Eric I-Chao Chang
Predicting breast tumor proliferation from whole-slide images: the TUPAC16 challenge
Jul 22, 2018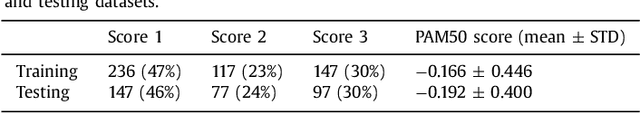
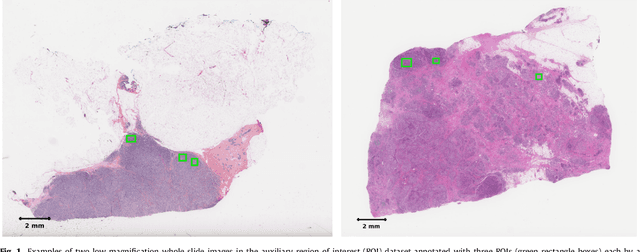
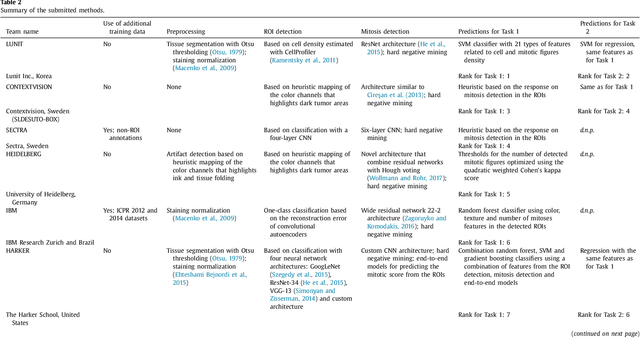
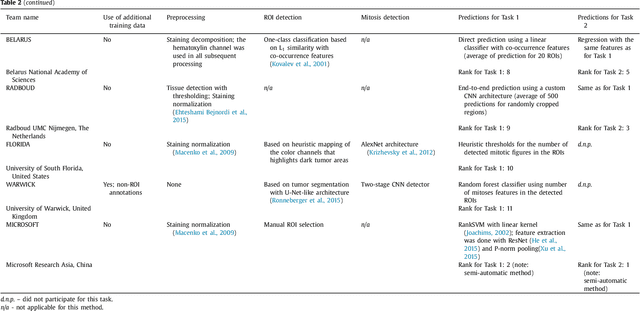
Abstract:Tumor proliferation is an important biomarker indicative of the prognosis of breast cancer patients. Assessment of tumor proliferation in a clinical setting is highly subjective and labor-intensive task. Previous efforts to automate tumor proliferation assessment by image analysis only focused on mitosis detection in predefined tumor regions. However, in a real-world scenario, automatic mitosis detection should be performed in whole-slide images (WSIs) and an automatic method should be able to produce a tumor proliferation score given a WSI as input. To address this, we organized the TUmor Proliferation Assessment Challenge 2016 (TUPAC16) on prediction of tumor proliferation scores from WSIs. The challenge dataset consisted of 500 training and 321 testing breast cancer histopathology WSIs. In order to ensure fair and independent evaluation, only the ground truth for the training dataset was provided to the challenge participants. The first task of the challenge was to predict mitotic scores, i.e., to reproduce the manual method of assessing tumor proliferation by a pathologist. The second task was to predict the gene expression based PAM50 proliferation scores from the WSI. The best performing automatic method for the first task achieved a quadratic-weighted Cohen's kappa score of $\kappa$ = 0.567, 95% CI [0.464, 0.671] between the predicted scores and the ground truth. For the second task, the predictions of the top method had a Spearman's correlation coefficient of r = 0.617, 95% CI [0.581 0.651] with the ground truth. This was the first study that investigated tumor proliferation assessment from WSIs. The achieved results are promising given the difficulty of the tasks and weakly-labelled nature of the ground truth. However, further research is needed to improve the practical utility of image analysis methods for this task.
Unsupervised Learning for Cell-level Visual Representation in Histopathology Images with Generative Adversarial Networks
Jul 07, 2018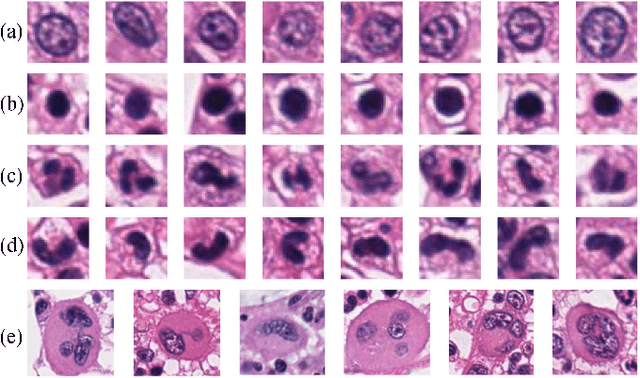

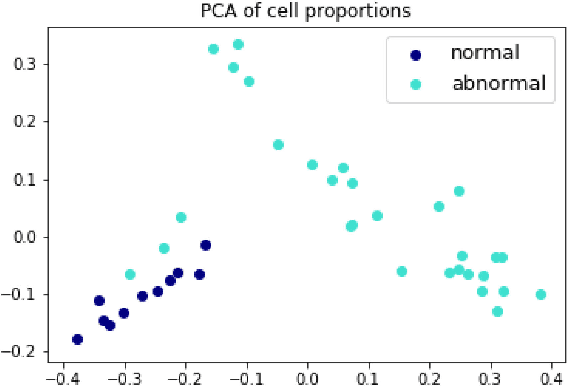
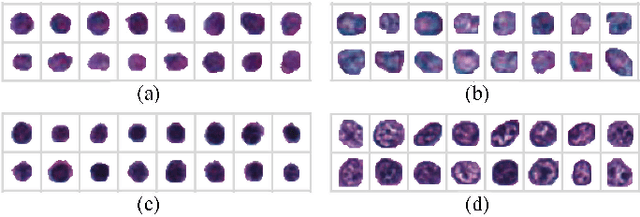
Abstract:The visual attributes of cells, such as the nuclear morphology and chromatin openness, are critical for histopathology image analysis. By learning cell-level visual representation, we can obtain a rich mix of features that are highly reusable for various tasks, such as cell-level classification, nuclei segmentation, and cell counting. In this paper, we propose a unified generative adversarial networks architecture with a new formulation of loss to perform robust cell-level visual representation learning in an unsupervised setting. Our model is not only label-free and easily trained but also capable of cell-level unsupervised classification with interpretable visualization, which achieves promising results in the unsupervised classification of bone marrow cellular components. Based on the proposed cell-level visual representation learning, we further develop a pipeline that exploits the varieties of cellular elements to perform histopathology image classification, the advantages of which are demonstrated on bone marrow datasets.
Unsupervised End-to-end Learning for Deformable Medical Image Registration
Jan 20, 2018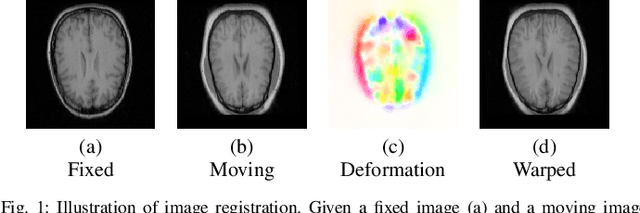
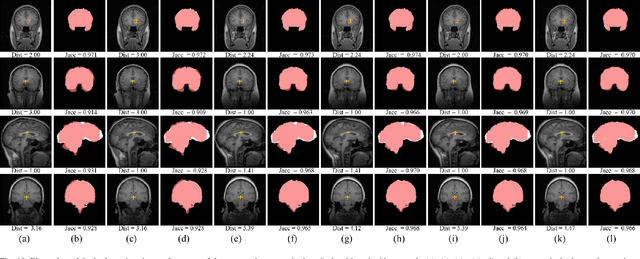
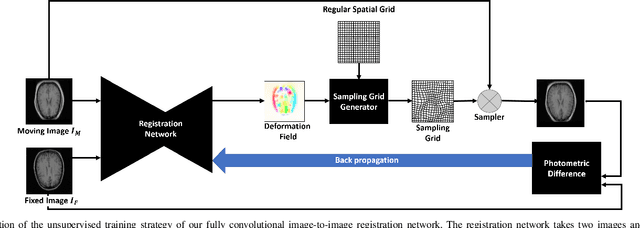
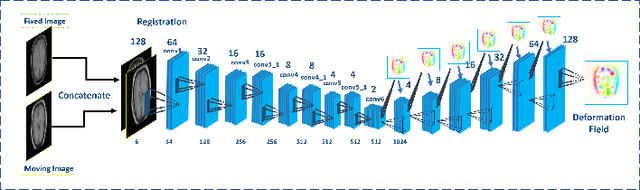
Abstract:We propose a registration algorithm for 2D CT/MRI medical images with a new unsupervised end-to-end strategy using convolutional neural networks. The contributions of our algorithm are threefold: (1) We transplant traditional image registration algorithms to an end-to-end convolutional neural network framework, while maintaining the unsupervised nature of image registration problems. The image-to-image integrated framework can simultaneously learn both image features and transformation matrix for registration. (2) Training with additional data without any label can further improve the registration performance by approximately 10 %. (3) The registration speed is 100x faster than traditional methods. The proposed network is easy to implement and can be trained efficiently. Experiments demonstrate that our system achieves state-of-the-art results on 2D brain registration and achieves comparable results on 2D liver registration. It can be extended to register other organs beyond liver and brain such as kidney, lung, and heart.
Gland Instance Segmentation Using Deep Multichannel Neural Networks
Nov 23, 2017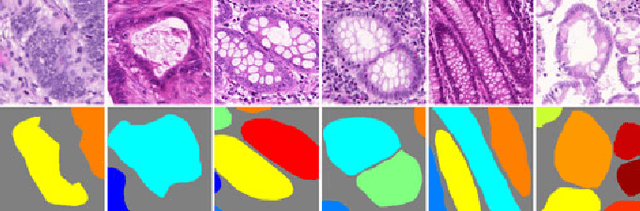
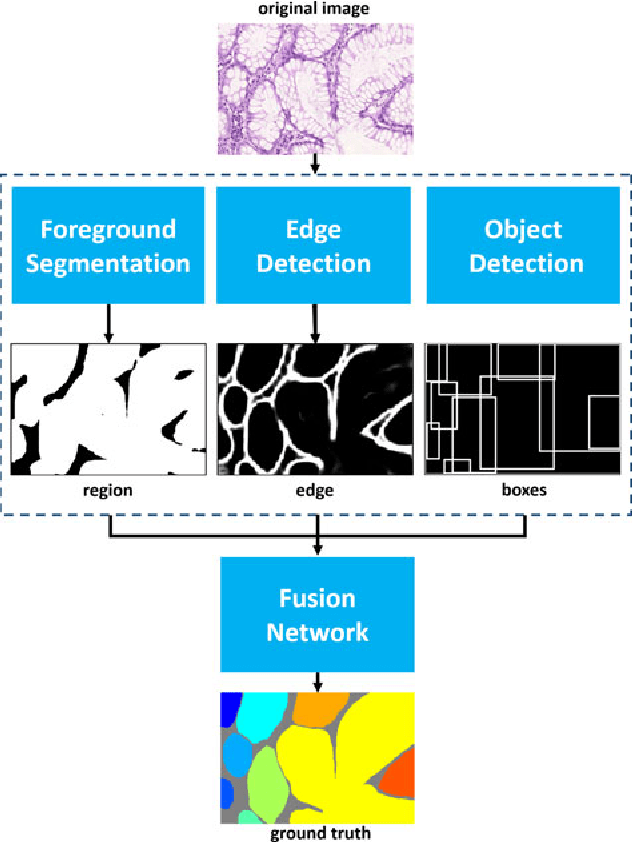
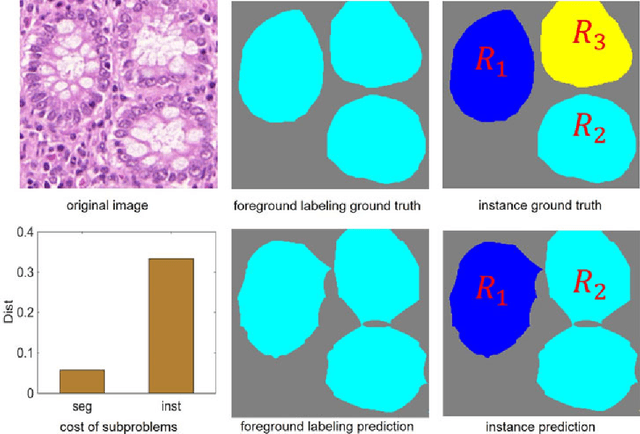
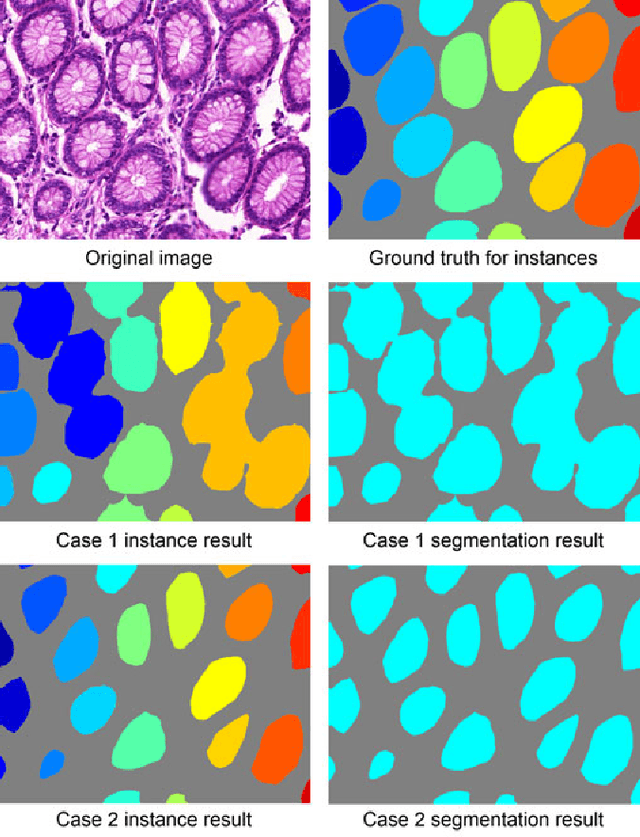
Abstract:Objective: A new image instance segmentation method is proposed to segment individual glands (instances) in colon histology images. This process is challenging since the glands not only need to be segmented from a complex background, they must also be individually identified. Methods: We leverage the idea of image-to-image prediction in recent deep learning by designing an algorithm that automatically exploits and fuses complex multichannel information - regional, location, and boundary cues - in gland histology images. Our proposed algorithm, a deep multichannel framework, alleviates heavy feature design due to the use of convolutional neural networks and is able to meet multifarious requirements by altering channels. Results: Compared with methods reported in the 2015 MICCAI Gland Segmentation Challenge and other currently prevalent instance segmentation methods, we observe state-of-the-art results based on the evaluation metrics. Conclusion: The proposed deep multichannel algorithm is an effective method for gland instance segmentation. Significance: The generalization ability of our model not only enable the algorithm to solve gland instance segmentation problems, but the channel is also alternative that can be replaced for a specific task.
* arXiv admin note: substantial text overlap with arXiv:1607.04889
Sleep Stage Classification Based on Multi-level Feature Learning and Recurrent Neural Networks via Wearable Device
Nov 02, 2017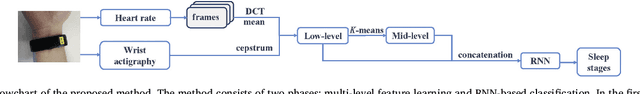
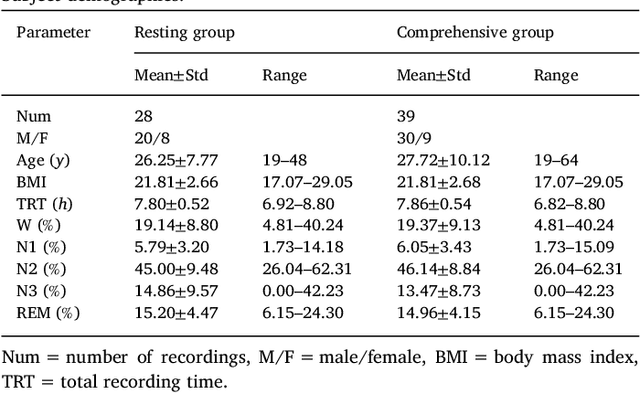
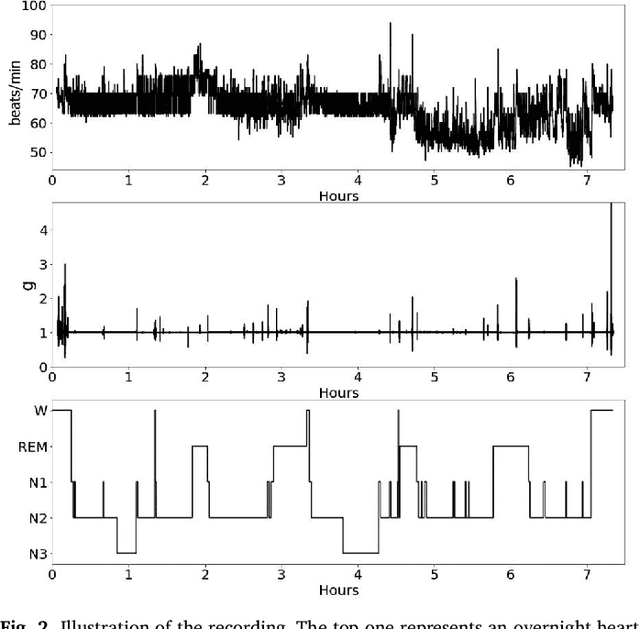
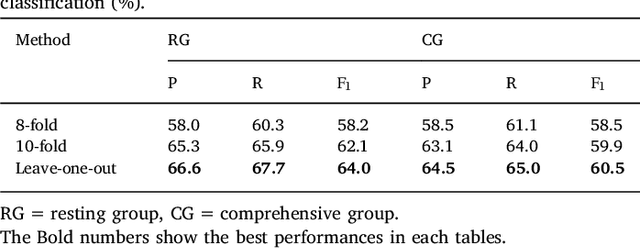
Abstract:This paper proposes a practical approach for automatic sleep stage classification based on a multi-level feature learning framework and Recurrent Neural Network (RNN) classifier using heart rate and wrist actigraphy derived from a wearable device. The feature learning framework is designed to extract low- and mid-level features. Low-level features capture temporal and frequency domain properties and mid-level features learn compositions and structural information of signals. Since sleep staging is a sequential problem with long-term dependencies, we take advantage of RNNs with Bidirectional Long Short-Term Memory (BLSTM) architectures for sequence data learning. To simulate the actual situation of daily sleep, experiments are conducted with a resting group in which sleep is recorded in resting state, and a comprehensive group in which both resting sleep and non-resting sleep are included.We evaluate the algorithm based on an eight-fold cross validation to classify five sleep stages (W, N1, N2, N3, and REM). The proposed algorithm achieves weighted precision, recall and F1 score of 58.0%, 60.3%, and 58.2% in the resting group and 58.5%, 61.1%, and 58.5% in the comprehensive group, respectively. Various comparison experiments demonstrate the effectiveness of feature learning and BLSTM. We further explore the influence of depth and width of RNNs on performance. Our method is specially proposed for wearable devices and is expected to be applicable for long-term sleep monitoring at home. Without using too much prior domain knowledge, our method has the potential to generalize sleep disorder detection.
Learning Multi-level Features For Sensor-based Human Action Recognition
Sep 02, 2017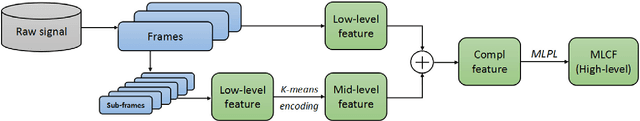
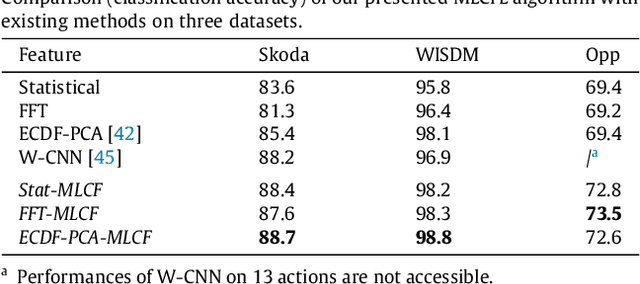
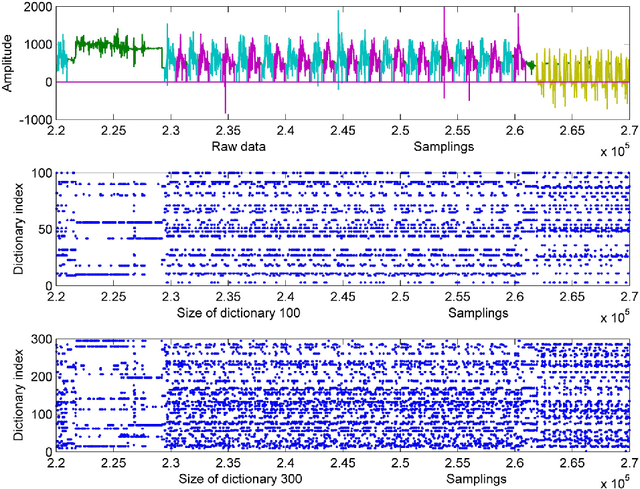
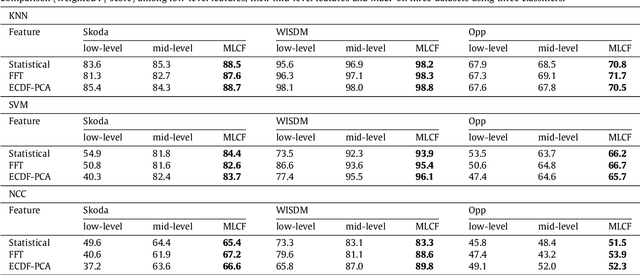
Abstract:This paper proposes a multi-level feature learning framework for human action recognition using a single body-worn inertial sensor. The framework consists of three phases, respectively designed to analyze signal-based (low-level), components (mid-level) and semantic (high-level) information. Low-level features capture the time and frequency domain property while mid-level representations learn the composition of the action. The Max-margin Latent Pattern Learning (MLPL) method is proposed to learn high-level semantic descriptions of latent action patterns as the output of our framework. The proposed method achieves the state-of-the-art performances, 88.7%, 98.8% and 72.6% (weighted F1 score) respectively, on Skoda, WISDM and OPP datasets.
* 26 pages, 23 figures
Constrained Deep Weak Supervision for Histopathology Image Segmentation
Jan 03, 2017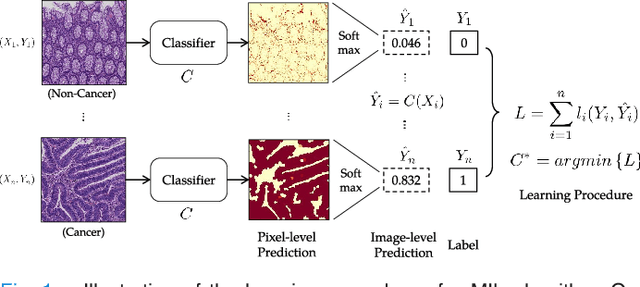

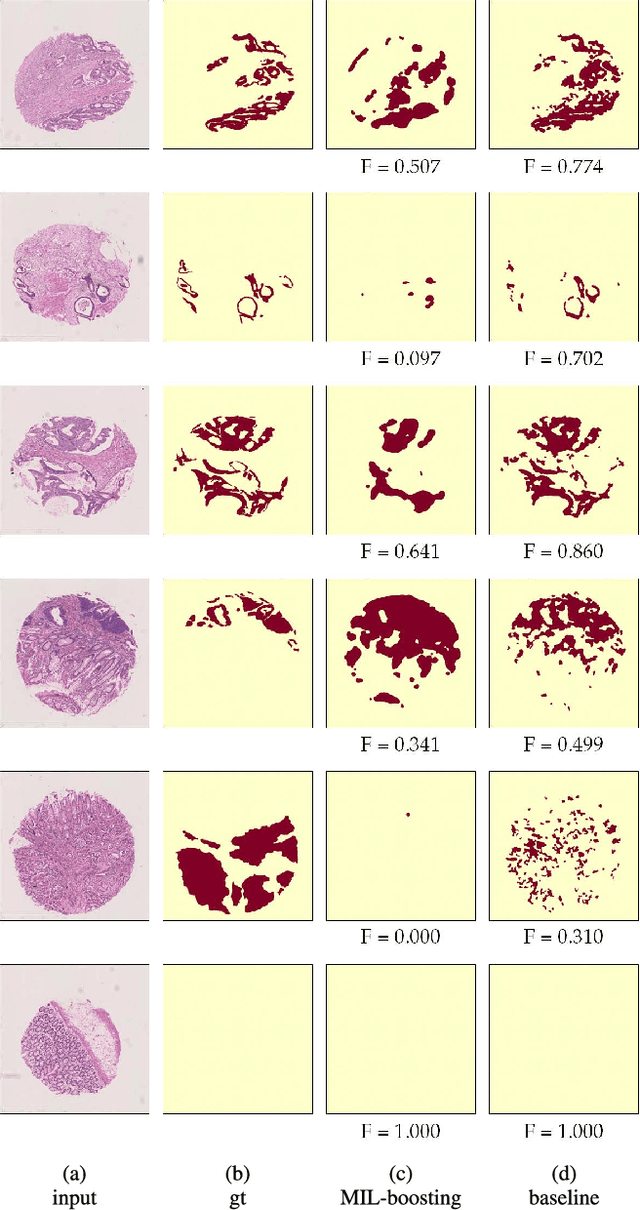
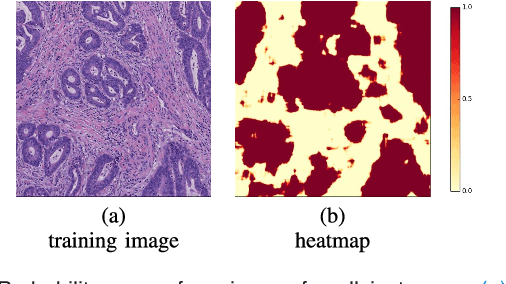
Abstract:In this paper, we develop a new weakly-supervised learning algorithm to learn to segment cancerous regions in histopathology images. Our work is under a multiple instance learning framework (MIL) with a new formulation, deep weak supervision (DWS); we also propose an effective way to introduce constraints to our neural networks to assist the learning process. The contributions of our algorithm are threefold: (1) We build an end-to-end learning system that segments cancerous regions with fully convolutional networks (FCN) in which image-to-image weakly-supervised learning is performed. (2) We develop a deep week supervision formulation to exploit multi-scale learning under weak supervision within fully convolutional networks. (3) Constraints about positive instances are introduced in our approach to effectively explore additional weakly-supervised information that is easy to obtain and enjoys a significant boost to the learning process. The proposed algorithm, abbreviated as DWS-MIL, is easy to implement and can be trained efficiently. Our system demonstrates state-of-the-art results on large-scale histopathology image datasets and can be applied to various applications in medical imaging beyond histopathology images such as MRI, CT, and ultrasound images.
End-to-End Subtitle Detection and Recognition for Videos in East Asian Languages via CNN Ensemble with Near-Human-Level Performance
Nov 18, 2016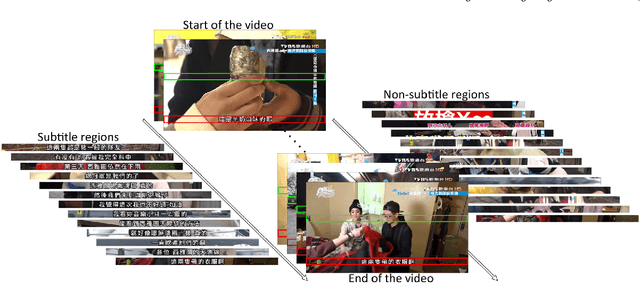
Abstract:In this paper, we propose an innovative end-to-end subtitle detection and recognition system for videos in East Asian languages. Our end-to-end system consists of multiple stages. Subtitles are firstly detected by a novel image operator based on the sequence information of consecutive video frames. Then, an ensemble of Convolutional Neural Networks (CNNs) trained on synthetic data is adopted for detecting and recognizing East Asian characters. Finally, a dynamic programming approach leveraging language models is applied to constitute results of the entire body of text lines. The proposed system achieves average end-to-end accuracies of 98.2% and 98.3% on 40 videos in Simplified Chinese and 40 videos in Traditional Chinese respectively, which is a significant outperformance of other existing methods. The near-perfect accuracy of our system dramatically narrows the gap between human cognitive ability and state-of-the-art algorithms used for such a task.
Gland Instance Segmentation by Deep Multichannel Neural Networks
Jul 19, 2016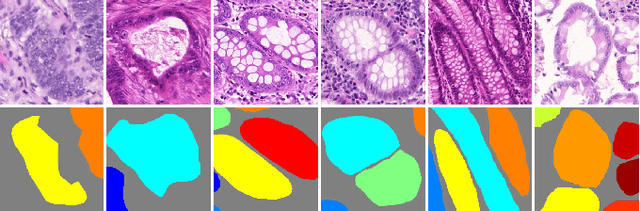
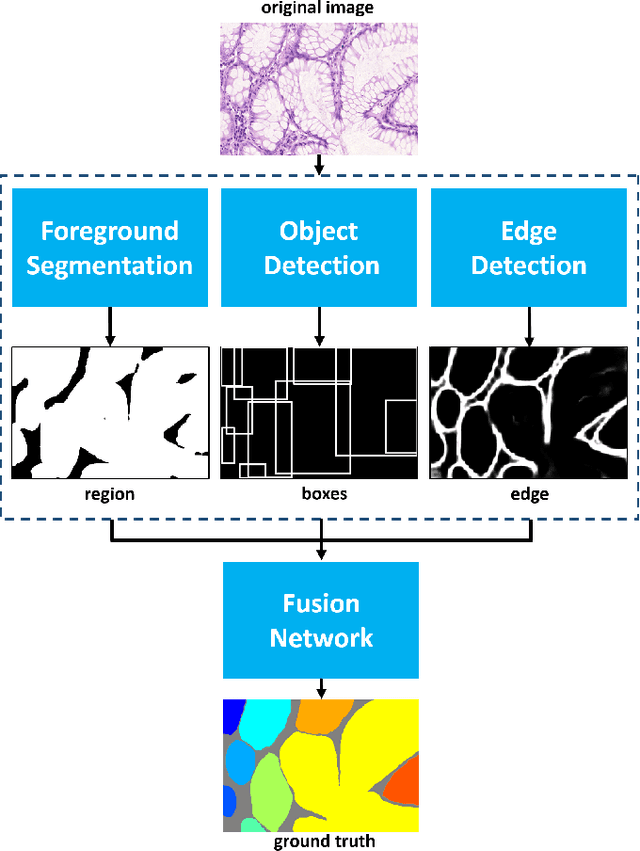
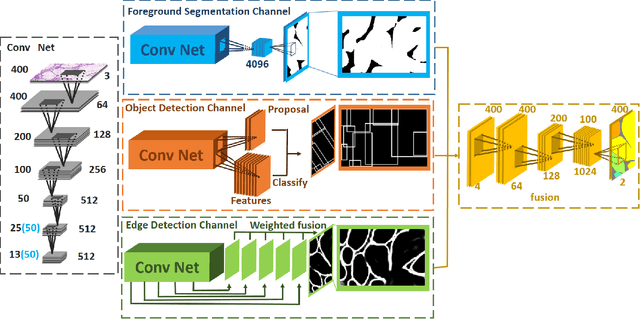

Abstract:In this paper, we propose a new image instance segmentation method that segments individual glands (instances) in colon histology images. This is a task called instance segmentation that has recently become increasingly important. The problem is challenging since not only do the glands need to be segmented from the complex background, they are also required to be individually identified. Here we leverage the idea of image-to-image prediction in recent deep learning by building a framework that automatically exploits and fuses complex multichannel information, regional, location and boundary patterns in gland histology images. Our proposed system, deep multichannel framework, alleviates heavy feature design due to the use of convolutional neural networks and is able to meet multifarious requirement by altering channels. Compared to methods reported in the 2015 MICCAI Gland Segmentation Challenge and other currently prevalent methods of instance segmentation, we observe state-of-the-art results based on a number of evaluation metrics.
Gland Instance Segmentation by Deep Multichannel Side Supervision
Jul 14, 2016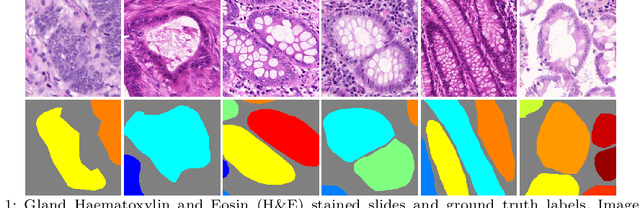
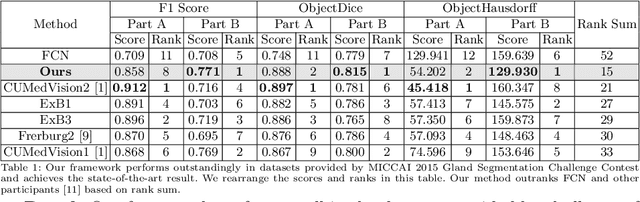
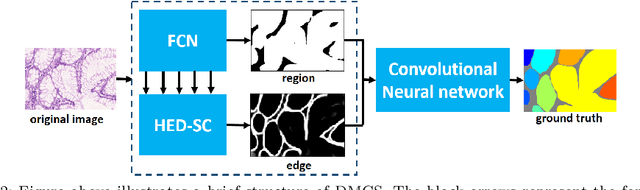
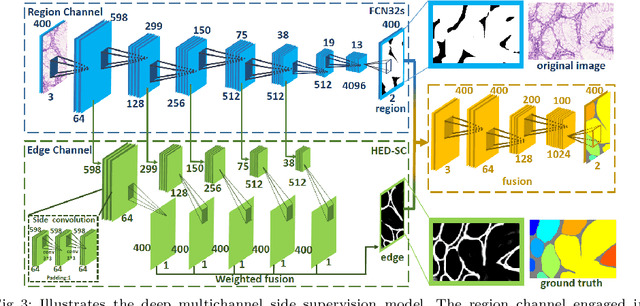
Abstract:In this paper, we propose a new image instance segmentation method that segments individual glands (instances) in colon histology images. This is a task called instance segmentation that has recently become increasingly important. The problem is challenging since not only do the glands need to be segmented from the complex background, they are also required to be individually identified. Here we leverage the idea of image-to-image prediction in recent deep learning by building a framework that automatically exploits and fuses complex multichannel information, regional and boundary patterns, with side supervision (deep supervision on side responses) in gland histology images. Our proposed system, deep multichannel side supervision (DMCS), alleviates heavy feature design due to the use of convolutional neural networks guided by side supervision. Compared to methods reported in the 2015 MICCAI Gland Segmentation Challenge, we observe state-of-the-art results based on a number of evaluation metrics.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge