Xuhai Xu
CoDaS: AI Co-Data-Scientist for Biomarker Discovery via Wearable Sensors
Apr 16, 2026Abstract:Scientific discovery in digital health requires converting continuous physiological signals from wearable devices into clinically actionable biomarkers. We introduce CoDaS (AI Co-Data-Scientist), a multi-agent system that structures biomarker discovery as an iterative process combining hypothesis generation, statistical analysis, adversarial validation, and literature-grounded reasoning with human oversight using large-scale wearable datasets. Across three cohorts totaling 9,279 participant-observations, CoDaS identified 41 candidate digital biomarkers for mental health and 25 for metabolic outcomes, each subjected to an internal validation battery spanning replication, stability, robustness, and discriminative power. Across two independent depression cohorts, CoDaS surfaced circadian instability-related features in both datasets, reflected in sleep duration variability (DWB, ρ= 0.252, p < 0.001) and sleep onset variability (GLOBEM, ρ= 0.126, p < 0.001). In a metabolic cohort, CoDaS derived a cardiovascular fitness index (steps/resting heart rate; ρ= -0.374, p < 0.001), and recovered established clinical associations, including the hepatic function ratio (AST/ALT; ρ= -0.375, p < 0.001), a known correlate of insulin resistance. Incorporating CoDaS-derived features alongside demographic variables led to modest but consistent improvements in predictive performance, with cross-validated ΔR^2 increases of 0.040 for depression and 0.021 for insulin resistance. These findings suggest that CoDaS enables systematic and traceable hypothesis generation and prioritization for biomarker discovery from large-scale wearable data.
EgoTL: Egocentric Think-Aloud Chains for Long-Horizon Tasks
Apr 10, 2026Abstract:Large foundation models have made significant advances in embodied intelligence, enabling synthesis and reasoning over egocentric input for household tasks. However, VLM-based auto-labeling is often noisy because the primary data sources lack accurate human action labels, chain-of-thought (CoT), and spatial annotations; these errors are amplified during long-horizon spatial instruction following. These issues stem from insufficient coverage of minute-long, daily household planning tasks and from inaccurate spatial grounding. As a result, VLM reasoning chains and world-model synthesis can hallucinate objects, skip steps, or fail to respect real-world physical attributes. To address these gaps, we introduce EgoTL. EgoTL builds a think-aloud capture pipeline for egocentric data. It uses a say-before-act protocol to record step-by-step goals and spoken reasoning with word-level timestamps, then calibrates physical properties with metric-scale spatial estimators, a memory-bank walkthrough for scene context, and clip-level tags for navigation instructions and detailed manipulation actions. With EgoTL, we are able to benchmark VLMs and World Models on six task dimensions from three layers and long-horizon generation over minute-long sequences across over 100 daily household tasks. We find that foundation models still fall short as egocentric assistants or open-world simulators. Finally, we finetune foundation models with human CoT aligned with metric labels on the training split of EgoTL, which improves long-horizon planning and reasoning, step-wise reasoning, instruction following, and spatial grounding.
Rethinking Health Agents: From Siloed AI to Collaborative Decision Mediators
Mar 26, 2026Abstract:Large language model based health agents are increasingly used by health consumers and clinicians to interpret health information and guide health decisions. However, most AI systems in healthcare operate in siloed configurations, supporting individual users rather than the multi-stakeholder relationships central to healthcare. Such use can fragment understanding and exacerbate misalignment among patients, caregivers, and clinicians. We reframe AI not as a standalone assistant, but as a collaborator embedded within multi-party care interactions. Through a clinically validated fictional pediatric chronic kidney disease case study, we show that breakdowns in adherence stem from fragmented situational awareness and misaligned goals, and that siloed use of general-purpose AI tools does little to address these collaboration gaps. We propose a conceptual framework for designing AI collaborators that surface contextual information, reconcile mental models, and scaffold shared understanding while preserving human decision authority.
Toward Global Large Language Models in Medicine
Jan 05, 2026Abstract:Despite continuous advances in medical technology, the global distribution of health care resources remains uneven. The development of large language models (LLMs) has transformed the landscape of medicine and holds promise for improving health care quality and expanding access to medical information globally. However, existing LLMs are primarily trained on high-resource languages, limiting their applicability in global medical scenarios. To address this gap, we constructed GlobMed, a large multilingual medical dataset, containing over 500,000 entries spanning 12 languages, including four low-resource languages. Building on this, we established GlobMed-Bench, which systematically assesses 56 state-of-the-art proprietary and open-weight LLMs across multiple multilingual medical tasks, revealing significant performance disparities across languages, particularly for low-resource languages. Additionally, we introduced GlobMed-LLMs, a suite of multilingual medical LLMs trained on GlobMed, with parameters ranging from 1.7B to 8B. GlobMed-LLMs achieved an average performance improvement of over 40% relative to baseline models, with a more than threefold increase in performance on low-resource languages. Together, these resources provide an important foundation for advancing the equitable development and application of LLMs globally, enabling broader language communities to benefit from technological advances.
Towards a Science of Scaling Agent Systems
Dec 17, 2025Abstract:Agents, language model-based systems that are capable of reasoning, planning, and acting are becoming the dominant paradigm for real-world AI applications. Despite this widespread adoption, the principles that determine their performance remain underexplored. We address this by deriving quantitative scaling principles for agent systems. We first formalize a definition for agentic evaluation and characterize scaling laws as the interplay between agent quantity, coordination structure, model capability, and task properties. We evaluate this across four benchmarks: Finance-Agent, BrowseComp-Plus, PlanCraft, and Workbench. With five canonical agent architectures (Single-Agent and four Multi-Agent Systems: Independent, Centralized, Decentralized, Hybrid), instantiated across three LLM families, we perform a controlled evaluation spanning 180 configurations. We derive a predictive model using coordination metrics, that achieves cross-validated R^2=0.524, enabling prediction on unseen task domains. We identify three effects: (1) a tool-coordination trade-off: under fixed computational budgets, tool-heavy tasks suffer disproportionately from multi-agent overhead. (2) a capability saturation: coordination yields diminishing or negative returns once single-agent baselines exceed ~45%. (3) topology-dependent error amplification: independent agents amplify errors 17.2x, while centralized coordination contains this to 4.4x. Centralized coordination improves performance by 80.8% on parallelizable tasks, while decentralized coordination excels on web navigation (+9.2% vs. +0.2%). Yet for sequential reasoning tasks, every multi-agent variants degraded performance by 39-70%. The framework predicts the optimal coordination strategy for 87% of held-out configurations. Out-of-sample validation on GPT-5.2, achieves MAE=0.071 and confirms four of five scaling principles generalize to unseen frontier models.
Explainable AI as a Double-Edged Sword in Dermatology: The Impact on Clinicians versus The Public
Dec 14, 2025Abstract:Artificial intelligence (AI) is increasingly permeating healthcare, from physician assistants to consumer applications. Since AI algorithm's opacity challenges human interaction, explainable AI (XAI) addresses this by providing AI decision-making insight, but evidence suggests XAI can paradoxically induce over-reliance or bias. We present results from two large-scale experiments (623 lay people; 153 primary care physicians, PCPs) combining a fairness-based diagnosis AI model and different XAI explanations to examine how XAI assistance, particularly multimodal large language models (LLMs), influences diagnostic performance. AI assistance balanced across skin tones improved accuracy and reduced diagnostic disparities. However, LLM explanations yielded divergent effects: lay users showed higher automation bias - accuracy boosted when AI was correct, reduced when AI erred - while experienced PCPs remained resilient, benefiting irrespective of AI accuracy. Presenting AI suggestions first also led to worse outcomes when the AI was incorrect for both groups. These findings highlight XAI's varying impact based on expertise and timing, underscoring LLMs as a "double-edged sword" in medical AI and informing future human-AI collaborative system design.
A Multi-Stage Large Language Model Framework for Extracting Suicide-Related Social Determinants of Health
Aug 07, 2025Abstract:Background: Understanding social determinants of health (SDoH) factors contributing to suicide incidents is crucial for early intervention and prevention. However, data-driven approaches to this goal face challenges such as long-tailed factor distributions, analyzing pivotal stressors preceding suicide incidents, and limited model explainability. Methods: We present a multi-stage large language model framework to enhance SDoH factor extraction from unstructured text. Our approach was compared to other state-of-the-art language models (i.e., pre-trained BioBERT and GPT-3.5-turbo) and reasoning models (i.e., DeepSeek-R1). We also evaluated how the model's explanations help people annotate SDoH factors more quickly and accurately. The analysis included both automated comparisons and a pilot user study. Results: We show that our proposed framework demonstrated performance boosts in the overarching task of extracting SDoH factors and in the finer-grained tasks of retrieving relevant context. Additionally, we show that fine-tuning a smaller, task-specific model achieves comparable or better performance with reduced inference costs. The multi-stage design not only enhances extraction but also provides intermediate explanations, improving model explainability. Conclusions: Our approach improves both the accuracy and transparency of extracting suicide-related SDoH from unstructured texts. These advancements have the potential to support early identification of individuals at risk and inform more effective prevention strategies.
SensorLM: Learning the Language of Wearable Sensors
Jun 10, 2025Abstract:We present SensorLM, a family of sensor-language foundation models that enable wearable sensor data understanding with natural language. Despite its pervasive nature, aligning and interpreting sensor data with language remains challenging due to the lack of paired, richly annotated sensor-text descriptions in uncurated, real-world wearable data. We introduce a hierarchical caption generation pipeline designed to capture statistical, structural, and semantic information from sensor data. This approach enabled the curation of the largest sensor-language dataset to date, comprising over 59.7 million hours of data from more than 103,000 people. Furthermore, SensorLM extends prominent multimodal pretraining architectures (e.g., CLIP, CoCa) and recovers them as specific variants within a generic architecture. Extensive experiments on real-world tasks in human activity analysis and healthcare verify the superior performance of SensorLM over state-of-the-art in zero-shot recognition, few-shot learning, and cross-modal retrieval. SensorLM also demonstrates intriguing capabilities including scaling behaviors, label efficiency, sensor captioning, and zero-shot generalization to unseen tasks.
RADAR: Benchmarking Language Models on Imperfect Tabular Data
Jun 09, 2025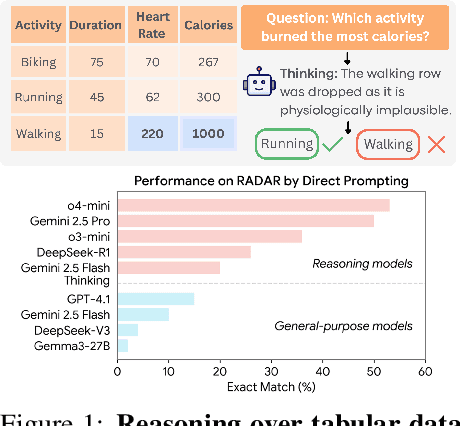
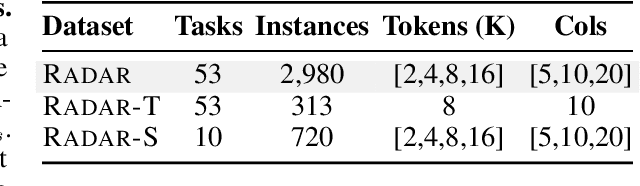
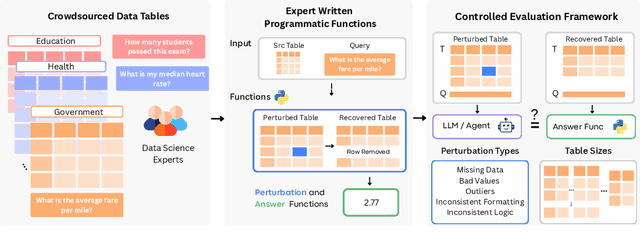
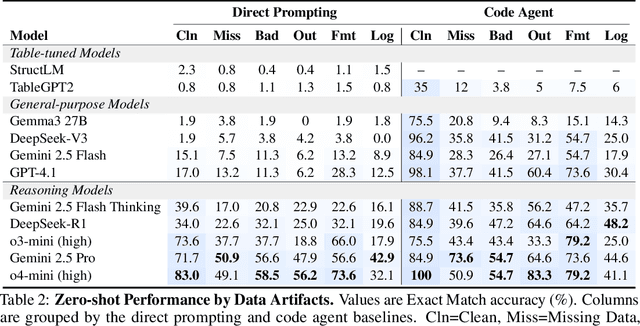
Abstract:Language models (LMs) are increasingly being deployed to perform autonomous data analyses. However, their data awareness -- the ability to recognize, reason over, and appropriately handle data artifacts such as missing values, outliers, and logical inconsistencies -- remains underexplored. These artifacts are especially common in real-world tabular data and, if mishandled, can significantly compromise the validity of analytical conclusions. To address this gap, we present RADAR, a benchmark for systematically evaluating data-aware reasoning on tabular data. We develop a framework to simulate data artifacts via programmatic perturbations to enable targeted evaluation of model behavior. RADAR comprises 2980 table query pairs, grounded in real-world data spanning 9 domains and 5 data artifact types. In addition to evaluating artifact handling, RADAR systematically varies table size to study how reasoning performance holds when increasing table size. Our evaluation reveals that, despite decent performance on tables without data artifacts, frontier models degrade significantly when data artifacts are introduced, exposing critical gaps in their capacity for robust, data-aware analysis. Designed to be flexible and extensible, RADAR supports diverse perturbation types and controllable table sizes, offering a valuable resource for advancing tabular reasoning.
LSM-2: Learning from Incomplete Wearable Sensor Data
Jun 05, 2025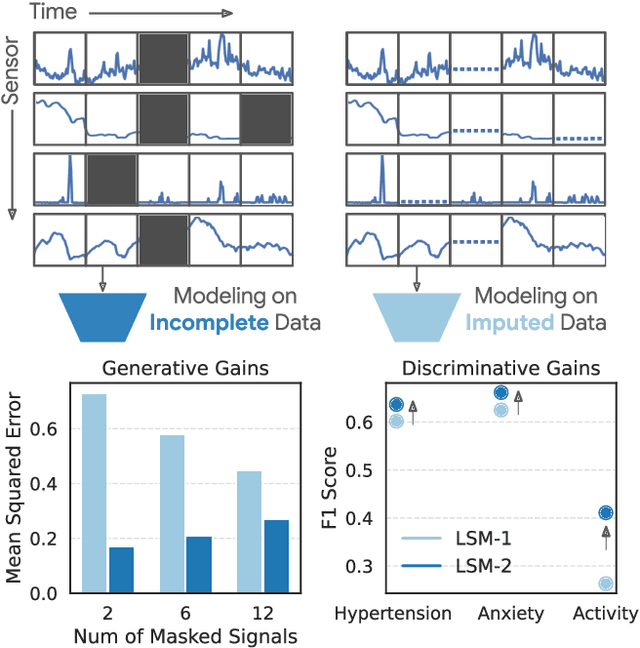
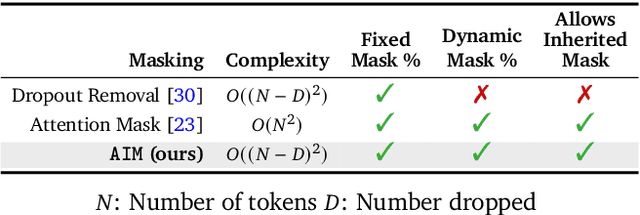
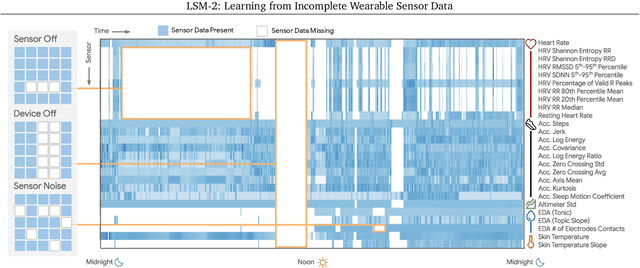
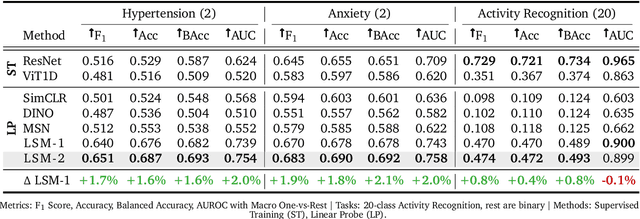
Abstract:Foundation models, a cornerstone of recent advancements in machine learning, have predominantly thrived on complete and well-structured data. Wearable sensor data frequently suffers from significant missingness, posing a substantial challenge for self-supervised learning (SSL) models that typically assume complete data inputs. This paper introduces the second generation of Large Sensor Model (LSM-2) with Adaptive and Inherited Masking (AIM), a novel SSL approach that learns robust representations directly from incomplete data without requiring explicit imputation. AIM's core novelty lies in its use of learnable mask tokens to model both existing ("inherited") and artificially introduced missingness, enabling it to robustly handle fragmented real-world data during inference. Pre-trained on an extensive dataset of 40M hours of day-long multimodal sensor data, our LSM-2 with AIM achieves the best performance across a diverse range of tasks, including classification, regression and generative modeling. Furthermore, LSM-2 with AIM exhibits superior scaling performance, and critically, maintains high performance even under targeted missingness scenarios, reflecting clinically coherent patterns, such as the diagnostic value of nighttime biosignals for hypertension prediction. This makes AIM a more reliable choice for real-world wearable data applications.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge