Adam Shephard
A Deep Learning Framework for Thyroid Nodule Segmentation and Malignancy Classification from Ultrasound Images
Nov 14, 2025



Abstract:Ultrasound-based risk stratification of thyroid nodules is a critical clinical task, but it suffers from high inter-observer variability. While many deep learning (DL) models function as "black boxes," we propose a fully automated, two-stage framework for interpretable malignancy prediction. Our method achieves interpretability by forcing the model to focus only on clinically relevant regions. First, a TransUNet model automatically segments the thyroid nodule. The resulting mask is then used to create a region of interest around the nodule, and this localised image is fed directly into a ResNet-18 classifier. We evaluated our framework using 5-fold cross-validation on a clinical dataset of 349 images, where it achieved a high F1-score of 0.852 for predicting malignancy. To validate its performance, we compared it against a strong baseline using a Random Forest classifier with hand-crafted morphological features, which achieved an F1-score of 0.829. The superior performance of our DL framework suggests that the implicit visual features learned from the localised nodule are more predictive than explicit shape features alone. This is the first fully automated end-to-end pipeline for both detecting thyroid nodules on ultrasound images and predicting their malignancy.
Synergy vs. Noise: Performance-Guided Multimodal Fusion For Biochemical Recurrence-Free Survival in Prostate Cancer
Nov 14, 2025



Abstract:Multimodal deep learning (MDL) has emerged as a transformative approach in computational pathology. By integrating complementary information from multiple data sources, MDL models have demonstrated superior predictive performance across diverse clinical tasks compared to unimodal models. However, the assumption that combining modalities inherently improves performance remains largely unexamined. We hypothesise that multimodal gains depend critically on the predictive quality of individual modalities, and that integrating weak modalities may introduce noise rather than complementary information. We test this hypothesis on a prostate cancer dataset with histopathology, radiology, and clinical data to predict time-to-biochemical recurrence. Our results confirm that combining high-performing modalities yield superior performance compared to unimodal approaches. However, integrating a poor-performing modality with other higher-performing modalities degrades predictive accuracy. These findings demonstrate that multimodal benefit requires selective, performance-guided integration rather than indiscriminate modality combination, with implications for MDL design across computational pathology and medical imaging.
CORE - A Cell-Level Coarse-to-Fine Image Registration Engine for Multi-stain Image Alignment
Nov 12, 2025Abstract:Accurate and efficient registration of whole slide images (WSIs) is essential for high-resolution, nuclei-level analysis in multi-stained tissue slides. We propose a novel coarse-to-fine framework CORE for accurate nuclei-level registration across diverse multimodal whole-slide image (WSI) datasets. The coarse registration stage leverages prompt-based tissue mask extraction to effectively filter out artefacts and non-tissue regions, followed by global alignment using tissue morphology and ac- celerated dense feature matching with a pre-trained feature extractor. From the coarsely aligned slides, nuclei centroids are detected and subjected to fine-grained rigid registration using a custom, shape-aware point-set registration model. Finally, non-rigid alignment at the cellular level is achieved by estimating a non-linear dis- placement field using Coherent Point Drift (CPD). Our approach benefits from automatically generated nuclei that enhance the accuracy of deformable registra- tion and ensure precise nuclei-level correspondence across modalities. The pro- posed model is evaluated on three publicly available WSI registration datasets, and two private datasets. We show that CORE outperforms current state-of-the-art methods in terms of generalisability, precision, and robustness in bright-field and immunofluorescence microscopy WSIs
MultiSurv: A Multimodal Deep Survival Framework for Prostrate and Bladder Cancer
Sep 05, 2025Abstract:Accurate prediction of time-to-event outcomes is a central challenge in oncology, with significant implications for treatment planning and patient management. In this work, we present MultiSurv, a multimodal deep survival model utilising DeepHit with a projection layer and inter-modality cross-attention, which integrates heterogeneous patient data, including clinical, MRI, RNA-seq and whole-slide pathology features. The model is designed to capture complementary prognostic signals across modalities and estimate individualised time-to-biochemical recurrence in prostate cancer and time-to-cancer recurrence in bladder cancer. Our approach was evaluated in the context of the CHIMERA Grand Challenge, across two of the three provided tasks. For Task 1 (prostate cancer bio-chemical recurrence prediction), the proposed framework achieved a concordance index (C-index) of 0.843 on 5-folds cross-validation and 0.818 on CHIMERA development set, demonstrating robust discriminatory ability. For Task 3 (bladder cancer recurrence prediction), the model obtained a C-index of 0.662 on 5-folds cross-validation and 0.457 on development set, highlighting its adaptability and potential for clinical translation. These results suggest that leveraging multimodal integration with deep survival learning provides a promising pathway toward personalised risk stratification in prostate and bladder cancer. Beyond the challenge setting, our framework is broadly applicable to survival prediction tasks involving heterogeneous biomedical data.
TIAViz: A Browser-based Visualization Tool for Computational Pathology Models
Feb 15, 2024


Abstract:Digital pathology has gained significant traction in modern healthcare systems. This shift from optical microscopes to digital imagery brings with it the potential for improved diagnosis, efficiency, and the integration of AI tools into the pathologists workflow. A critical aspect of this is visualization. Throughout the development of a machine learning (ML) model in digital pathology, it is crucial to have flexible, openly available tools to visualize models, from their outputs and predictions to the underlying annotations and images used to train or test a model. We introduce TIAViz, a Python-based visualization tool built into TIAToolbox which allows flexible, interactive, fully zoomable overlay of a wide variety of information onto whole slide images, including graphs, heatmaps, segmentations, annotations and other WSIs. The UI is browser-based, allowing use either locally, on a remote machine, or on a server to provide publicly available demos. This tool is open source and is made available at: https://github.com/TissueImageAnalytics/tiatoolbox and via pip installation (pip install tiatoolbox) and conda as part of TIAToolbox.
Domain Generalization in Computational Pathology: Survey and Guidelines
Oct 30, 2023



Abstract:Deep learning models have exhibited exceptional effectiveness in Computational Pathology (CPath) by tackling intricate tasks across an array of histology image analysis applications. Nevertheless, the presence of out-of-distribution data (stemming from a multitude of sources such as disparate imaging devices and diverse tissue preparation methods) can cause \emph{domain shift} (DS). DS decreases the generalization of trained models to unseen datasets with slightly different data distributions, prompting the need for innovative \emph{domain generalization} (DG) solutions. Recognizing the potential of DG methods to significantly influence diagnostic and prognostic models in cancer studies and clinical practice, we present this survey along with guidelines on achieving DG in CPath. We rigorously define various DS types, systematically review and categorize existing DG approaches and resources in CPath, and provide insights into their advantages, limitations, and applicability. We also conduct thorough benchmarking experiments with 28 cutting-edge DG algorithms to address a complex DG problem. Our findings suggest that careful experiment design and CPath-specific Stain Augmentation technique can be very effective. However, there is no one-size-fits-all solution for DG in CPath. Therefore, we establish clear guidelines for detecting and managing DS depending on different scenarios. While most of the concepts, guidelines, and recommendations are given for applications in CPath, we believe that they are applicable to most medical image analysis tasks as well.
Domain generalization across tumor types, laboratories, and species -- insights from the 2022 edition of the Mitosis Domain Generalization Challenge
Sep 27, 2023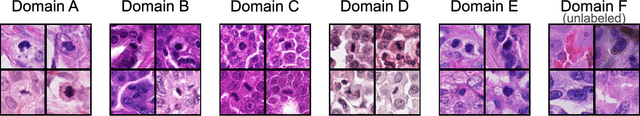
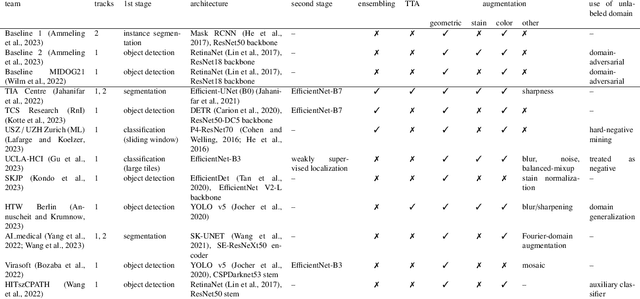
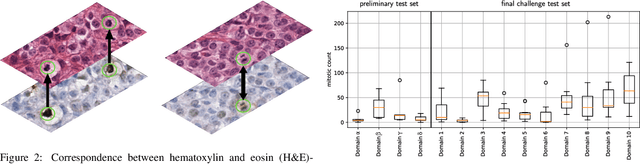
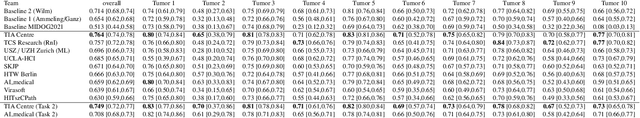
Abstract:Recognition of mitotic figures in histologic tumor specimens is highly relevant to patient outcome assessment. This task is challenging for algorithms and human experts alike, with deterioration of algorithmic performance under shifts in image representations. Considerable covariate shifts occur when assessment is performed on different tumor types, images are acquired using different digitization devices, or specimens are produced in different laboratories. This observation motivated the inception of the 2022 challenge on MItosis Domain Generalization (MIDOG 2022). The challenge provided annotated histologic tumor images from six different domains and evaluated the algorithmic approaches for mitotic figure detection provided by nine challenge participants on ten independent domains. Ground truth for mitotic figure detection was established in two ways: a three-expert consensus and an independent, immunohistochemistry-assisted set of labels. This work represents an overview of the challenge tasks, the algorithmic strategies employed by the participants, and potential factors contributing to their success. With an $F_1$ score of 0.764 for the top-performing team, we summarize that domain generalization across various tumor domains is possible with today's deep learning-based recognition pipelines. When assessed against the immunohistochemistry-assisted reference standard, all methods resulted in reduced recall scores, but with only minor changes in the order of participants in the ranking.
Why is the winner the best?
Mar 30, 2023



Abstract:International benchmarking competitions have become fundamental for the comparative performance assessment of image analysis methods. However, little attention has been given to investigating what can be learnt from these competitions. Do they really generate scientific progress? What are common and successful participation strategies? What makes a solution superior to a competing method? To address this gap in the literature, we performed a multi-center study with all 80 competitions that were conducted in the scope of IEEE ISBI 2021 and MICCAI 2021. Statistical analyses performed based on comprehensive descriptions of the submitted algorithms linked to their rank as well as the underlying participation strategies revealed common characteristics of winning solutions. These typically include the use of multi-task learning (63%) and/or multi-stage pipelines (61%), and a focus on augmentation (100%), image preprocessing (97%), data curation (79%), and postprocessing (66%). The "typical" lead of a winning team is a computer scientist with a doctoral degree, five years of experience in biomedical image analysis, and four years of experience in deep learning. Two core general development strategies stood out for highly-ranked teams: the reflection of the metrics in the method design and the focus on analyzing and handling failure cases. According to the organizers, 43% of the winning algorithms exceeded the state of the art but only 11% completely solved the respective domain problem. The insights of our study could help researchers (1) improve algorithm development strategies when approaching new problems, and (2) focus on open research questions revealed by this work.
CoNIC Challenge: Pushing the Frontiers of Nuclear Detection, Segmentation, Classification and Counting
Mar 14, 2023



Abstract:Nuclear detection, segmentation and morphometric profiling are essential in helping us further understand the relationship between histology and patient outcome. To drive innovation in this area, we setup a community-wide challenge using the largest available dataset of its kind to assess nuclear segmentation and cellular composition. Our challenge, named CoNIC, stimulated the development of reproducible algorithms for cellular recognition with real-time result inspection on public leaderboards. We conducted an extensive post-challenge analysis based on the top-performing models using 1,658 whole-slide images of colon tissue. With around 700 million detected nuclei per model, associated features were used for dysplasia grading and survival analysis, where we demonstrated that the challenge's improvement over the previous state-of-the-art led to significant boosts in downstream performance. Our findings also suggest that eosinophils and neutrophils play an important role in the tumour microevironment. We release challenge models and WSI-level results to foster the development of further methods for biomarker discovery.
Stain-Robust Mitotic Figure Detection for MIDOG 2022 Challenge
Aug 26, 2022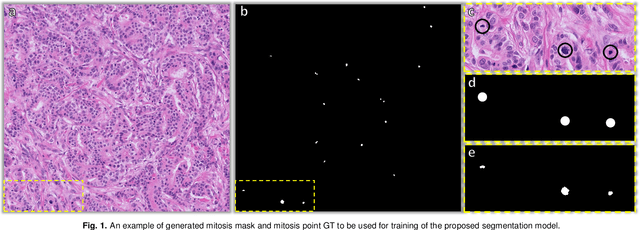
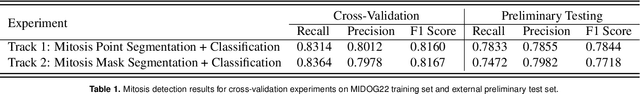
Abstract:The detection of mitotic figures from different scanners/sites remains an important topic of research, owing to its potential in assisting clinicians with tumour grading. The MItosis DOmain Generalization (MIDOG) 2022 challenge aims to test the robustness of detection models on unseen data from multiple scanners and tissue types for this task. We present a short summary of the approach employed by the TIA Centre team to address this challenge. Our approach is based on a hybrid detection model, where mitotic candidates are segmented, before being refined by a deep learning classifier. Cross-validation on the training images achieved the F1-score of 0.816 and 0.784 on the preliminary test set, demonstrating the generalizability of our model to unseen data from new scanners.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge