Zheren Li
Learning Better Contrastive View from Radiologist's Gaze
May 15, 2023



Abstract:Recent self-supervised contrastive learning methods greatly benefit from the Siamese structure that aims to minimizing distances between positive pairs. These methods usually apply random data augmentation to input images, expecting the augmented views of the same images to be similar and positively paired. However, random augmentation may overlook image semantic information and degrade the quality of augmented views in contrastive learning. This issue becomes more challenging in medical images since the abnormalities related to diseases can be tiny, and are easy to be corrupted (e.g., being cropped out) in the current scheme of random augmentation. In this work, we first demonstrate that, for widely-used X-ray images, the conventional augmentation prevalent in contrastive pre-training can affect the performance of the downstream diagnosis or classification tasks. Then, we propose a novel augmentation method, i.e., FocusContrast, to learn from radiologists' gaze in diagnosis and generate contrastive views for medical images with guidance from radiologists' visual attention. Specifically, we track the gaze movement of radiologists and model their visual attention when reading to diagnose X-ray images. The learned model can predict visual attention of the radiologists given a new input image, and further guide the attention-aware augmentation that hardly neglects the disease-related abnormalities. As a plug-and-play and framework-agnostic module, FocusContrast consistently improves state-of-the-art contrastive learning methods of SimCLR, MoCo, and BYOL by 4.0~7.0% in classification accuracy on a knee X-ray dataset.
Domain Generalization for Mammographic Image Analysis via Contrastive Learning
Apr 20, 2023Abstract:Mammographic image analysis is a fundamental problem in the computer-aided diagnosis scheme, which has recently made remarkable progress with the advance of deep learning. However, the construction of a deep learning model requires training data that are large and sufficiently diverse in terms of image style and quality. In particular, the diversity of image style may be majorly attributed to the vendor factor. However, mammogram collection from vendors as many as possible is very expensive and sometimes impractical for laboratory-scale studies. Accordingly, to further augment the generalization capability of deep learning models to various vendors with limited resources, a new contrastive learning scheme is developed. Specifically, the backbone network is firstly trained with a multi-style and multi-view unsupervised self-learning scheme for the embedding of invariant features to various vendor styles. Afterward, the backbone network is then recalibrated to the downstream tasks of mass detection, multi-view mass matching, BI-RADS classification and breast density classification with specific supervised learning. The proposed method is evaluated with mammograms from four vendors and two unseen public datasets. The experimental results suggest that our approach can effectively improve analysis performance on both seen and unseen domains, and outperforms many state-of-the-art (SOTA) generalization methods.
TBI-GAN: An Adversarial Learning Approach for Data Synthesis on Traumatic Brain Segmentation
Aug 12, 2022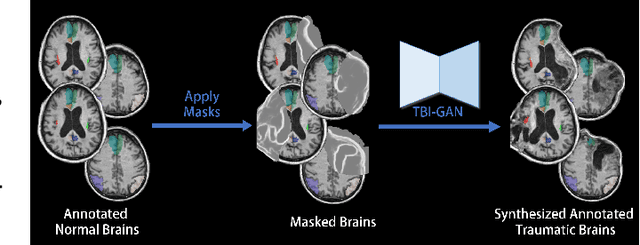
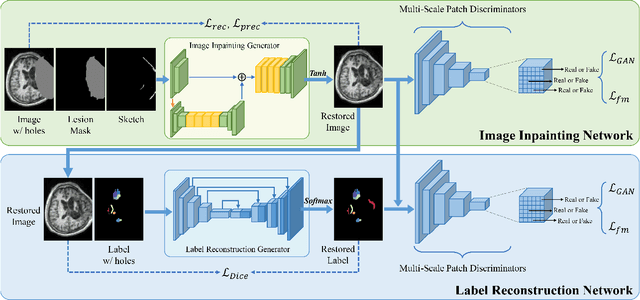
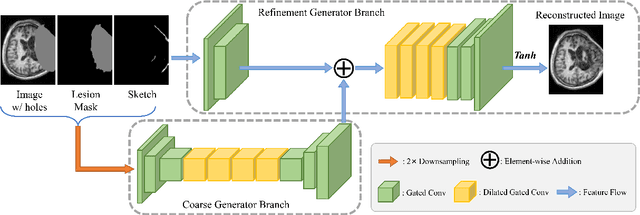
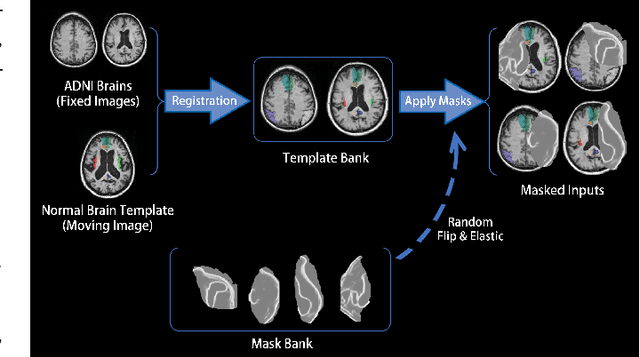
Abstract:Brain network analysis for traumatic brain injury (TBI) patients is critical for its consciousness level assessment and prognosis evaluation, which requires the segmentation of certain consciousness-related brain regions. However, it is difficult to construct a TBI segmentation model as manually annotated MR scans of TBI patients are hard to collect. Data augmentation techniques can be applied to alleviate the issue of data scarcity. However, conventional data augmentation strategies such as spatial and intensity transformation are unable to mimic the deformation and lesions in traumatic brains, which limits the performance of the subsequent segmentation task. To address these issues, we propose a novel medical image inpainting model named TBI-GAN to synthesize TBI MR scans with paired brain label maps. The main strength of our TBI-GAN method is that it can generate TBI images and corresponding label maps simultaneously, which has not been achieved in the previous inpainting methods for medical images. We first generate the inpainted image under the guidance of edge information following a coarse-to-fine manner, and then the synthesized intensity image is used as the prior for label inpainting. Furthermore, we introduce a registration-based template augmentation pipeline to increase the diversity of the synthesized image pairs and enhance the capacity of data augmentation. Experimental results show that the proposed TBI-GAN method can produce sufficient synthesized TBI images with high quality and valid label maps, which can greatly improve the 2D and 3D traumatic brain segmentation performance compared with the alternatives.
Domain Generalization for Mammography Detection via Multi-style and Multi-view Contrastive Learning
Nov 21, 2021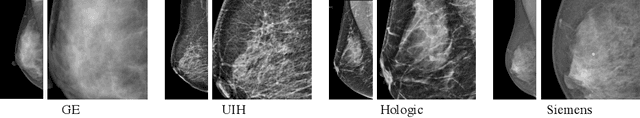
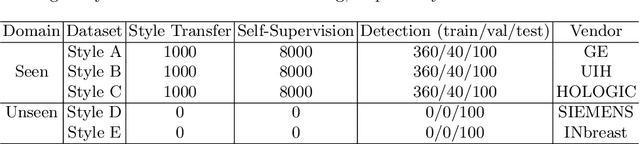
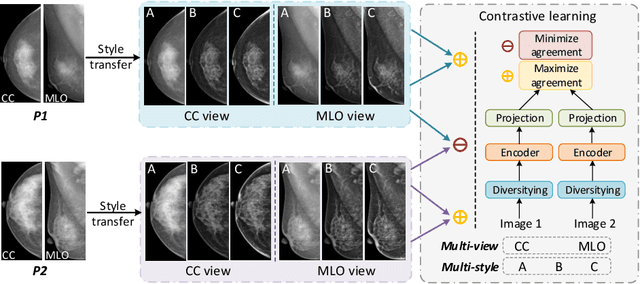
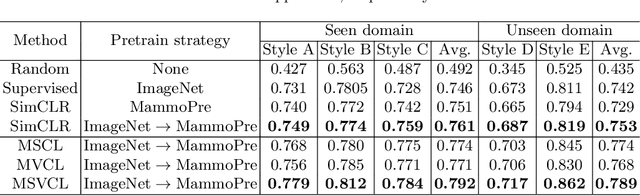
Abstract:Lesion detection is a fundamental problem in the computer-aided diagnosis scheme for mammography. The advance of deep learning techniques have made a remarkable progress for this task, provided that the training data are large and sufficiently diverse in terms of image style and quality. In particular, the diversity of image style may be majorly attributed to the vendor factor. However, the collection of mammograms from vendors as many as possible is very expensive and sometimes impractical for laboratory-scale studies. Accordingly, to further augment the generalization capability of deep learning model to various vendors with limited resources, a new contrastive learning scheme is developed. Specifically, the backbone network is firstly trained with a multi-style and multi-view unsupervised self-learning scheme for the embedding of invariant features to various vendor-styles. Afterward, the backbone network is then recalibrated to the downstream task of lesion detection with the specific supervised learning. The proposed method is evaluated with mammograms from four vendors and one unseen public dataset. The experimental results suggest that our approach can effectively improve detection performance on both seen and unseen domains, and outperforms many state-of-the-art (SOTA) generalization methods.
* Pages 98-108
Automated Segmentation of Lesions in Ultrasound Using Semi-pixel-wise Cycle Generative Adversarial Nets
May 27, 2019
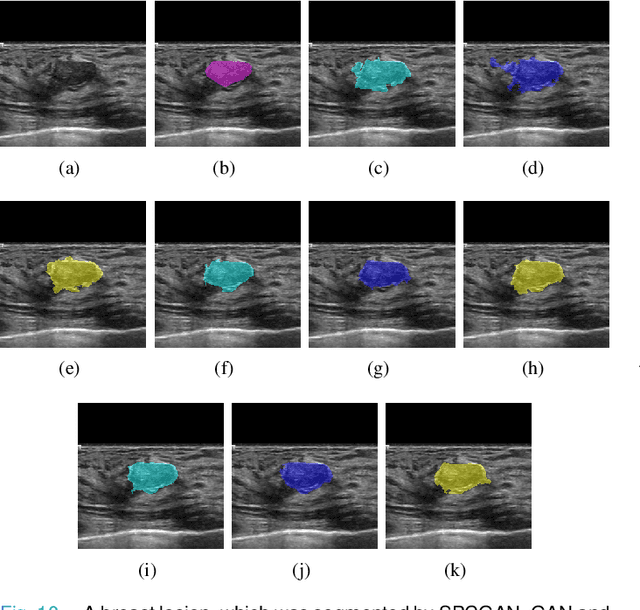
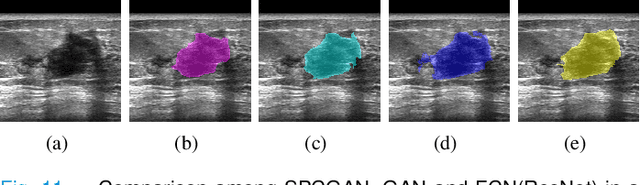
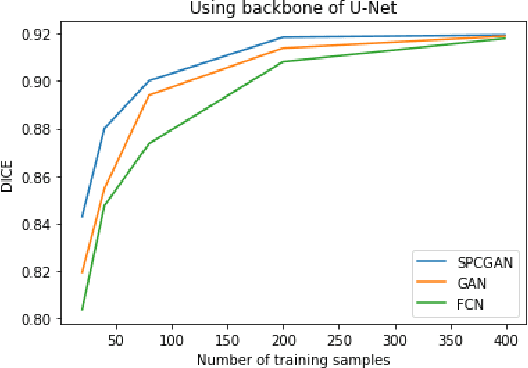
Abstract:Breast cancer is the most common invasive cancer with the highest cancer occurrence in females. Handheld ultrasound is one of the most efficient ways to identify and diagnose the breast cancer. The area and the shape information of a lesion is very helpful for clinicians to make diagnostic decisions. In this study we propose a new deep-learning scheme, semi-pixel-wise cycle generative adversarial net (SPCGAN) for segmenting the lesion in 2D ultrasound. The method takes the advantage of a fully connected convolutional neural network (FCN) and a generative adversarial net to segment a lesion by using prior knowledge. We compared the proposed method to a fully connected neural network and the level set segmentation method on a test dataset consisting of 32 malignant lesions and 109 benign lesions. Our proposed method achieved a Dice similarity coefficient (DSC) of 0.92 while FCN and the level set achieved 0.90 and 0.79 respectively. Particularly, for malignant lesions, our method increases the DSC (0.90) of the fully connected neural network to 0.93 significantly (p$<$0.001). The results show that our SPCGAN can obtain robust segmentation results and may be used to relieve the radiologists' burden for annotation.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge