M. Jorge Cardoso
School of Biomedical Engineering and Imaging Science, King's College London, UK
Transformer-based normative modelling for anomaly detection of early schizophrenia
Dec 08, 2022Abstract:Despite the impact of psychiatric disorders on clinical health, early-stage diagnosis remains a challenge. Machine learning studies have shown that classifiers tend to be overly narrow in the diagnosis prediction task. The overlap between conditions leads to high heterogeneity among participants that is not adequately captured by classification models. To address this issue, normative approaches have surged as an alternative method. By using a generative model to learn the distribution of healthy brain data patterns, we can identify the presence of pathologies as deviations or outliers from the distribution learned by the model. In particular, deep generative models showed great results as normative models to identify neurological lesions in the brain. However, unlike most neurological lesions, psychiatric disorders present subtle changes widespread in several brain regions, making these alterations challenging to identify. In this work, we evaluate the performance of transformer-based normative models to detect subtle brain changes expressed in adolescents and young adults. We trained our model on 3D MRI scans of neurotypical individuals (N=1,765). Then, we obtained the likelihood of neurotypical controls and psychiatric patients with early-stage schizophrenia from an independent dataset (N=93) from the Human Connectome Project. Using the predicted likelihood of the scans as a proxy for a normative score, we obtained an AUROC of 0.82 when assessing the difference between controls and individuals with early-stage schizophrenia. Our approach surpassed recent normative methods based on brain age and Gaussian Process, showing the promising use of deep generative models to help in individualised analyses.
Denoising Diffusion Models for Out-of-Distribution Detection
Nov 30, 2022Abstract:Out-of-distribution detection is crucial to the safe deployment of machine learning systems. Currently, the state-of-the-art in unsupervised out-of-distribution detection is dominated by generative-based approaches that make use of estimates of the likelihood or other measurements from a generative model. Reconstruction-based methods offer an alternative approach, in which a measure of reconstruction error is used to determine if a sample is out-of-distribution. However, reconstruction-based approaches are less favoured, as they require careful tuning of the model's information bottleneck - such as the size of the latent dimension - to produce good results. In this work, we exploit the view of denoising diffusion probabilistic models (DDPM) as denoising autoencoders where the bottleneck is controlled externally, by means of the amount of noise applied. We propose to use DDPMs to reconstruct an input that has been noised to a range of noise levels, and use the resulting multi-dimensional reconstruction error to classify out-of-distribution inputs. Our approach outperforms not only reconstruction-based methods, but also state-of-the-art generative-based approaches.
MONAI: An open-source framework for deep learning in healthcare
Nov 04, 2022



Abstract:Artificial Intelligence (AI) is having a tremendous impact across most areas of science. Applications of AI in healthcare have the potential to improve our ability to detect, diagnose, prognose, and intervene on human disease. For AI models to be used clinically, they need to be made safe, reproducible and robust, and the underlying software framework must be aware of the particularities (e.g. geometry, physiology, physics) of medical data being processed. This work introduces MONAI, a freely available, community-supported, and consortium-led PyTorch-based framework for deep learning in healthcare. MONAI extends PyTorch to support medical data, with a particular focus on imaging, and provide purpose-specific AI model architectures, transformations and utilities that streamline the development and deployment of medical AI models. MONAI follows best practices for software-development, providing an easy-to-use, robust, well-documented, and well-tested software framework. MONAI preserves the simple, additive, and compositional approach of its underlying PyTorch libraries. MONAI is being used by and receiving contributions from research, clinical and industrial teams from around the world, who are pursuing applications spanning nearly every aspect of healthcare.
Brain Imaging Generation with Latent Diffusion Models
Sep 15, 2022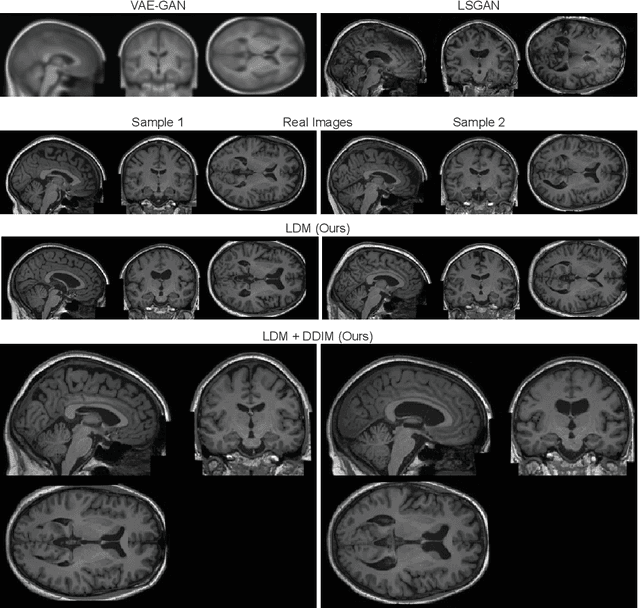
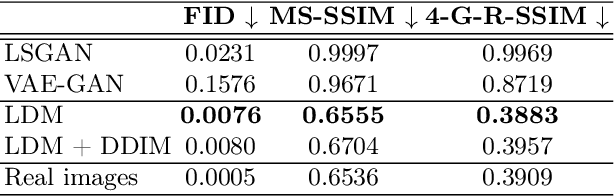
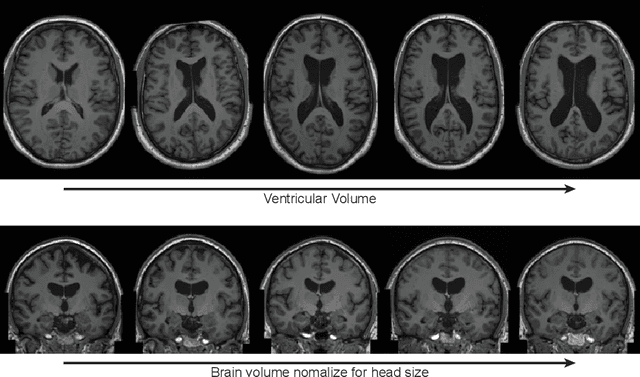
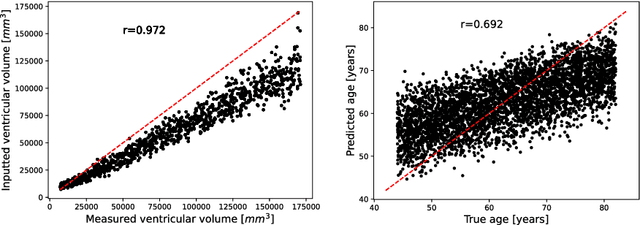
Abstract:Deep neural networks have brought remarkable breakthroughs in medical image analysis. However, due to their data-hungry nature, the modest dataset sizes in medical imaging projects might be hindering their full potential. Generating synthetic data provides a promising alternative, allowing to complement training datasets and conducting medical image research at a larger scale. Diffusion models recently have caught the attention of the computer vision community by producing photorealistic synthetic images. In this study, we explore using Latent Diffusion Models to generate synthetic images from high-resolution 3D brain images. We used T1w MRI images from the UK Biobank dataset (N=31,740) to train our models to learn about the probabilistic distribution of brain images, conditioned on covariables, such as age, sex, and brain structure volumes. We found that our models created realistic data, and we could use the conditioning variables to control the data generation effectively. Besides that, we created a synthetic dataset with 100,000 brain images and made it openly available to the scientific community.
Fitting Segmentation Networks on Varying Image Resolutions using Splatting
Jun 15, 2022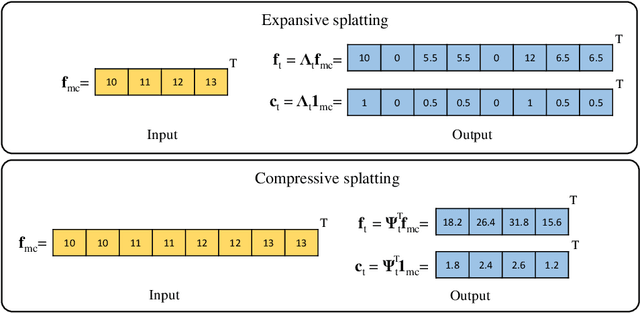
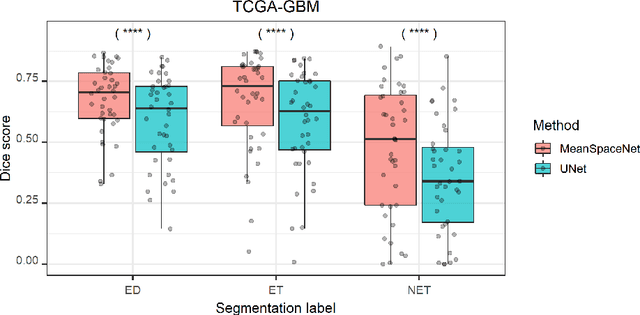
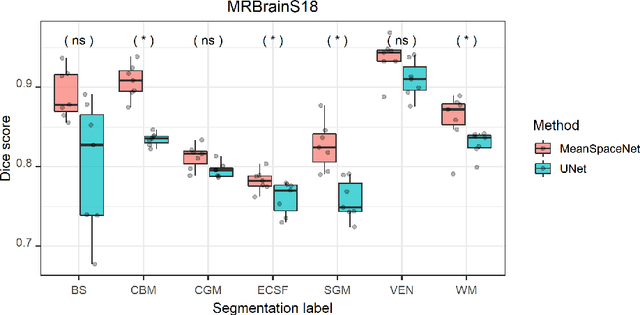
Abstract:Data used in image segmentation are not always defined on the same grid. This is particularly true for medical images, where the resolution, field-of-view and orientation can differ across channels and subjects. Images and labels are therefore commonly resampled onto the same grid, as a pre-processing step. However, the resampling operation introduces partial volume effects and blurring, thereby changing the effective resolution and reducing the contrast between structures. In this paper we propose a splat layer, which automatically handles resolution mismatches in the input data. This layer pushes each image onto a mean space where the forward pass is performed. As the splat operator is the adjoint to the resampling operator, the mean-space prediction can be pulled back to the native label space, where the loss function is computed. Thus, the need for explicit resolution adjustment using interpolation is removed. We show on two publicly available datasets, with simulated and real multi-modal magnetic resonance images, that this model improves segmentation results compared to resampling as a pre-processing step.
Fast Unsupervised Brain Anomaly Detection and Segmentation with Diffusion Models
Jun 07, 2022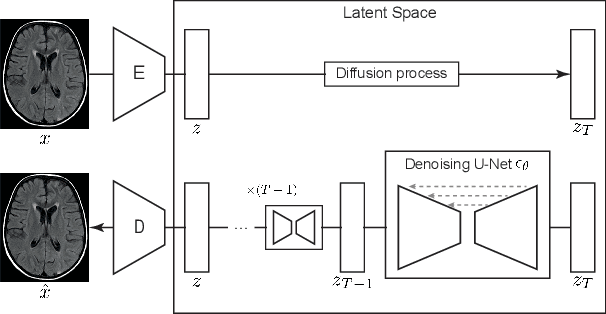
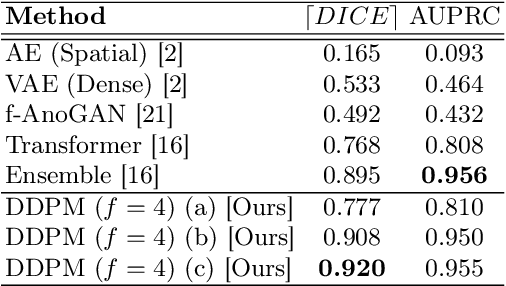
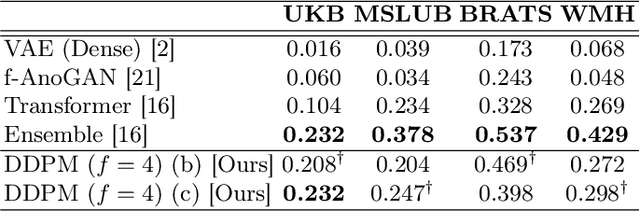
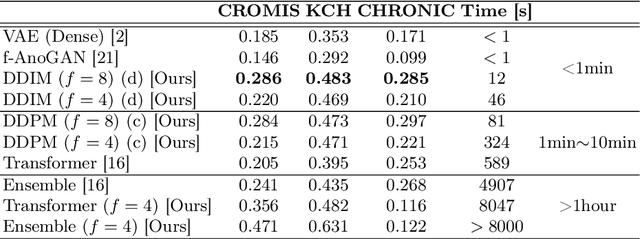
Abstract:Deep generative models have emerged as promising tools for detecting arbitrary anomalies in data, dispensing with the necessity for manual labelling. Recently, autoregressive transformers have achieved state-of-the-art performance for anomaly detection in medical imaging. Nonetheless, these models still have some intrinsic weaknesses, such as requiring images to be modelled as 1D sequences, the accumulation of errors during the sampling process, and the significant inference times associated with transformers. Denoising diffusion probabilistic models are a class of non-autoregressive generative models recently shown to produce excellent samples in computer vision (surpassing Generative Adversarial Networks), and to achieve log-likelihoods that are competitive with transformers while having fast inference times. Diffusion models can be applied to the latent representations learnt by autoencoders, making them easily scalable and great candidates for application to high dimensional data, such as medical images. Here, we propose a method based on diffusion models to detect and segment anomalies in brain imaging. By training the models on healthy data and then exploring its diffusion and reverse steps across its Markov chain, we can identify anomalous areas in the latent space and hence identify anomalies in the pixel space. Our diffusion models achieve competitive performance compared with autoregressive approaches across a series of experiments with 2D CT and MRI data involving synthetic and real pathological lesions with much reduced inference times, making their usage clinically viable.
Metrics reloaded: Pitfalls and recommendations for image analysis validation
Jun 03, 2022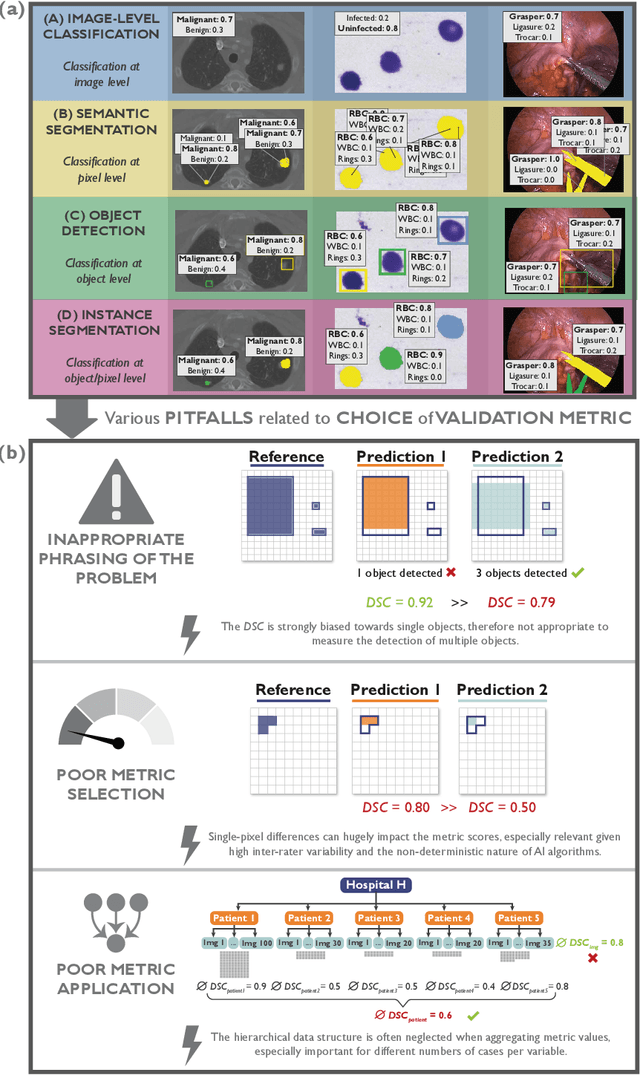
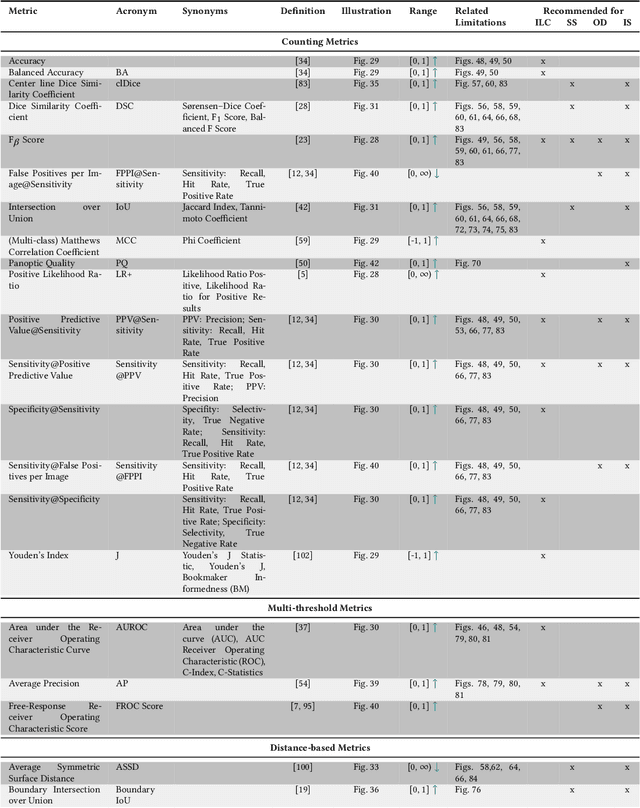
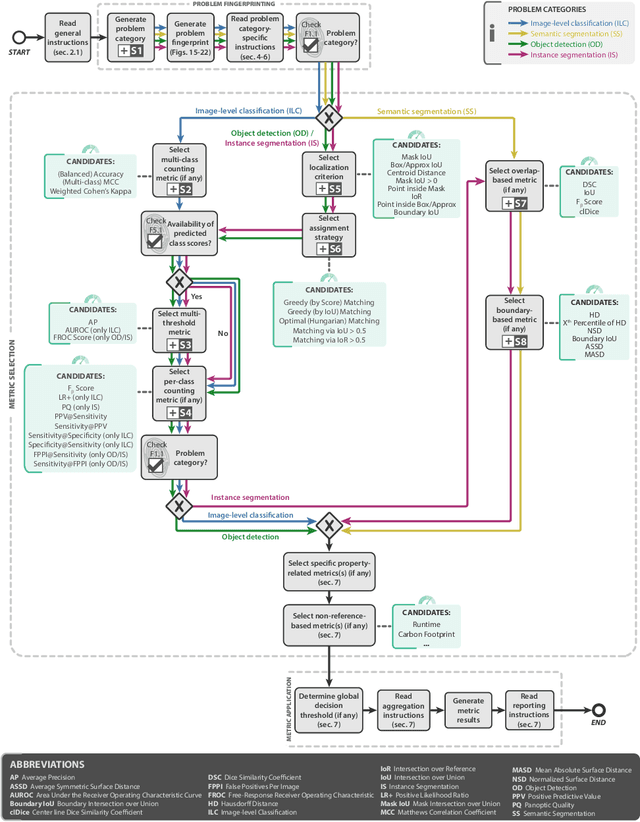
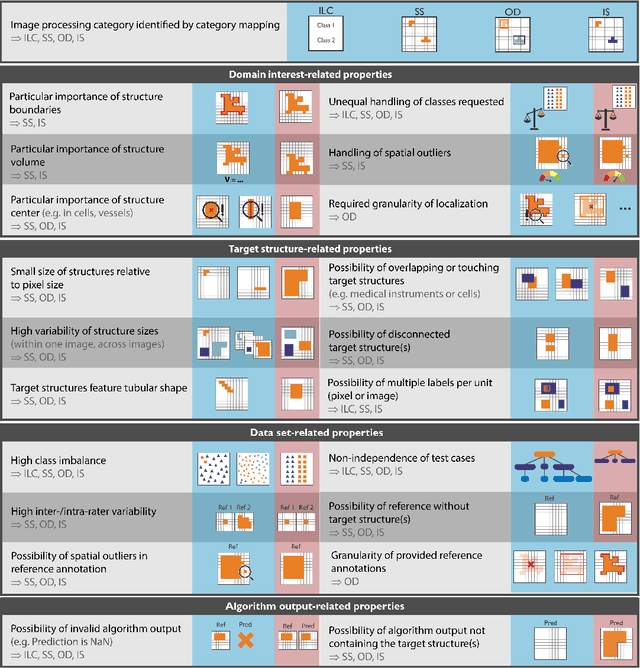
Abstract:The field of automatic biomedical image analysis crucially depends on robust and meaningful performance metrics for algorithm validation. Current metric usage, however, is often ill-informed and does not reflect the underlying domain interest. Here, we present a comprehensive framework that guides researchers towards choosing performance metrics in a problem-aware manner. Specifically, we focus on biomedical image analysis problems that can be interpreted as a classification task at image, object or pixel level. The framework first compiles domain interest-, target structure-, data set- and algorithm output-related properties of a given problem into a problem fingerprint, while also mapping it to the appropriate problem category, namely image-level classification, semantic segmentation, instance segmentation, or object detection. It then guides users through the process of selecting and applying a set of appropriate validation metrics while making them aware of potential pitfalls related to individual choices. In this paper, we describe the current status of the Metrics Reloaded recommendation framework, with the goal of obtaining constructive feedback from the image analysis community. The current version has been developed within an international consortium of more than 60 image analysis experts and will be made openly available as a user-friendly toolkit after community-driven optimization.
MONAI Label: A framework for AI-assisted Interactive Labeling of 3D Medical Images
Mar 23, 2022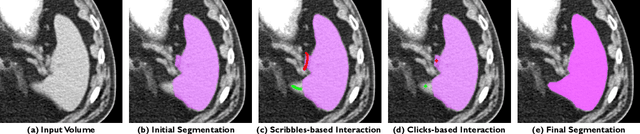
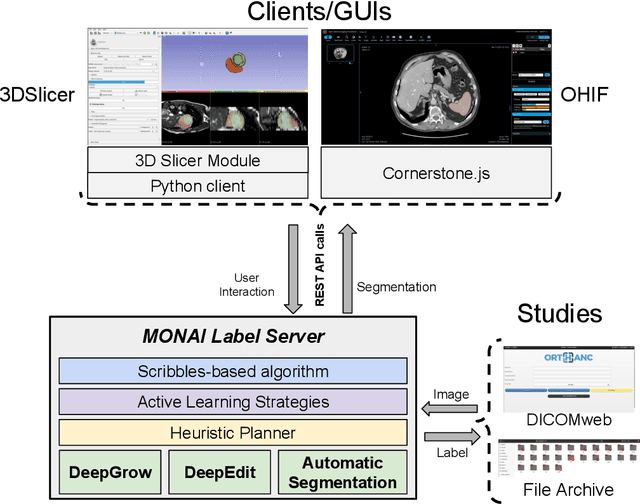
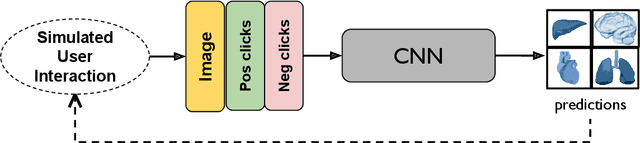
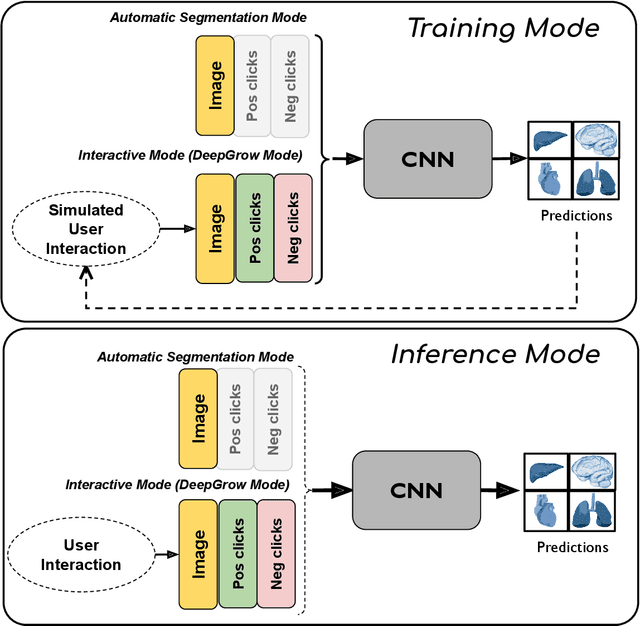
Abstract:The lack of annotated datasets is a major challenge in training new task-specific supervised AI algorithms as manual annotation is expensive and time-consuming. To address this problem, we present MONAI Label, a free and open-source platform that facilitates the development of AI-based applications that aim at reducing the time required to annotate 3D medical image datasets. Through MONAI Label researchers can develop annotation applications focusing on their domain of expertise. It allows researchers to readily deploy their apps as services, which can be made available to clinicians via their preferred user-interface. Currently, MONAI Label readily supports locally installed (3DSlicer) and web-based (OHIF) frontends, and offers two Active learning strategies to facilitate and speed up the training of segmentation algorithms. MONAI Label allows researchers to make incremental improvements to their labeling apps by making them available to other researchers and clinicians alike. Lastly, MONAI Label provides sample labeling apps, namely DeepEdit and DeepGrow, demonstrating dramatically reduced annotation times.
Deep forecasting of translational impact in medical research
Oct 17, 2021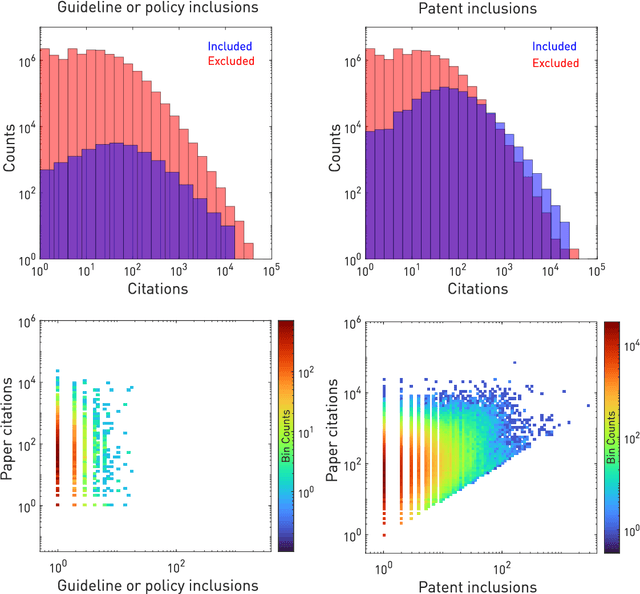
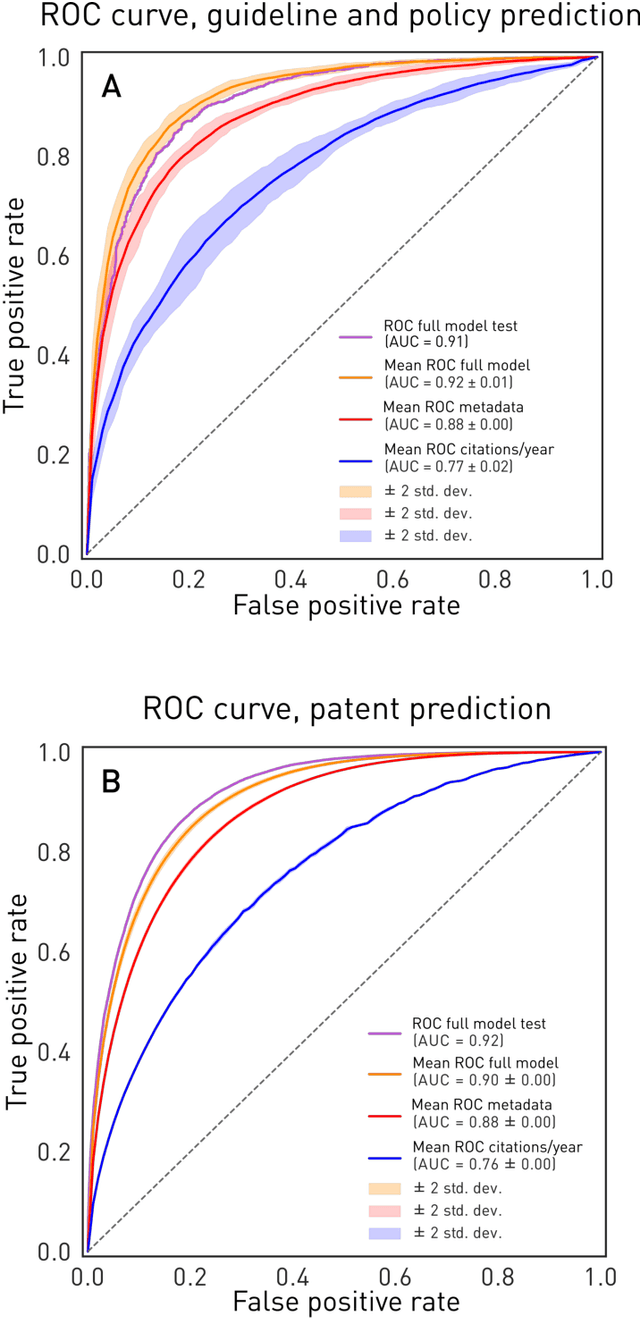
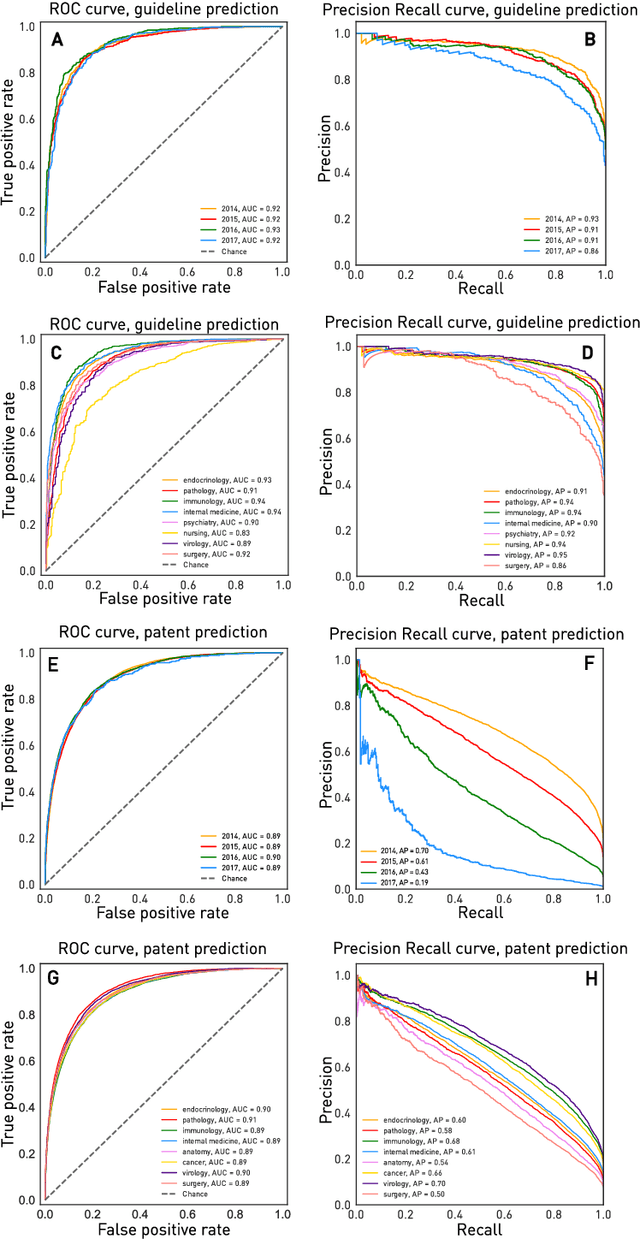
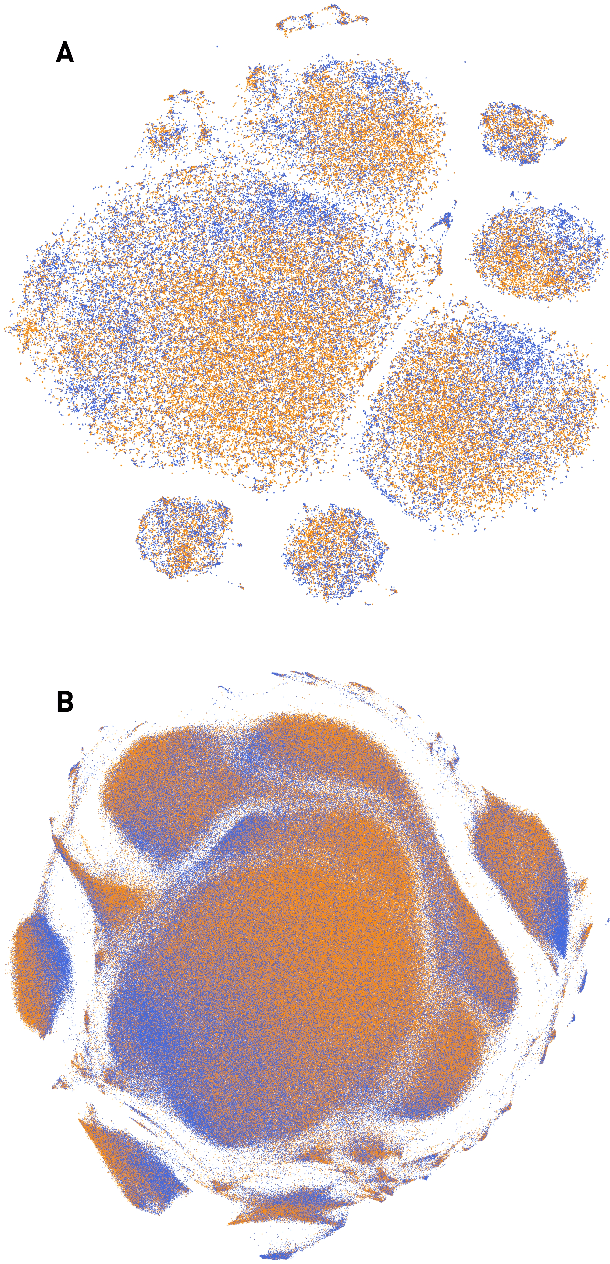
Abstract:The value of biomedical research--a $1.7 trillion annual investment--is ultimately determined by its downstream, real-world impact. Current objective predictors of impact rest on proxy, reductive metrics of dissemination, such as paper citation rates, whose relation to real-world translation remains unquantified. Here we sought to determine the comparative predictability of future real-world translation--as indexed by inclusion in patents, guidelines or policy documents--from complex models of the abstract-level content of biomedical publications versus citations and publication meta-data alone. We develop a suite of representational and discriminative mathematical models of multi-scale publication data, quantifying predictive performance out-of-sample, ahead-of-time, across major biomedical domains, using the entire corpus of biomedical research captured by Microsoft Academic Graph from 1990 to 2019, encompassing 43.3 million papers across all domains. We show that citations are only moderately predictive of translational impact as judged by inclusion in patents, guidelines, or policy documents. By contrast, high-dimensional models of publication titles, abstracts and metadata exhibit high fidelity (AUROC > 0.9), generalise across time and thematic domain, and transfer to the task of recognising papers of Nobel Laureates. The translational impact of a paper indexed by inclusion in patents, guidelines, or policy documents can be predicted--out-of-sample and ahead-of-time--with substantially higher fidelity from complex models of its abstract-level content than from models of publication meta-data or citation metrics. We argue that content-based models of impact are superior in performance to conventional, citation-based measures, and sustain a stronger evidence-based claim to the objective measurement of translational potential.
A Decoupled Uncertainty Model for MRI Segmentation Quality Estimation
Sep 06, 2021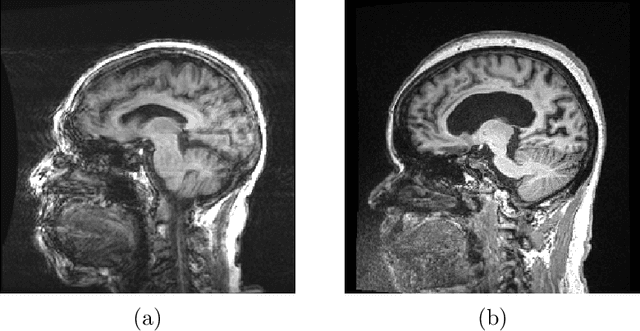
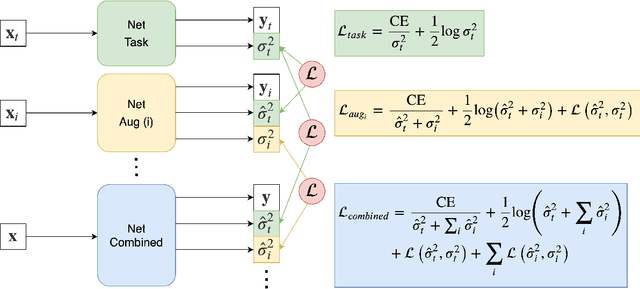
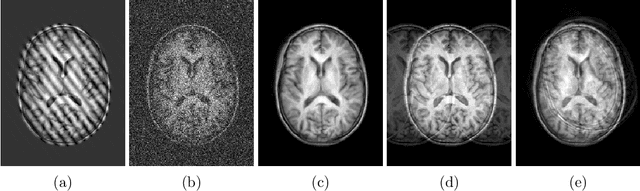
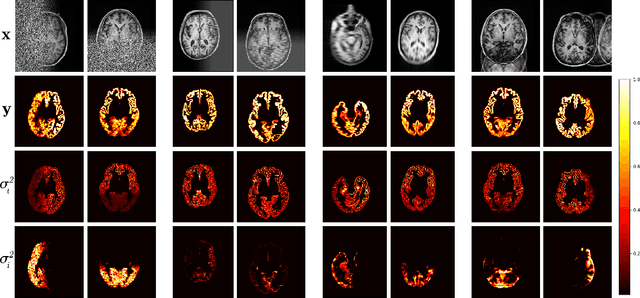
Abstract:Quality control (QC) of MR images is essential to ensure that downstream analyses such as segmentation can be performed successfully. Currently, QC is predominantly performed visually and subjectively, at significant time and operator cost. We aim to automate the process using a probabilistic network that estimates segmentation uncertainty through a heteroscedastic noise model, providing a measure of task-specific quality. By augmenting training images with k-space artefacts, we propose a novel CNN architecture to decouple sources of uncertainty related to the task and different k-space artefacts in a self-supervised manner. This enables the prediction of separate uncertainties for different types of data degradation. While the uncertainty predictions reflect the presence and severity of artefacts, the network provides more robust and generalisable segmentation predictions given the quality of the data. We show that models trained with artefact augmentation provide informative measures of uncertainty on both simulated artefacts and problematic real-world images identified by human raters, both qualitatively and quantitatively in the form of error bars on volume measurements. Relating artefact uncertainty to segmentation Dice scores, we observe that our uncertainty predictions provide a better estimate of MRI quality from the point of view of the task (gray matter segmentation) compared to commonly used metrics of quality including signal-to-noise ratio (SNR) and contrast-to-noise ratio (CNR), hence providing a real-time quality metric indicative of segmentation quality.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge