Yipeng Hu
Active learning using adaptable task-based prioritisation
Dec 03, 2022



Abstract:Supervised machine learning-based medical image computing applications necessitate expert label curation, while unlabelled image data might be relatively abundant. Active learning methods aim to prioritise a subset of available image data for expert annotation, for label-efficient model training. We develop a controller neural network that measures priority of images in a sequence of batches, as in batch-mode active learning, for multi-class segmentation tasks. The controller is optimised by rewarding positive task-specific performance gain, within a Markov decision process (MDP) environment that also optimises the task predictor. In this work, the task predictor is a segmentation network. A meta-reinforcement learning algorithm is proposed with multiple MDPs, such that the pre-trained controller can be adapted to a new MDP that contains data from different institutes and/or requires segmentation of different organs or structures within the abdomen. We present experimental results using multiple CT datasets from more than one thousand patients, with segmentation tasks of nine different abdominal organs, to demonstrate the efficacy of the learnt prioritisation controller function and its cross-institute and cross-organ adaptability. We show that the proposed adaptable prioritisation metric yields converging segmentation accuracy for the novel class of kidney, unseen in training, using between approximately 40\% to 60\% of labels otherwise required with other heuristic or random prioritisation metrics. For clinical datasets of limited size, the proposed adaptable prioritisation offers a performance improvement of 22.6\% and 10.2\% in Dice score, for tasks of kidney and liver vessel segmentation, respectively, compared to random prioritisation and alternative active sampling strategies.
Trackerless freehand ultrasound with sequence modelling and auxiliary transformation over past and future frames
Nov 09, 2022



Abstract:Three-dimensional (3D) freehand ultrasound (US) reconstruction without a tracker can be advantageous over its two-dimensional or tracked counterparts in many clinical applications. In this paper, we propose to estimate 3D spatial transformation between US frames from both past and future 2D images, using feed-forward and recurrent neural networks (RNNs). With the temporally available frames, a further multi-task learning algorithm is proposed to utilise a large number of auxiliary transformation-predicting tasks between them. Using more than 40,000 US frames acquired from 228 scans on 38 forearms of 19 volunteers in a volunteer study, the hold-out test performance is quantified by frame prediction accuracy, volume reconstruction overlap, accumulated tracking error and final drift, based on ground-truth from an optical tracker. The results show the importance of modelling the temporal-spatially correlated input frames as well as output transformations, with further improvement owing to additional past and/or future frames. The best performing model was associated with predicting transformation between moderately-spaced frames, with an interval of less than ten frames at 20 frames per second (fps). Little benefit was observed by adding frames more than one second away from the predicted transformation, with or without LSTM-based RNNs. Interestingly, with the proposed approach, explicit within-sequence loss that encourages consistency in composing transformations or minimises accumulated error may no longer be required. The implementation code and volunteer data will be made publicly available ensuring reproducibility and further research.
MONAI: An open-source framework for deep learning in healthcare
Nov 04, 2022



Abstract:Artificial Intelligence (AI) is having a tremendous impact across most areas of science. Applications of AI in healthcare have the potential to improve our ability to detect, diagnose, prognose, and intervene on human disease. For AI models to be used clinically, they need to be made safe, reproducible and robust, and the underlying software framework must be aware of the particularities (e.g. geometry, physiology, physics) of medical data being processed. This work introduces MONAI, a freely available, community-supported, and consortium-led PyTorch-based framework for deep learning in healthcare. MONAI extends PyTorch to support medical data, with a particular focus on imaging, and provide purpose-specific AI model architectures, transformations and utilities that streamline the development and deployment of medical AI models. MONAI follows best practices for software-development, providing an easy-to-use, robust, well-documented, and well-tested software framework. MONAI preserves the simple, additive, and compositional approach of its underlying PyTorch libraries. MONAI is being used by and receiving contributions from research, clinical and industrial teams from around the world, who are pursuing applications spanning nearly every aspect of healthcare.
Meta-Learning Initializations for Interactive Medical Image Registration
Oct 27, 2022



Abstract:We present a meta-learning framework for interactive medical image registration. Our proposed framework comprises three components: a learning-based medical image registration algorithm, a form of user interaction that refines registration at inference, and a meta-learning protocol that learns a rapidly adaptable network initialization. This paper describes a specific algorithm that implements the registration, interaction and meta-learning protocol for our exemplar clinical application: registration of magnetic resonance (MR) imaging to interactively acquired, sparsely-sampled transrectal ultrasound (TRUS) images. Our approach obtains comparable registration error (4.26 mm) to the best-performing non-interactive learning-based 3D-to-3D method (3.97 mm) while requiring only a fraction of the data, and occurring in real-time during acquisition. Applying sparsely sampled data to non-interactive methods yields higher registration errors (6.26 mm), demonstrating the effectiveness of interactive MR-TRUS registration, which may be applied intraoperatively given the real-time nature of the adaptation process.
Prototypical few-shot segmentation for cross-institution male pelvic structures with spatial registration
Sep 13, 2022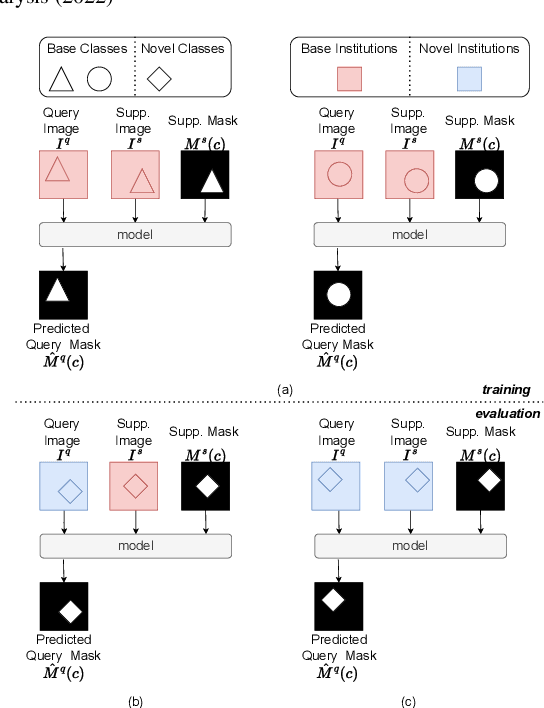

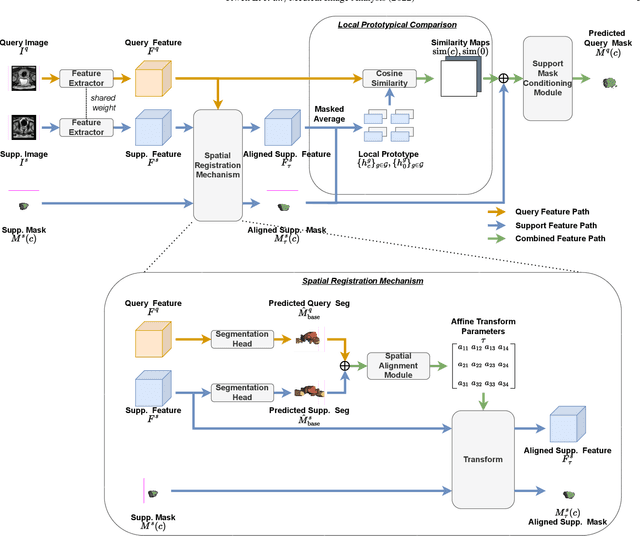

Abstract:The prowess that makes few-shot learning desirable in medical image analysis is the efficient use of the support image data, which are labelled to classify or segment new classes, a task that otherwise requires substantially more training images and expert annotations. This work describes a fully 3D prototypical few-shot segmentation algorithm, such that the trained networks can be effectively adapted to clinically interesting structures that are absent in training, using only a few labelled images from a different institute. First, to compensate for the widely recognised spatial variability between institutions in episodic adaptation of novel classes, a novel spatial registration mechanism is integrated into prototypical learning, consisting of a segmentation head and an spatial alignment module. Second, to assist the training with observed imperfect alignment, support mask conditioning module is proposed to further utilise the annotation available from the support images. Extensive experiments are presented in an application of segmenting eight anatomical structures important for interventional planning, using a data set of 589 pelvic T2-weighted MR images, acquired at seven institutes. The results demonstrate the efficacy in each of the 3D formulation, the spatial registration, and the support mask conditioning, all of which made positive contributions independently or collectively. Compared with the previously proposed 2D alternatives, the few-shot segmentation performance was improved with statistical significance, regardless whether the support data come from the same or different institutes.
Domain Generalization for Prostate Segmentation in Transrectal Ultrasound Images: A Multi-center Study
Sep 05, 2022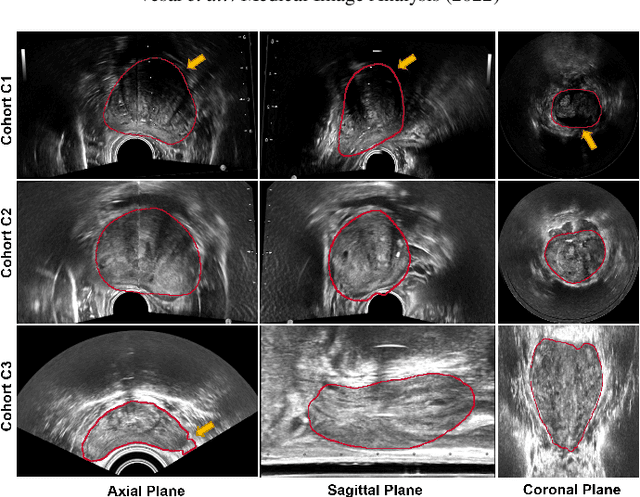
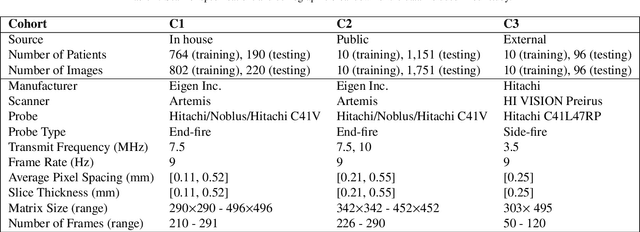
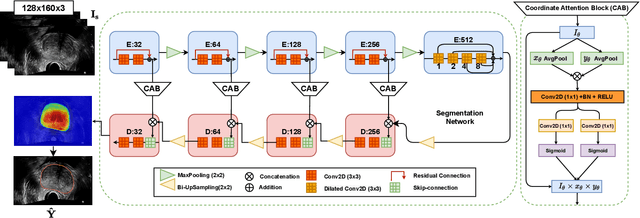
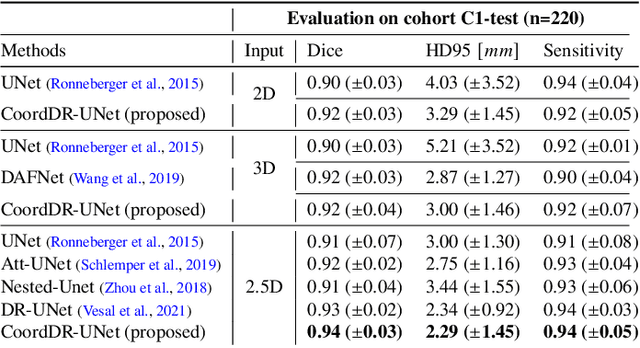
Abstract:Prostate biopsy and image-guided treatment procedures are often performed under the guidance of ultrasound fused with magnetic resonance images (MRI). Accurate image fusion relies on accurate segmentation of the prostate on ultrasound images. Yet, the reduced signal-to-noise ratio and artifacts (e.g., speckle and shadowing) in ultrasound images limit the performance of automated prostate segmentation techniques and generalizing these methods to new image domains is inherently difficult. In this study, we address these challenges by introducing a novel 2.5D deep neural network for prostate segmentation on ultrasound images. Our approach addresses the limitations of transfer learning and finetuning methods (i.e., drop in performance on the original training data when the model weights are updated) by combining a supervised domain adaptation technique and a knowledge distillation loss. The knowledge distillation loss allows the preservation of previously learned knowledge and reduces the performance drop after model finetuning on new datasets. Furthermore, our approach relies on an attention module that considers model feature positioning information to improve the segmentation accuracy. We trained our model on 764 subjects from one institution and finetuned our model using only ten subjects from subsequent institutions. We analyzed the performance of our method on three large datasets encompassing 2067 subjects from three different institutions. Our method achieved an average Dice Similarity Coefficient (Dice) of $94.0\pm0.03$ and Hausdorff Distance (HD95) of 2.28 $mm$ in an independent set of subjects from the first institution. Moreover, our model generalized well in the studies from the other two institutions (Dice: $91.0\pm0.03$; HD95: 3.7$mm$ and Dice: $82.0\pm0.03$; HD95: 7.1 $mm$).
Optimising Chest X-Rays for Image Analysis by Identifying and Removing Confounding Factors
Aug 22, 2022
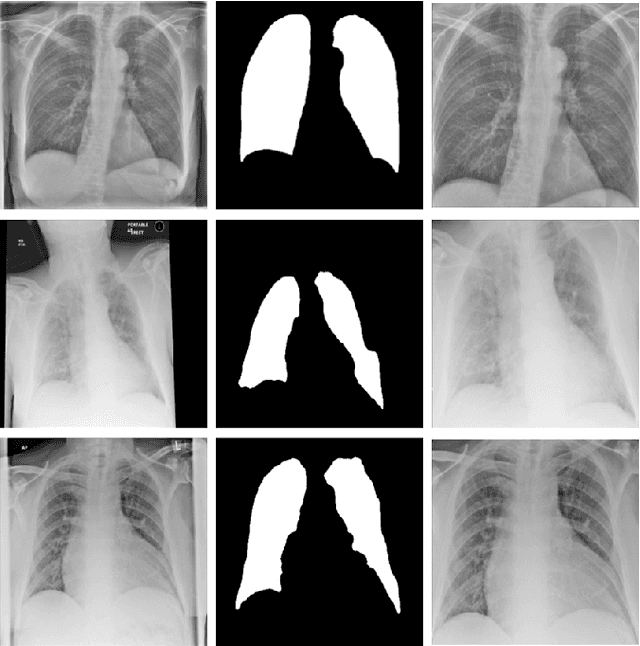
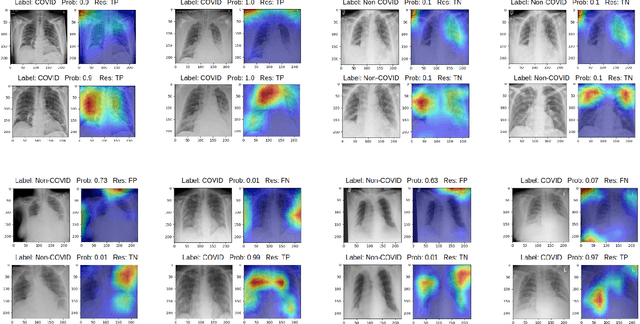
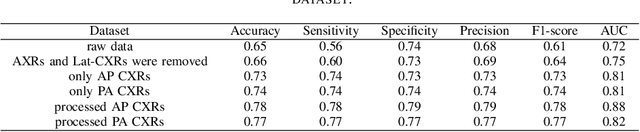
Abstract:During the COVID-19 pandemic, the sheer volume of imaging performed in an emergency setting for COVID-19 diagnosis has resulted in a wide variability of clinical CXR acquisitions. This variation is seen in the CXR projections used, image annotations added and in the inspiratory effort and degree of rotation of clinical images. The image analysis community has attempted to ease the burden on overstretched radiology departments during the pandemic by developing automated COVID-19 diagnostic algorithms, the input for which has been CXR imaging. Large publicly available CXR datasets have been leveraged to improve deep learning algorithms for COVID-19 diagnosis. Yet the variable quality of clinically-acquired CXRs within publicly available datasets could have a profound effect on algorithm performance. COVID-19 diagnosis may be inferred by an algorithm from non-anatomical features on an image such as image labels. These imaging shortcuts may be dataset-specific and limit the generalisability of AI systems. Understanding and correcting key potential biases in CXR images is therefore an essential first step prior to CXR image analysis. In this study, we propose a simple and effective step-wise approach to pre-processing a COVID-19 chest X-ray dataset to remove undesired biases. We perform ablation studies to show the impact of each individual step. The results suggest that using our proposed pipeline could increase accuracy of the baseline COVID-19 detection algorithm by up to 13%.
Bayesian Pseudo Labels: Expectation Maximization for Robust and Efficient Semi-Supervised Segmentation
Aug 08, 2022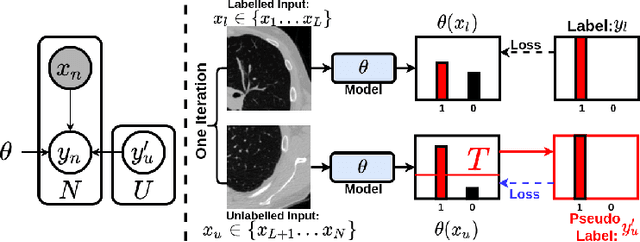
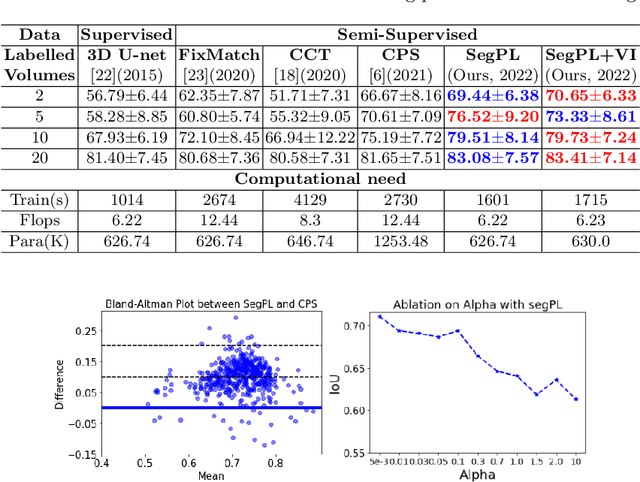
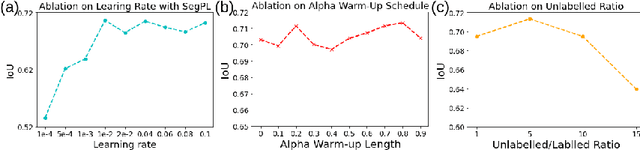
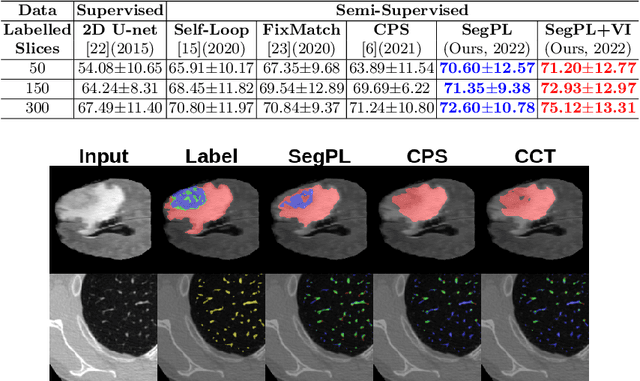
Abstract:This paper concerns pseudo labelling in segmentation. Our contribution is fourfold. Firstly, we present a new formulation of pseudo-labelling as an Expectation-Maximization (EM) algorithm for clear statistical interpretation. Secondly, we propose a semi-supervised medical image segmentation method purely based on the original pseudo labelling, namely SegPL. We demonstrate SegPL is a competitive approach against state-of-the-art consistency regularisation based methods on semi-supervised segmentation on a 2D multi-class MRI brain tumour segmentation task and a 3D binary CT lung vessel segmentation task. The simplicity of SegPL allows less computational cost comparing to prior methods. Thirdly, we demonstrate that the effectiveness of SegPL may originate from its robustness against out-of-distribution noises and adversarial attacks. Lastly, under the EM framework, we introduce a probabilistic generalisation of SegPL via variational inference, which learns a dynamic threshold for pseudo labelling during the training. We show that SegPL with variational inference can perform uncertainty estimation on par with the gold-standard method Deep Ensemble.
Cross-Modality Image Registration using a Training-Time Privileged Third Modality
Jul 26, 2022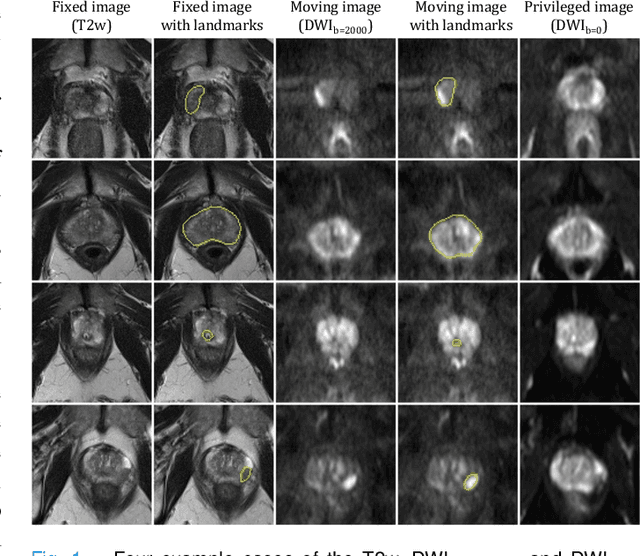
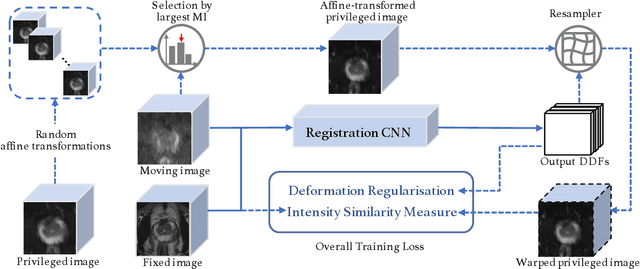
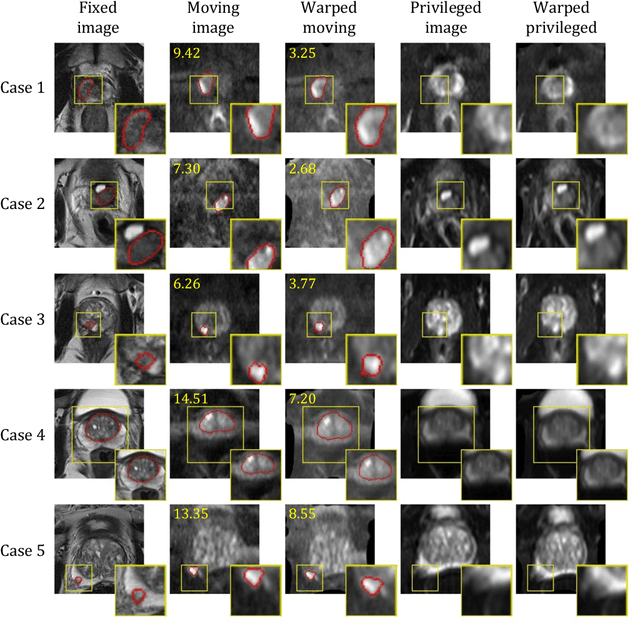
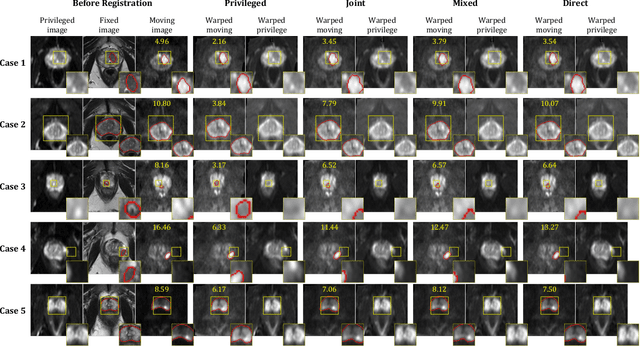
Abstract:In this work, we consider the task of pairwise cross-modality image registration, which may benefit from exploiting additional images available only at training time from an additional modality that is different to those being registered. As an example, we focus on aligning intra-subject multiparametric Magnetic Resonance (mpMR) images, between T2-weighted (T2w) scans and diffusion-weighted scans with high b-value (DWI$_{high-b}$). For the application of localising tumours in mpMR images, diffusion scans with zero b-value (DWI$_{b=0}$) are considered easier to register to T2w due to the availability of corresponding features. We propose a learning from privileged modality algorithm, using a training-only imaging modality DWI$_{b=0}$, to support the challenging multi-modality registration problems. We present experimental results based on 369 sets of 3D multiparametric MRI images from 356 prostate cancer patients and report, with statistical significance, a lowered median target registration error of 4.34 mm, when registering the holdout DWI$_{high-b}$ and T2w image pairs, compared with that of 7.96 mm before registration. Results also show that the proposed learning-based registration networks enabled efficient registration with comparable or better accuracy, compared with a classical iterative algorithm and other tested learning-based methods with/without the additional modality. These compared algorithms also failed to produce any significantly improved alignment between DWI$_{high-b}$ and T2w in this challenging application.
Rapid Lung Ultrasound COVID-19 Severity Scoring with Resource-Efficient Deep Feature Extraction
Jul 22, 2022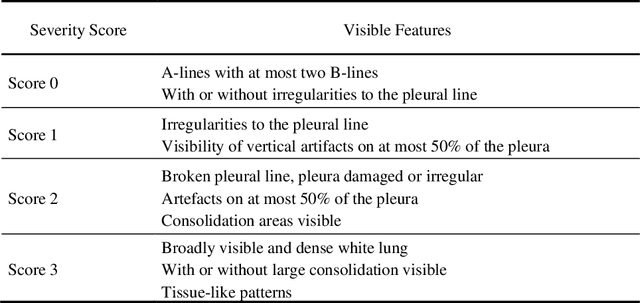
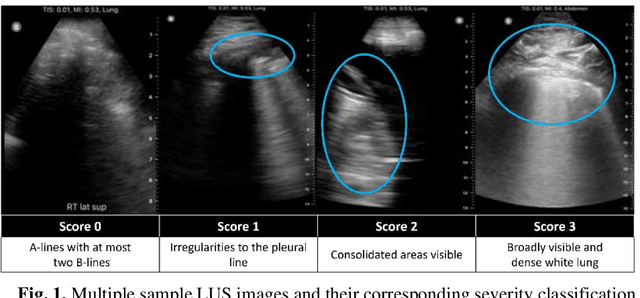
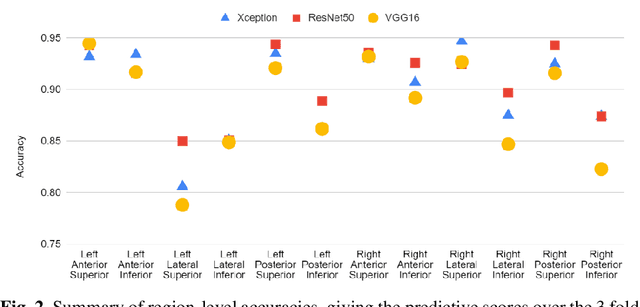
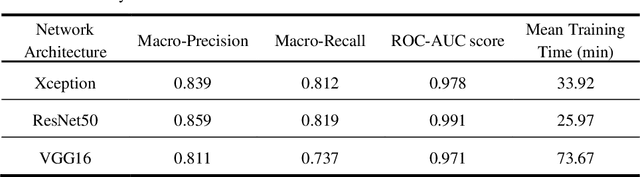
Abstract:Artificial intelligence-based analysis of lung ultrasound imaging has been demonstrated as an effective technique for rapid diagnostic decision support throughout the COVID-19 pandemic. However, such techniques can require days- or weeks-long training processes and hyper-parameter tuning to develop intelligent deep learning image analysis models. This work focuses on leveraging 'off-the-shelf' pre-trained models as deep feature extractors for scoring disease severity with minimal training time. We propose using pre-trained initializations of existing methods ahead of simple and compact neural networks to reduce reliance on computational capacity. This reduction of computational capacity is of critical importance in time-limited or resource-constrained circumstances, such as the early stages of a pandemic. On a dataset of 49 patients, comprising over 20,000 images, we demonstrate that the use of existing methods as feature extractors results in the effective classification of COVID-19-related pneumonia severity while requiring only minutes of training time. Our methods can achieve an accuracy of over 0.93 on a 4-level severity score scale and provides comparable per-patient region and global scores compared to expert annotated ground truths. These results demonstrate the capability for rapid deployment and use of such minimally-adapted methods for progress monitoring, patient stratification and management in clinical practice for COVID-19 patients, and potentially in other respiratory diseases.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge