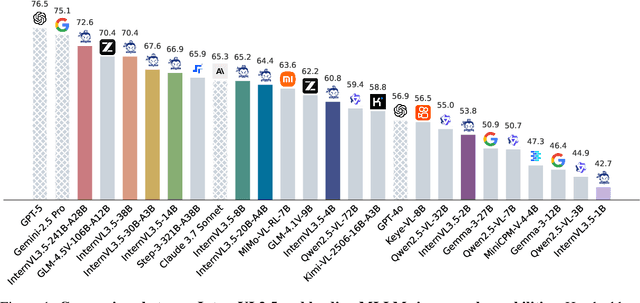
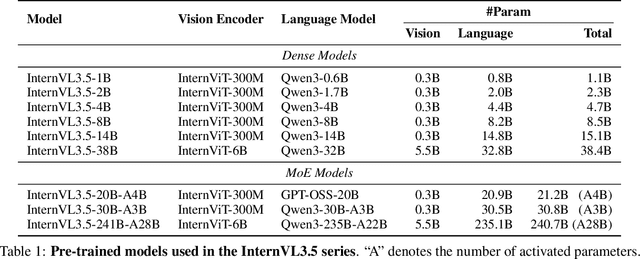
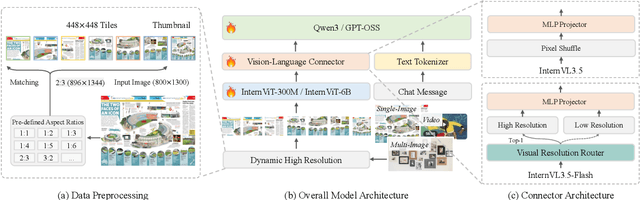
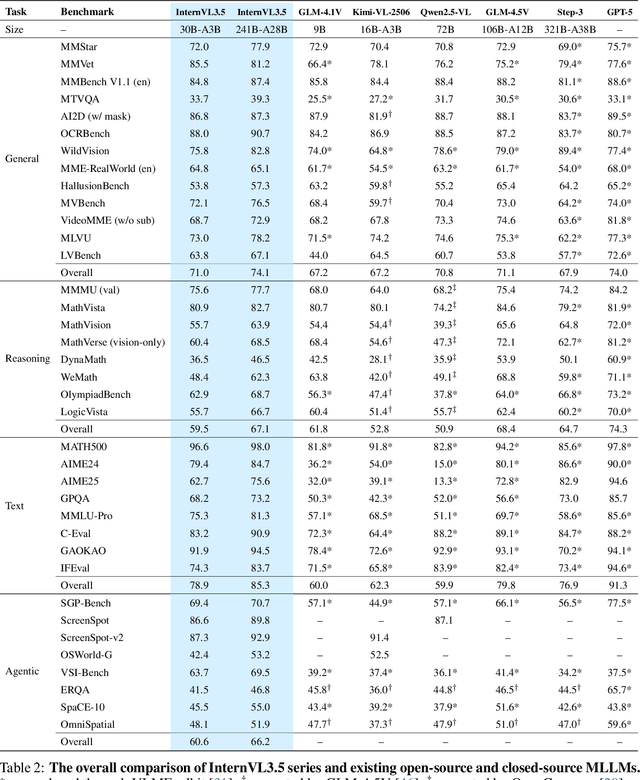
Abstract:We introduce InternVL 3.5, a new family of open-source multimodal models that significantly advances versatility, reasoning capability, and inference efficiency along the InternVL series. A key innovation is the Cascade Reinforcement Learning (Cascade RL) framework, which enhances reasoning through a two-stage process: offline RL for stable convergence and online RL for refined alignment. This coarse-to-fine training strategy leads to substantial improvements on downstream reasoning tasks, e.g., MMMU and MathVista. To optimize efficiency, we propose a Visual Resolution Router (ViR) that dynamically adjusts the resolution of visual tokens without compromising performance. Coupled with ViR, our Decoupled Vision-Language Deployment (DvD) strategy separates the vision encoder and language model across different GPUs, effectively balancing computational load. These contributions collectively enable InternVL3.5 to achieve up to a +16.0\% gain in overall reasoning performance and a 4.05$\times$ inference speedup compared to its predecessor, i.e., InternVL3. In addition, InternVL3.5 supports novel capabilities such as GUI interaction and embodied agency. Notably, our largest model, i.e., InternVL3.5-241B-A28B, attains state-of-the-art results among open-source MLLMs across general multimodal, reasoning, text, and agentic tasks -- narrowing the performance gap with leading commercial models like GPT-5. All models and code are publicly released.
Can One Domain Help Others? A Data-Centric Study on Multi-Domain Reasoning via Reinforcement Learning
Jul 23, 2025Abstract:Reinforcement Learning with Verifiable Rewards (RLVR) has emerged as a powerful paradigm for enhancing the reasoning capabilities of LLMs. Existing research has predominantly concentrated on isolated reasoning domains such as mathematical problem-solving, coding tasks, or logical reasoning. However, real world reasoning scenarios inherently demand an integrated application of multiple cognitive skills. Despite this, the interplay among these reasoning skills under reinforcement learning remains poorly understood. To bridge this gap, we present a systematic investigation of multi-domain reasoning within the RLVR framework, explicitly focusing on three primary domains: mathematical reasoning, code generation, and logical puzzle solving. We conduct a comprehensive study comprising four key components: (1) Leveraging the GRPO algorithm and the Qwen-2.5-7B model family, our study thoroughly evaluates the models' in-domain improvements and cross-domain generalization capabilities when trained on single-domain datasets. (2) Additionally, we examine the intricate interactions including mutual enhancements and conflicts that emerge during combined cross-domain training. (3) To further understand the influence of SFT on RL, we also analyze and compare performance differences between base and instruct models under identical RL configurations. (4) Furthermore, we delve into critical RL training details, systematically exploring the impacts of curriculum learning strategies, variations in reward design, and language-specific factors. Through extensive experiments, our results offer significant insights into the dynamics governing domain interactions, revealing key factors influencing both specialized and generalizable reasoning performance. These findings provide valuable guidance for optimizing RL methodologies to foster comprehensive, multi-domain reasoning capabilities in LLMs.
GTR-CoT: Graph Traversal as Visual Chain of Thought for Molecular Structure Recognition
Jun 09, 2025



Abstract:Optical Chemical Structure Recognition (OCSR) is crucial for digitizing chemical knowledge by converting molecular images into machine-readable formats. While recent vision-language models (VLMs) have shown potential in this task, their image-captioning approach often struggles with complex molecular structures and inconsistent annotations. To overcome these challenges, we introduce GTR-Mol-VLM, a novel framework featuring two key innovations: (1) the \textit{Graph Traversal as Visual Chain of Thought} mechanism that emulates human reasoning by incrementally parsing molecular graphs through sequential atom-bond predictions, and (2) the data-centric principle of \textit{Faithfully Recognize What You've Seen}, which addresses the mismatch between abbreviated structures in images and their expanded annotations. To support model development, we constructed GTR-CoT-1.3M, a large-scale instruction-tuning dataset with meticulously corrected annotations, and introduced MolRec-Bench, the first benchmark designed for a fine-grained evaluation of graph-parsing accuracy in OCSR. Comprehensive experiments demonstrate that GTR-Mol-VLM achieves superior results compared to specialist models, chemistry-domain VLMs, and commercial general-purpose VLMs. Notably, in scenarios involving molecular images with functional group abbreviations, GTR-Mol-VLM outperforms the second-best baseline by approximately 14 percentage points, both in SMILES-based and graph-based metrics. We hope that this work will drive OCSR technology to more effectively meet real-world needs, thereby advancing the fields of cheminformatics and AI for Science. We will release GTR-CoT at https://github.com/opendatalab/GTR-CoT.
Evaluating Large Language Model with Knowledge Oriented Language Specific Simple Question Answering
May 22, 2025
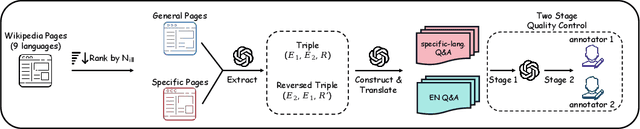


Abstract:We introduce KoLasSimpleQA, the first benchmark evaluating the multilingual factual ability of Large Language Models (LLMs). Inspired by existing research, we created the question set with features such as single knowledge point coverage, absolute objectivity, unique answers, and temporal stability. These questions enable efficient evaluation using the LLM-as-judge paradigm, testing both the LLMs' factual memory and self-awareness ("know what they don't know"). KoLasSimpleQA expands existing research in two key dimensions: (1) Breadth (Multilingual Coverage): It includes 9 languages, supporting global applicability evaluation. (2) Depth (Dual Domain Design): It covers both the general domain (global facts) and the language-specific domain (such as history, culture, and regional traditions) for a comprehensive assessment of multilingual capabilities. We evaluated mainstream LLMs, including traditional LLM and emerging Large Reasoning Models. Results show significant performance differences between the two domains, particularly in performance metrics, ranking, calibration, and robustness. This highlights the need for targeted evaluation and optimization in multilingual contexts. We hope KoLasSimpleQA will help the research community better identify LLM capability boundaries in multilingual contexts and provide guidance for model optimization. We will release KoLasSimpleQA at https://github.com/opendatalab/KoLasSimpleQA .
IDEAL: Data Equilibrium Adaptation for Multi-Capability Language Model Alignment
May 19, 2025



Abstract:Large Language Models (LLMs) have achieved impressive performance through Supervised Fine-tuning (SFT) on diverse instructional datasets. When training on multiple capabilities simultaneously, the mixture training dataset, governed by volumes of data from different domains, is a critical factor that directly impacts the final model's performance. Unlike many studies that focus on enhancing the quality of training datasets through data selection methods, few works explore the intricate relationship between the compositional quantity of mixture training datasets and the emergent capabilities of LLMs. Given the availability of a high-quality multi-domain training dataset, understanding the impact of data from each domain on the model's overall capabilities is crucial for preparing SFT data and training a well-balanced model that performs effectively across diverse domains. In this work, we introduce IDEAL, an innovative data equilibrium adaptation framework designed to effectively optimize volumes of data from different domains within mixture SFT datasets, thereby enhancing the model's alignment and performance across multiple capabilities. IDEAL employs a gradient-based approach to iteratively refine the training data distribution, dynamically adjusting the volumes of domain-specific data based on their impact on downstream task performance. By leveraging this adaptive mechanism, IDEAL ensures a balanced dataset composition, enabling the model to achieve robust generalization and consistent proficiency across diverse tasks. Experiments across different capabilities demonstrate that IDEAL outperforms conventional uniform data allocation strategies, achieving a comprehensive improvement of approximately 7% in multi-task evaluation scores.
CipherBank: Exploring the Boundary of LLM Reasoning Capabilities through Cryptography Challenges
Apr 27, 2025



Abstract:Large language models (LLMs) have demonstrated remarkable capabilities, especially the recent advancements in reasoning, such as o1 and o3, pushing the boundaries of AI. Despite these impressive achievements in mathematics and coding, the reasoning abilities of LLMs in domains requiring cryptographic expertise remain underexplored. In this paper, we introduce CipherBank, a comprehensive benchmark designed to evaluate the reasoning capabilities of LLMs in cryptographic decryption tasks. CipherBank comprises 2,358 meticulously crafted problems, covering 262 unique plaintexts across 5 domains and 14 subdomains, with a focus on privacy-sensitive and real-world scenarios that necessitate encryption. From a cryptographic perspective, CipherBank incorporates 3 major categories of encryption methods, spanning 9 distinct algorithms, ranging from classical ciphers to custom cryptographic techniques. We evaluate state-of-the-art LLMs on CipherBank, e.g., GPT-4o, DeepSeek-V3, and cutting-edge reasoning-focused models such as o1 and DeepSeek-R1. Our results reveal significant gaps in reasoning abilities not only between general-purpose chat LLMs and reasoning-focused LLMs but also in the performance of current reasoning-focused models when applied to classical cryptographic decryption tasks, highlighting the challenges these models face in understanding and manipulating encrypted data. Through detailed analysis and error investigations, we provide several key observations that shed light on the limitations and potential improvement areas for LLMs in cryptographic reasoning. These findings underscore the need for continuous advancements in LLM reasoning capabilities.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge