Le Lu
Structured Landmark Detection via Topology-Adapting Deep Graph Learning
Apr 23, 2020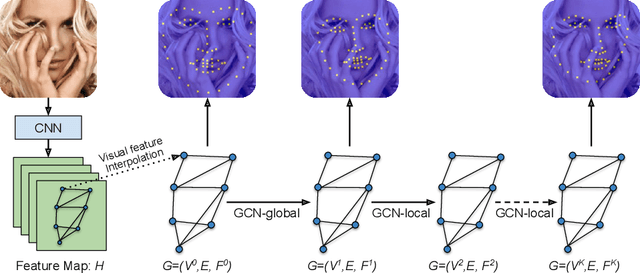
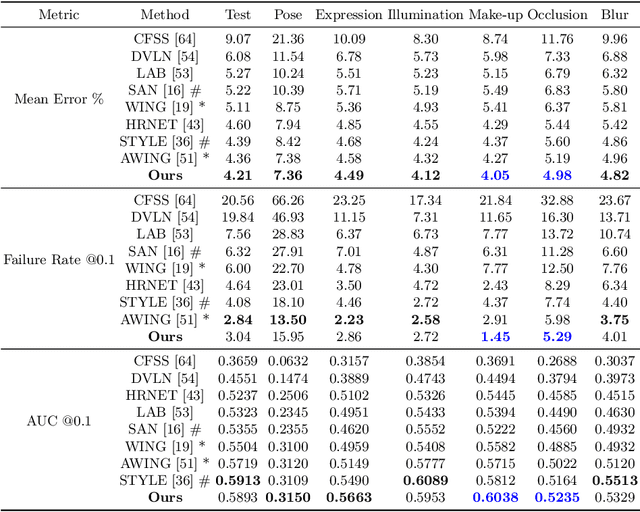

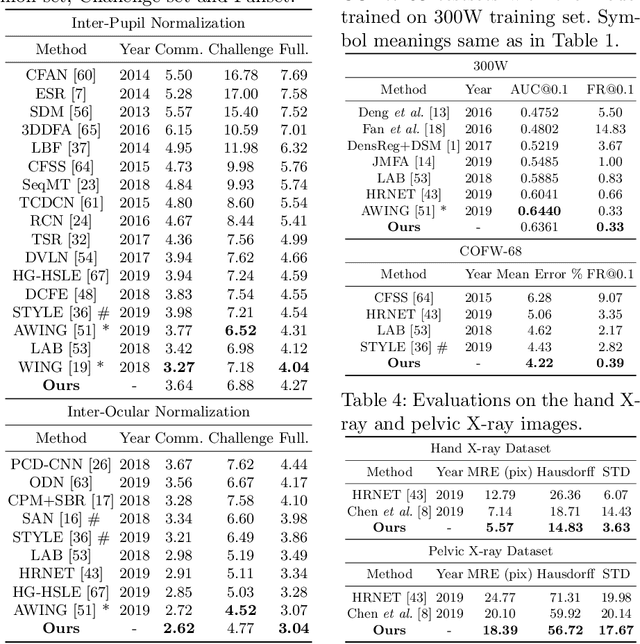
Abstract:Image landmark detection aims to automatically identify the locations of predefined fiducial points. Despite recent success in this filed, higher-ordered structural modeling to capture implicit or explicit relationships among anatomical landmarks has not been adequately exploited. In this work, we present a new topology-adapting deep graph learning approach for accurate anatomical facial and medical (e.g., hand, pelvis) landmark detection. The proposed method constructs graph signals leveraging both local image features and global shape features. The adaptive graph topology naturally explores and lands on task-specific structures which is learned end-to-end with two Graph Convolutional Networks (GCNs). Extensive experiments are conducted on three public facial image datasets (WFLW, 300W and COFW-68) as well as three real-world X-ray medical datasets (Cephalometric (public), Hand and Pelvis). Quantitative results comparing with the previous state-of-the-art approaches across all studied datasets indicating the superior performance in both robustness and accuracy. Qualitative visualizations of the learned graph topologies demonstrate a physically plausible connectivity laying behind the landmarks.
Organ at Risk Segmentation for Head and Neck Cancer using Stratified Learning and Neural Architecture Search
Apr 17, 2020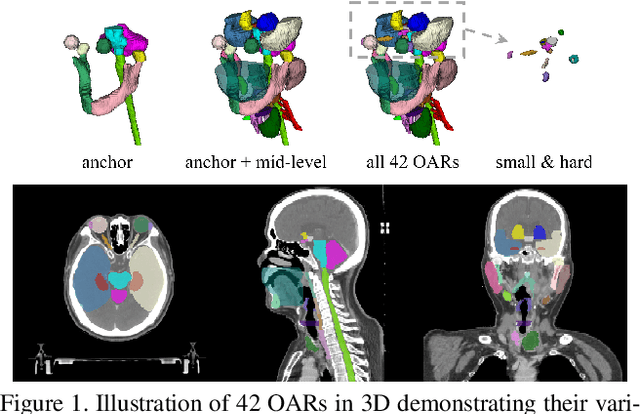
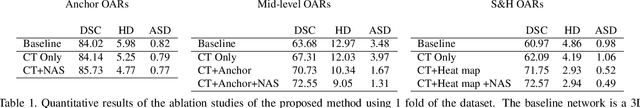
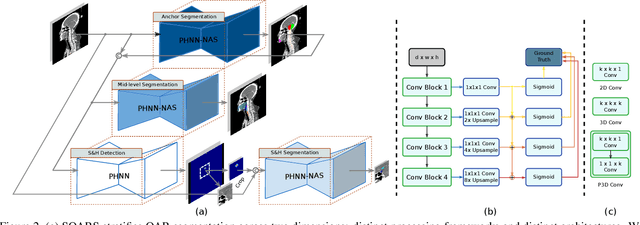
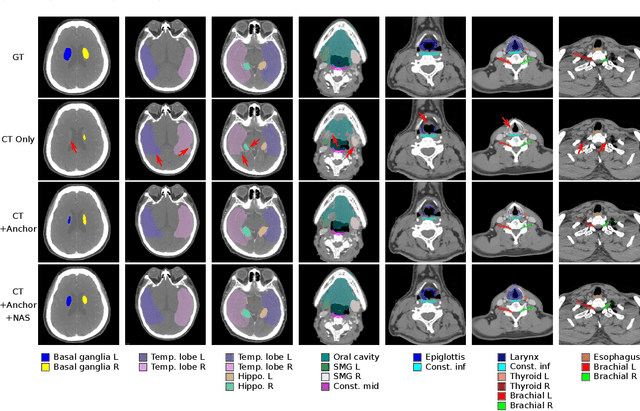
Abstract:OAR segmentation is a critical step in radiotherapy of head and neck (H&N) cancer, where inconsistencies across radiation oncologists and prohibitive labor costs motivate automated approaches. However, leading methods using standard fully convolutional network workflows that are challenged when the number of OARs becomes large, e.g. > 40. For such scenarios, insights can be gained from the stratification approaches seen in manual clinical OAR delineation. This is the goal of our work, where we introduce stratified organ at risk segmentation (SOARS), an approach that stratifies OARs into anchor, mid-level, and small & hard (S&H) categories. SOARS stratifies across two dimensions. The first dimension is that distinct processing pipelines are used for each OAR category. In particular, inspired by clinical practices, anchor OARs are used to guide the mid-level and S&H categories. The second dimension is that distinct network architectures are used to manage the significant contrast, size, and anatomy variations between different OARs. We use differentiable neural architecture search (NAS), allowing the network to choose among 2D, 3D or Pseudo-3D convolutions. Extensive 4-fold cross-validation on 142 H&N cancer patients with 42 manually labeled OARs, the most comprehensive OAR dataset to date, demonstrates that both pipeline- and NAS-stratification significantly improves quantitative performance over the state-of-the-art (from 69.52% to 73.68% in absolute Dice scores). Thus, SOARS provides a powerful and principled means to manage the highly complex segmentation space of OARs.
Unsupervised Learning of Landmarks based on Inter-Intra Subject Consistencies
Apr 16, 2020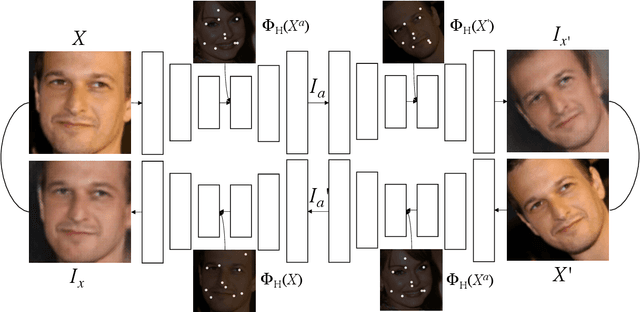

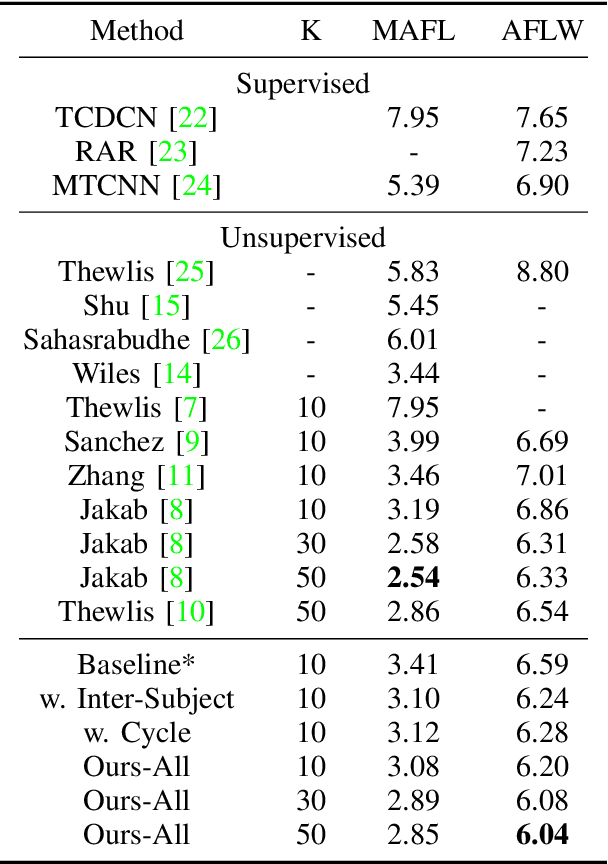
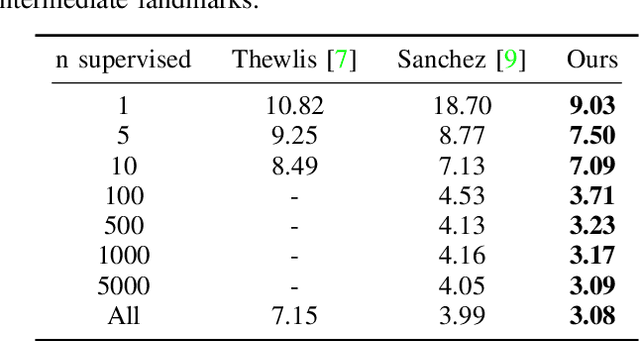
Abstract:We present a novel unsupervised learning approach to image landmark discovery by incorporating the inter-subject landmark consistencies on facial images. This is achieved via an inter-subject mapping module that transforms original subject landmarks based on an auxiliary subject-related structure. To recover from the transformed images back to the original subject, the landmark detector is forced to learn spatial locations that contain the consistent semantic meanings both for the paired intra-subject images and between the paired inter-subject images. Our proposed method is extensively evaluated on two public facial image datasets (MAFL, AFLW) with various settings. Experimental results indicate that our method can extract the consistent landmarks for both datasets and achieve better performances compared to the previous state-of-the-art methods quantitatively and qualitatively.
Lesion Harvester: Iteratively Mining Unlabeled Lesions and Hard-Negative Examples at Scale
Jan 28, 2020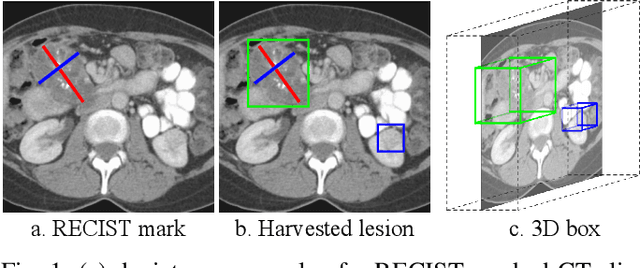
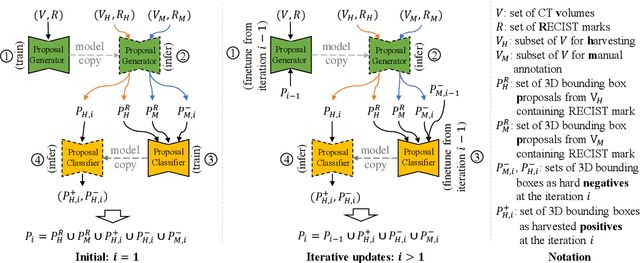
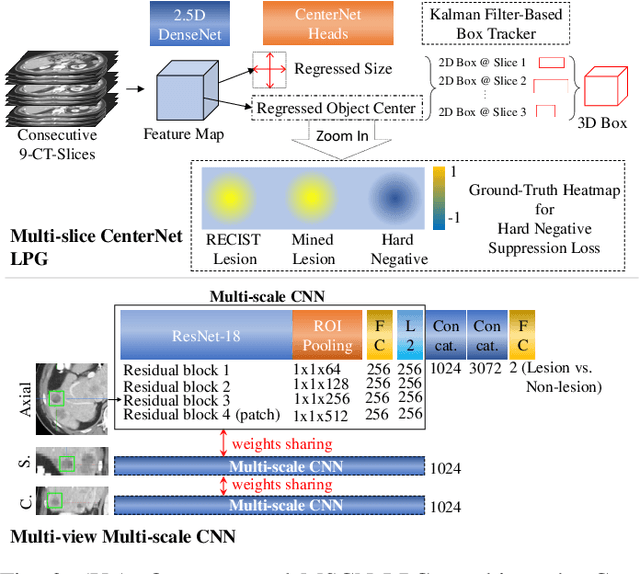
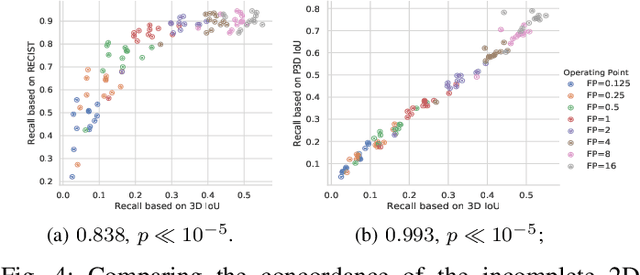
Abstract:Acquiring large-scale medical image data, necessary for training machine learning algorithms, is frequently intractable, due to prohibitive expert-driven annotation costs. Recent datasets extracted from hospital archives, e.g., DeepLesion, have begun to address this problem. However, these are often incompletely or noisily labeled, e.g., DeepLesion leaves over 50% of its lesions unlabeled. Thus, effective methods to harvest missing annotations are critical for continued progress in medical image analysis. This is the goal of our work, where we develop a powerful system to harvest missing lesions from the DeepLesion dataset at high precision. Accepting the need for some degree of expert labor to achieve high fidelity, we exploit a small fully-labeled subset of medical image volumes and use it to intelligently mine annotations from the remainder. To do this, we chain together a highly sensitive lesion proposal generator and a very selective lesion proposal classifier. While our framework is generic, we optimize our performance by proposing a 3D contextual lesion proposal generator and by using a multi-view multi-scale lesion proposal classifier. These produce harvested and hard-negative proposals, which we then re-use to finetune our proposal generator by using a novel hard negative suppression loss, continuing this process until no extra lesions are found. Extensive experimental analysis demonstrates that our method can harvest an additional 9,805 lesions while keeping precision above 90%. To demonstrate the benefits of our approach, we show that lesion detectors trained on our harvested lesions can significantly outperform the same variants only trained on the original annotations, with boost of average precision of 7% to 10%. We open source our annotations at https://github.com/JimmyCai91/DeepLesionAnnotation.
CT Data Curation for Liver Patients: Phase Recognition in Dynamic Contrast-Enhanced CT
Sep 27, 2019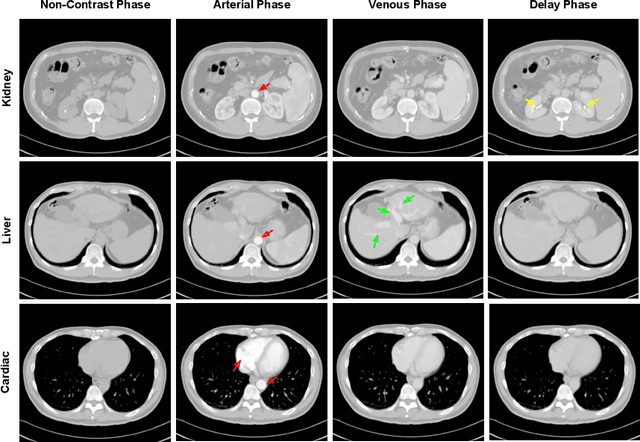
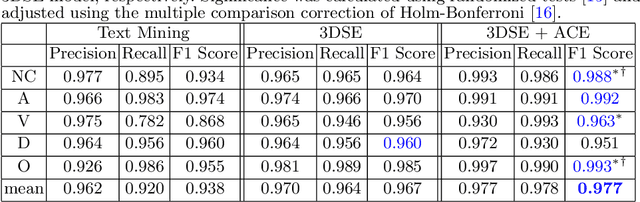
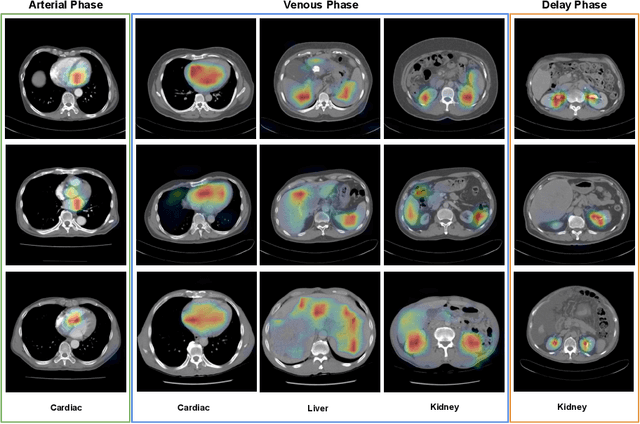

Abstract:As the demand for more descriptive machine learning models grows within medical imaging, bottlenecks due to data paucity will exacerbate. Thus, collecting enough large-scale data will require automated tools to harvest data/label pairs from messy and real-world datasets, such as hospital PACS. This is the focus of our work, where we present a principled data curation tool to extract multi-phase CT liver studies and identify each scan's phase from a real-world and heterogenous hospital PACS dataset. Emulating a typical deployment scenario, we first obtain a set of noisy labels from our institutional partners that are text mined using simple rules from DICOM tags. We train a deep learning system, using a customized and streamlined 3D SE architecture, to identify non-contrast, arterial, venous, and delay phase dynamic CT liver scans, filtering out anything else, including other types of liver contrast studies. To exploit as much training data as possible, we also introduce an aggregated cross entropy loss that can learn from scans only identified as "contrast". Extensive experiments on a dataset of 43K scans of 7680 patient imaging studies demonstrate that our 3DSE architecture, armed with our aggregated loss, can achieve a mean F1 of 0.977 and can correctly harvest up to 92.7% of studies, which significantly outperforms the text-mined and standard-loss approach, and also outperforms other, and more complex, model architectures.
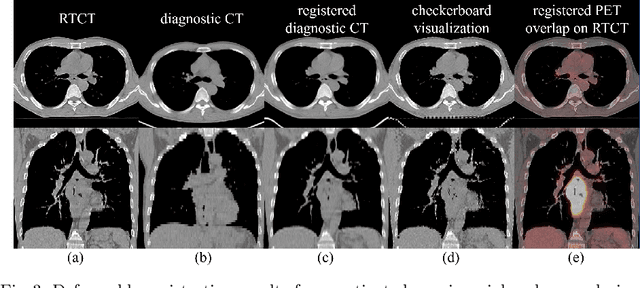
Deep Esophageal Clinical Target Volume Delineation using Encoded 3D Spatial Context of Tumors, Lymph Nodes, and Organs At Risk
Sep 06, 2019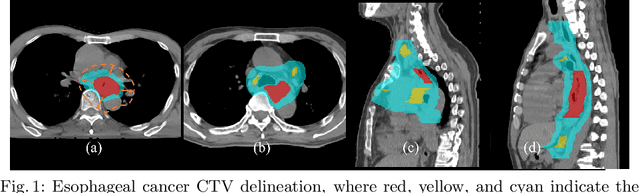

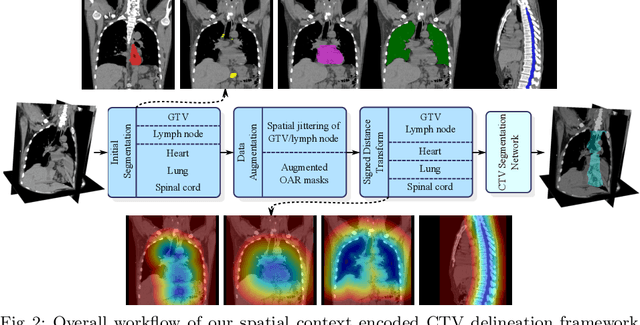
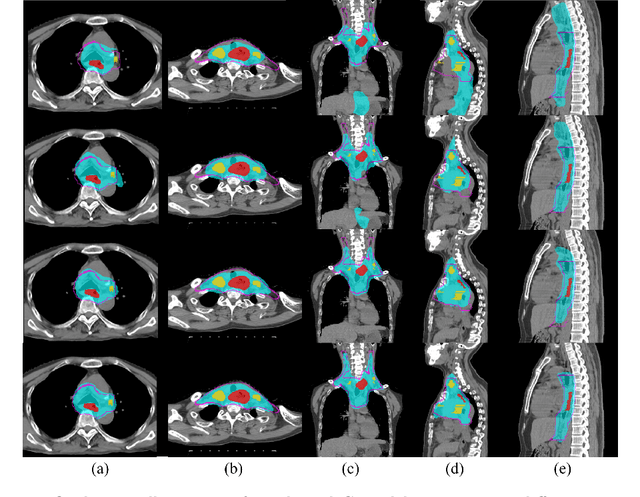
Abstract:Clinical target volume (CTV) delineation from radiotherapy computed tomography (RTCT) images is used to define the treatment areas containing the gross tumor volume (GTV) and/or sub-clinical malignant disease for radiotherapy (RT). High intra- and inter-user variability makes this a particularly difficult task for esophageal cancer. This motivates automated solutions, which is the aim of our work. Because CTV delineation is highly context-dependent--it must encompass the GTV and regional lymph nodes (LNs) while also avoiding excessive exposure to the organs at risk (OARs)--we formulate it as a deep contextual appearance-based problem using encoded spatial contexts of these anatomical structures. This allows the deep network to better learn from and emulate the margin- and appearance-based delineation performed by human physicians. Additionally, we develop domain-specific data augmentation to inject robustness to our system. Finally, we show that a simple 3D progressive holistically nested network (PHNN), which avoids computationally heavy decoding paths while still aggregating features at different levels of context, can outperform more complicated networks. Cross-validated experiments on a dataset of 135 esophageal cancer patients demonstrate that our encoded spatial context approach can produce concrete performance improvements, with an average Dice score of 83.9% and an average surface distance of 4.2 mm, representing improvements of 3.8% and 2.4 mm, respectively, over the state-of-the-art approach.
Accurate Esophageal Gross Tumor Volume Segmentation in PET/CT using Two-Stream Chained 3D Deep Network Fusion
Sep 06, 2019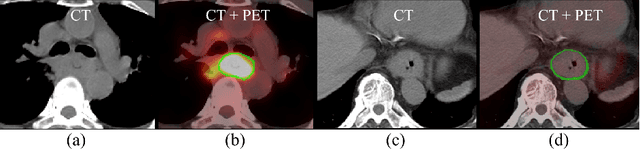
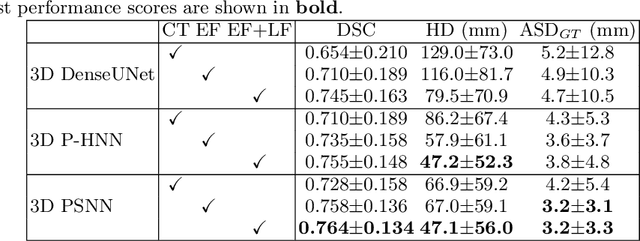
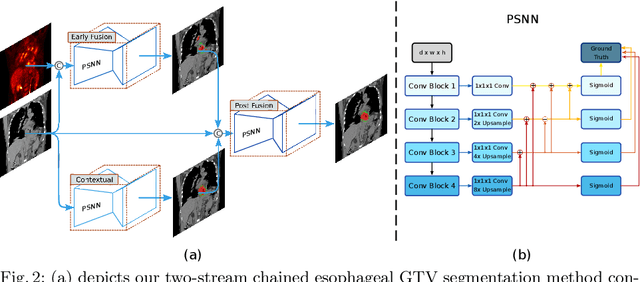
Abstract:Gross tumor volume (GTV) segmentation is a critical step in esophageal cancer radiotherapy treatment planning. Inconsistencies across oncologists and prohibitive labor costs motivate automated approaches for this task. However, leading approaches are only applied to radiotherapy computed tomography (RTCT) images taken prior to treatment. This limits the performance as RTCT suffers from low contrast between the esophagus, tumor, and surrounding tissues. In this paper, we aim to exploit both RTCT and positron emission tomography (PET) imaging modalities to facilitate more accurate GTV segmentation. By utilizing PET, we emulate medical professionals who frequently delineate GTV boundaries through observation of the RTCT images obtained after prescribing radiotherapy and PET/CT images acquired earlier for cancer staging. To take advantage of both modalities, we present a two-stream chained segmentation approach that effectively fuses the CT and PET modalities via early and late 3D deep-network-based fusion. Furthermore, to effect the fusion and segmentation we propose a simple yet effective progressive semantically nested network (PSNN) model that outperforms more complicated models. Extensive 5-fold cross-validation on 110 esophageal cancer patients, the largest analysis to date, demonstrates that both the proposed two-stream chained segmentation pipeline and the PSNN model can significantly improve the quantitative performance over the previous state-of-the-art work by 11% in absolute Dice score (DSC) (from 0.654 to 0.764) and, at the same time, reducing the Hausdorff distance from 129 mm to 47 mm.
Weakly Supervised Universal Fracture Detection in Pelvic X-rays
Sep 04, 2019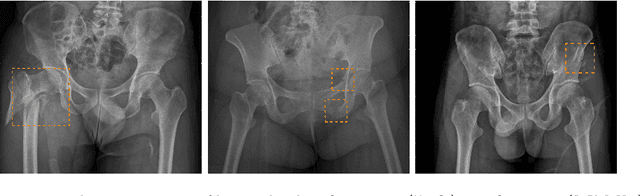
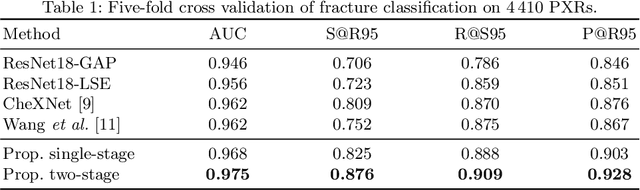
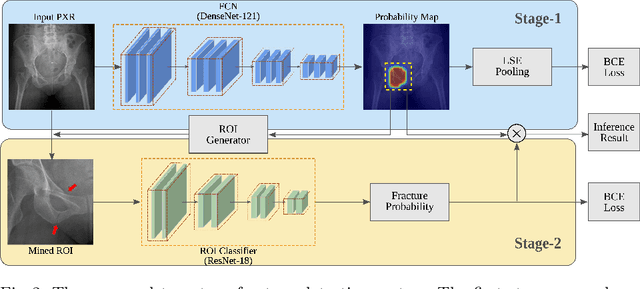
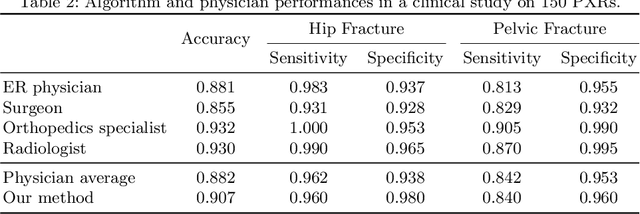
Abstract:Hip and pelvic fractures are serious injuries with life-threatening complications. However, diagnostic errors of fractures in pelvic X-rays (PXRs) are very common, driving the demand for computer-aided diagnosis (CAD) solutions. A major challenge lies in the fact that fractures are localized patterns that require localized analyses. Unfortunately, the PXRs residing in hospital picture archiving and communication system do not typically specify region of interests. In this paper, we propose a two-stage hip and pelvic fracture detection method that executes localized fracture classification using weakly supervised ROI mining. The first stage uses a large capacity fully-convolutional network, i.e., deep with high levels of abstraction, in a multiple instance learning setting to automatically mine probable true positive and definite hard negative ROIs from the whole PXR in the training data. The second stage trains a smaller capacity model, i.e., shallower and more generalizable, with the mined ROIs to perform localized analyses to classify fractures. During inference, our method detects hip and pelvic fractures in one pass by chaining the probability outputs of the two stages together. We evaluate our method on 4 410 PXRs, reporting an area under the ROC curve value of 0.975, the highest among state-of-the-art fracture detection methods. Moreover, we show that our two-stage approach can perform comparably to human physicians (even outperforming emergency physicians and surgeons), in a preliminary reader study of 23 readers.
Radiotherapy Target Contouring with Convolutional Gated Graph Neural Network
Apr 05, 2019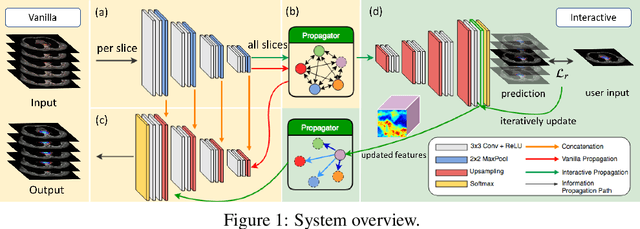
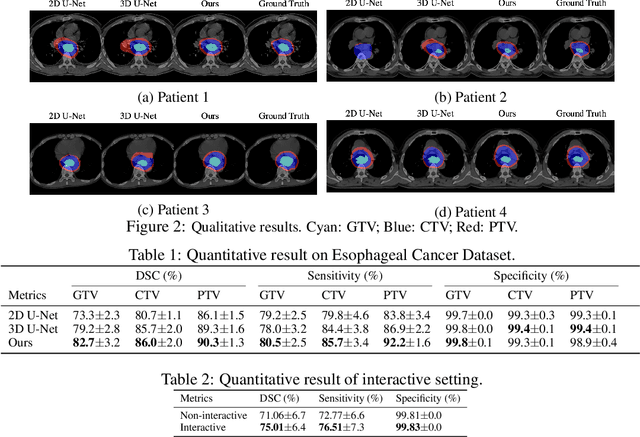
Abstract:Tomography medical imaging is essential in the clinical workflow of modern cancer radiotherapy. Radiation oncologists identify cancerous tissues, applying delineation on treatment regions throughout all image slices. This kind of task is often formulated as a volumetric segmentation task by means of 3D convolutional networks with considerable computational cost. Instead, inspired by the treating methodology of considering meaningful information across slices, we used Gated Graph Neural Network to frame this problem more efficiently. More specifically, we propose convolutional recurrent Gated Graph Propagator (GGP) to propagate high-level information through image slices, with learnable adjacency weighted matrix. Furthermore, as physicians often investigate a few specific slices to refine their decision, we model this slice-wise interaction procedure to further improve our segmentation result. This can be set by editing any slice effortlessly as updating predictions of other slices using GGP. To evaluate our method, we collect an Esophageal Cancer Radiotherapy Target Treatment Contouring dataset of 81 patients which includes tomography images with radiotherapy target. On this dataset, our convolutional graph network produces state-of-the-art results and outperforms the baselines. With the addition of interactive setting, performance is improved even further. Our method has the potential to be easily applied to diverse kinds of medical tasks with volumetric images. Incorporating both the ability to make a feasible prediction and to consider the human interactive input, the proposed method is suitable for clinical scenarios.
Spatial-Temporal Convolutional LSTMs for Tumor Growth Prediction by Learning 4D Longitudinal Patient Data
Feb 23, 2019
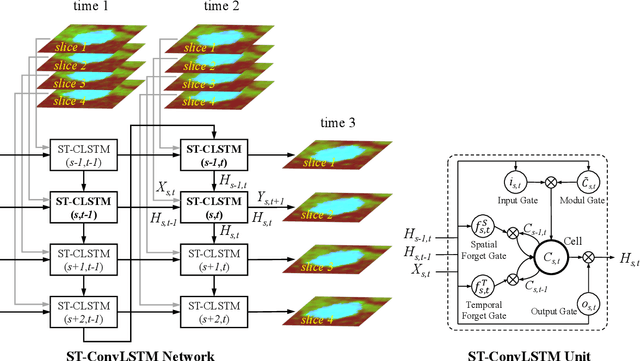
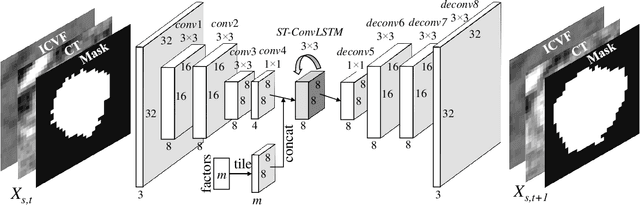

Abstract:Prognostic tumor growth modeling via medical imaging observations is a challenging yet important problem in precision and predictive medicine. Traditionally, this problem is tackled through mathematical modeling and evaluated using relatively small patient datasets. Recent advances of convolutional networks (ConvNets) have demonstrated their higher accuracy than mathematical models in predicting future tumor volumes. This indicates that deep learning may have great potentials on addressing such problem. The state-of-the-art work models the cell invasion and mass-effect of tumor growth by training separate ConvNets on 2D image patches. Nevertheless such a 2D modeling approach cannot make full use of the spatial-temporal imaging context of the tumor's longitudinal 4D (3D + time) patient data. Moreover, previous methods are incapable to predict clinically-relevant tumor properties, other than the tumor volumes. In this paper, we exploit to formulate the tumor growth process through convolutional LSTMs (ConvLSTM) that extract tumor's static imaging appearances and simultaneously capture its temporal dynamic changes within a single network. We extend ConvLSTM into the spatial-temporal domain (ST-ConvLSTM) by jointly learning the inter-slice 3D contexts and the longitudinal dynamics. Our approach can incorporate other non-imaging patient information in an end-to-end trainable manner. Experiments are conducted on the largest 4D longitudinal tumor dataset of 33 patients to date. Results validate that the proposed ST-ConvLSTM model produces a Dice score of 83.2%+-5.1% and a RVD of 11.2%+-10.8%, both statistically significantly outperforming (p<0.05) other compared methods of traditional linear model, ConvLSTM, and generative adversarial network (GAN) under the metric of predicting future tumor volumes. Last, our new method enables the prediction of both cell density and CT intensity numbers.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge