Tobias Czempiel
Location-Free Scene Graph Generation
Mar 20, 2023



Abstract:Scene Graph Generation (SGG) is a challenging visual understanding task. It combines the detection of entities and relationships between them in a scene. Both previous works and existing evaluation metrics rely on bounding box labels, even though many downstream scene graph applications do not need location information. The need for localization labels significantly increases the annotation cost and hampers the creation of more and larger scene graph datasets. We suggest breaking the dependency of scene graphs on bounding box labels by proposing location-free scene graph generation (LF-SGG). This new task aims at predicting instances of entities, as well as their relationships, without spatial localization. To objectively evaluate the task, the predicted and ground truth scene graphs need to be compared. We solve this NP-hard problem through an efficient algorithm using branching. Additionally, we design the first LF-SGG method, Pix2SG, using autoregressive sequence modeling. Our proposed method is evaluated on Visual Genome and 4D-OR. Although using significantly fewer labels during training, we achieve 74.12\% of the location-supervised SOTA performance on Visual Genome and even outperform the best method on 4D-OR.
CholecTriplet2022: Show me a tool and tell me the triplet -- an endoscopic vision challenge for surgical action triplet detection
Feb 13, 2023



Abstract:Formalizing surgical activities as triplets of the used instruments, actions performed, and target anatomies is becoming a gold standard approach for surgical activity modeling. The benefit is that this formalization helps to obtain a more detailed understanding of tool-tissue interaction which can be used to develop better Artificial Intelligence assistance for image-guided surgery. Earlier efforts and the CholecTriplet challenge introduced in 2021 have put together techniques aimed at recognizing these triplets from surgical footage. Estimating also the spatial locations of the triplets would offer a more precise intraoperative context-aware decision support for computer-assisted intervention. This paper presents the CholecTriplet2022 challenge, which extends surgical action triplet modeling from recognition to detection. It includes weakly-supervised bounding box localization of every visible surgical instrument (or tool), as the key actors, and the modeling of each tool-activity in the form of <instrument, verb, target> triplet. The paper describes a baseline method and 10 new deep learning algorithms presented at the challenge to solve the task. It also provides thorough methodological comparisons of the methods, an in-depth analysis of the obtained results, their significance, and useful insights for future research directions and applications in surgery.
CholecTriplet2021: A benchmark challenge for surgical action triplet recognition
Apr 10, 2022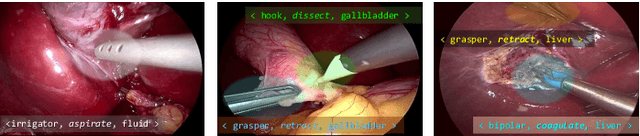
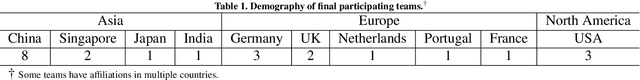
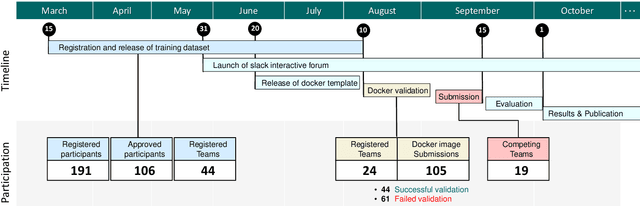
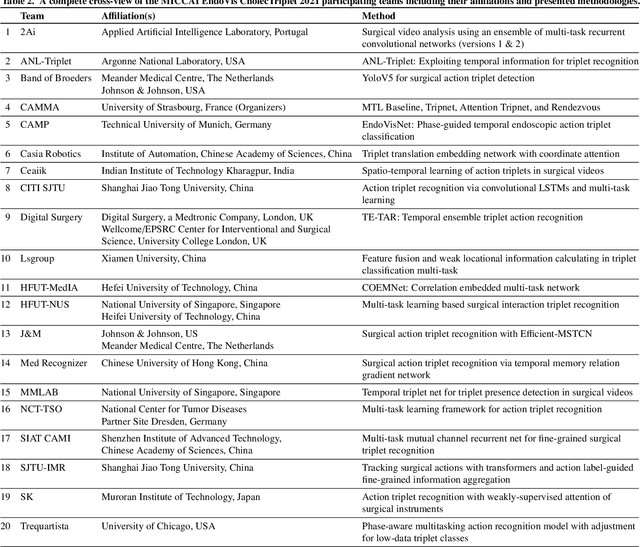
Abstract:Context-aware decision support in the operating room can foster surgical safety and efficiency by leveraging real-time feedback from surgical workflow analysis. Most existing works recognize surgical activities at a coarse-grained level, such as phases, steps or events, leaving out fine-grained interaction details about the surgical activity; yet those are needed for more helpful AI assistance in the operating room. Recognizing surgical actions as triplets of <instrument, verb, target> combination delivers comprehensive details about the activities taking place in surgical videos. This paper presents CholecTriplet2021: an endoscopic vision challenge organized at MICCAI 2021 for the recognition of surgical action triplets in laparoscopic videos. The challenge granted private access to the large-scale CholecT50 dataset, which is annotated with action triplet information. In this paper, we present the challenge setup and assessment of the state-of-the-art deep learning methods proposed by the participants during the challenge. A total of 4 baseline methods from the challenge organizers and 19 new deep learning algorithms by competing teams are presented to recognize surgical action triplets directly from surgical videos, achieving mean average precision (mAP) ranging from 4.2% to 38.1%. This study also analyzes the significance of the results obtained by the presented approaches, performs a thorough methodological comparison between them, in-depth result analysis, and proposes a novel ensemble method for enhanced recognition. Our analysis shows that surgical workflow analysis is not yet solved, and also highlights interesting directions for future research on fine-grained surgical activity recognition which is of utmost importance for the development of AI in surgery.
Few-shot Structured Radiology Report Generation Using Natural Language Prompts
Mar 29, 2022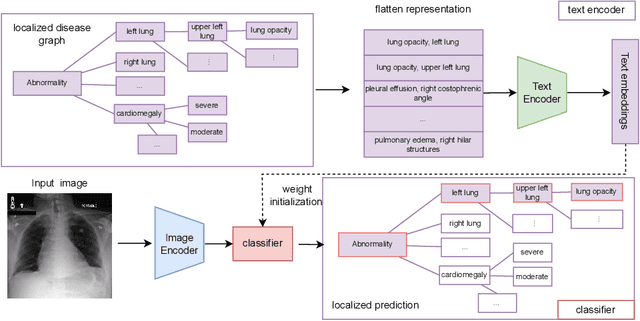
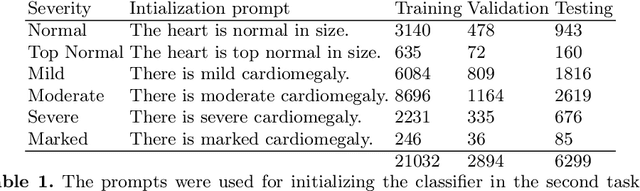
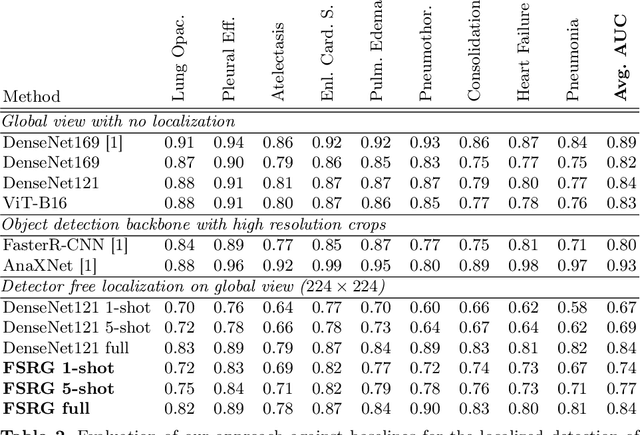
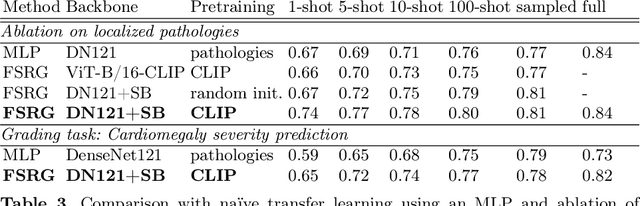
Abstract:Chest radiograph reporting is time-consuming, and numerous solutions to automate this process have been proposed. Due to the complexity of medical information, the variety of writing styles, and free text being prone to typos and inconsistencies, the efficacy of quantifying the clinical accuracy of free-text reports using natural language processing measures is challenging. On the other hand, structured reports ensure consistency and can more easily be used as a quality assurance tool. To accomplish this, we present a strategy for predicting clinical observations and their anatomical location that is easily extensible to other structured findings. First, we train a contrastive language-image model using related chest radiographs and free-text radiological reports. Then, we create textual prompts for each structured finding and optimize a classifier for predicting clinical findings and their associations within the medical image. The results indicate that even when only a few image-level annotations are used for training, the method can localize pathologies in chest radiographs and generate structured reports.
4D-OR: Semantic Scene Graphs for OR Domain Modeling
Mar 22, 2022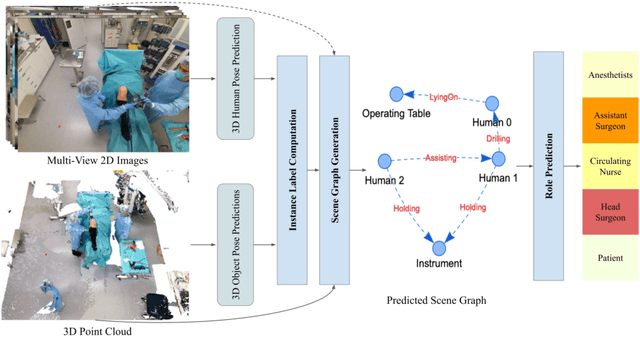


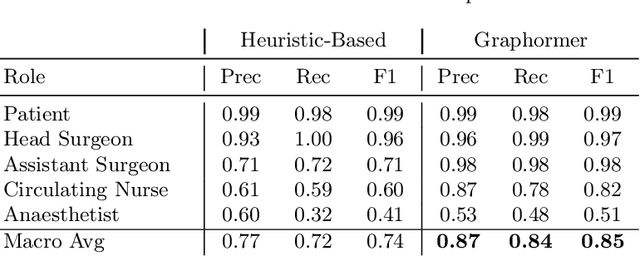
Abstract:Surgical procedures are conducted in highly complex operating rooms (OR), comprising different actors, devices, and interactions. To date, only medically trained human experts are capable of understanding all the links and interactions in such a demanding environment. This paper aims to bring the community one step closer to automated, holistic and semantic understanding and modeling of OR domain. Towards this goal, for the first time, we propose using semantic scene graphs (SSG) to describe and summarize the surgical scene. The nodes of the scene graphs represent different actors and objects in the room, such as medical staff, patients, and medical equipment, whereas edges are the relationships between them. To validate the possibilities of the proposed representation, we create the first publicly available 4D surgical SSG dataset, 4D-OR, containing ten simulated total knee replacement surgeries recorded with six RGB-D sensors in a realistic OR simulation center. 4D-OR includes 6734 frames and is richly annotated with SSGs, human and object poses, and clinical roles. We propose an end-to-end neural network-based SSG generation pipeline, with a rate of success of 0.75 macro F1, indeed being able to infer semantic reasoning in the OR. We further demonstrate the representation power of our scene graphs by using it for the problem of clinical role prediction, where we achieve 0.85 macro F1. The code and dataset will be made available upon acceptance.
Know your sensORs -- A Modality Study For Surgical Action Classification
Mar 22, 2022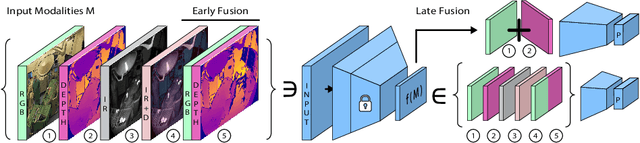
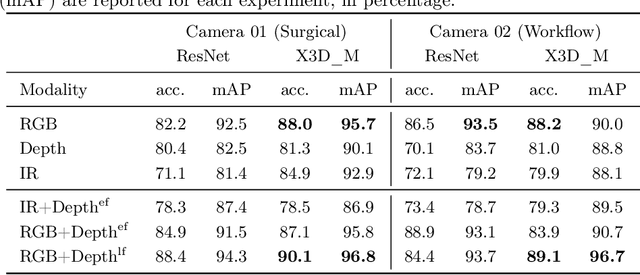
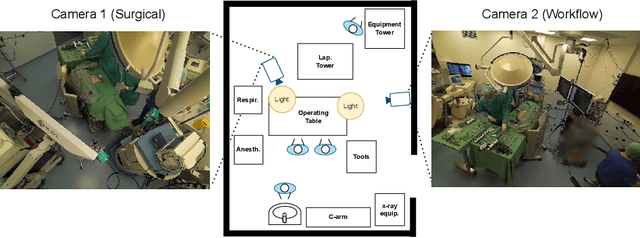
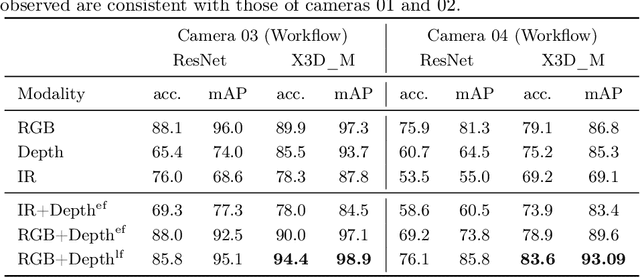
Abstract:The surgical operating room (OR) presents many opportunities for automation and optimization. Videos from various sources in the OR are becoming increasingly available. The medical community seeks to leverage this wealth of data to develop automated methods to advance interventional care, lower costs, and improve overall patient outcomes. Existing datasets from OR room cameras are thus far limited in size or modalities acquired, leaving it unclear which sensor modalities are best suited for tasks such as recognizing surgical action from videos. This study demonstrates that surgical action recognition performance can vary depending on the image modalities used. We perform a methodical analysis on several commonly available sensor modalities, presenting two fusion approaches that improve classification performance. The analyses are carried out on a set of multi-view RGB-D video recordings of 18 laparoscopic procedures.
Longitudinal Self-Supervision for COVID-19 Pathology Quantification
Mar 21, 2022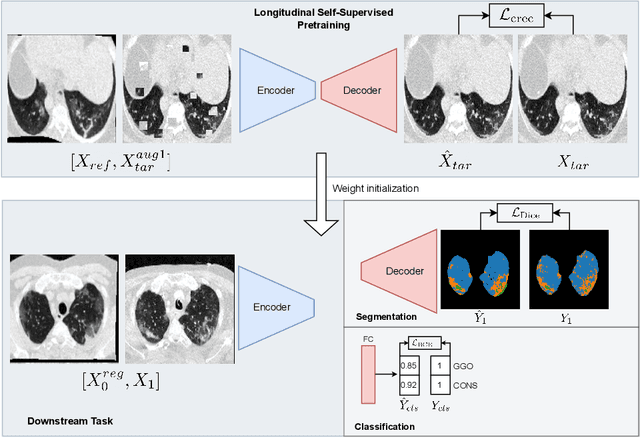
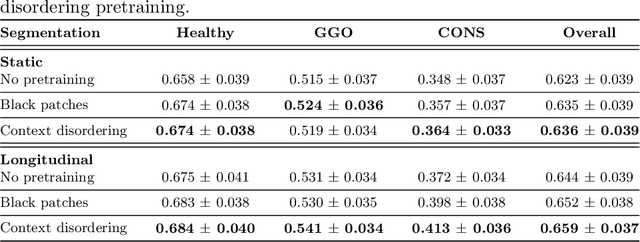
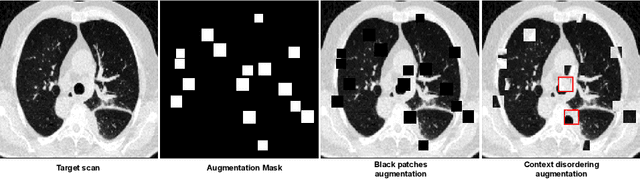

Abstract:Quantifying COVID-19 infection over time is an important task to manage the hospitalization of patients during a global pandemic. Recently, deep learning-based approaches have been proposed to help radiologists automatically quantify COVID-19 pathologies on longitudinal CT scans. However, the learning process of deep learning methods demands extensive training data to learn the complex characteristics of infected regions over longitudinal scans. It is challenging to collect a large-scale dataset, especially for longitudinal training. In this study, we want to address this problem by proposing a new self-supervised learning method to effectively train longitudinal networks for the quantification of COVID-19 infections. For this purpose, longitudinal self-supervision schemes are explored on clinical longitudinal COVID-19 CT scans. Experimental results show that the proposed method is effective, helping the model better exploit the semantics of longitudinal data and improve two COVID-19 quantification tasks.
Surgical Workflow Recognition: from Analysis of Challenges to Architectural Study
Mar 17, 2022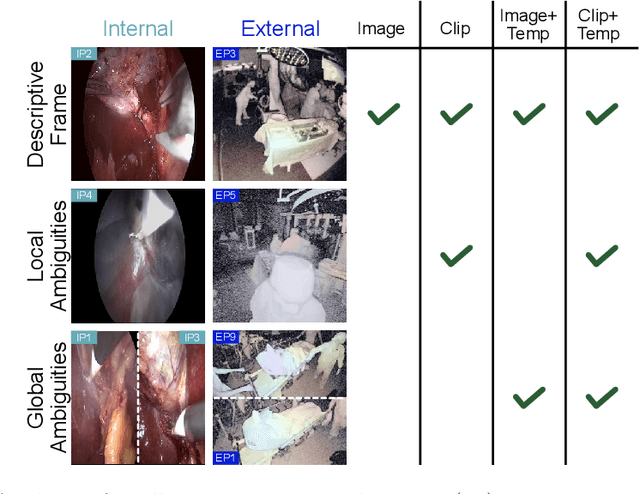
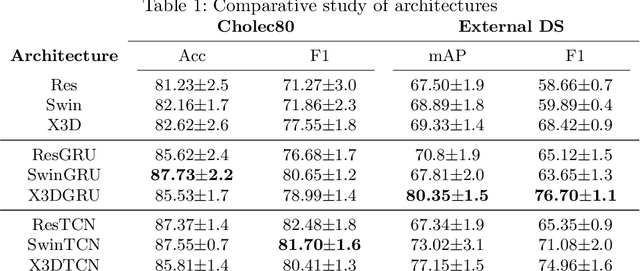
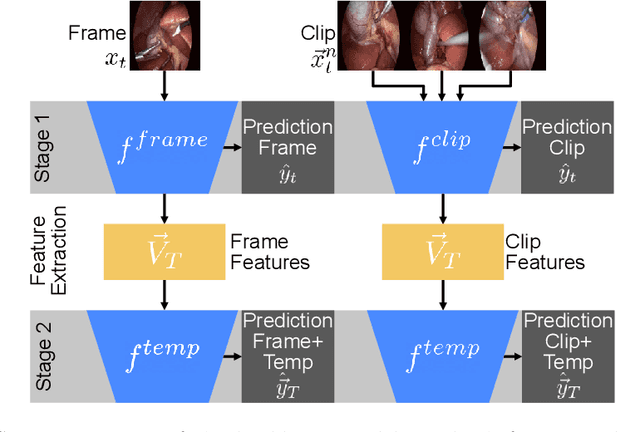
Abstract:Algorithmic surgical workflow recognition is an ongoing research field and can be divided into laparoscopic (Internal) and operating room (External) analysis. So far many different works for the internal analysis have been proposed with the combination of a frame-level and an additional temporal model to address the temporal ambiguities between different workflow phases. For the External recognition task, Clip-level methods are in the focus of researchers targeting the local ambiguities present in the OR scene. In this work we evaluate combinations of different model architectures for the task of surgical workflow recognition to provide a fair comparison of the methods for both Internal and External analysis. We show that methods designed for the Internal analysis can be transferred to the external task with comparable performance gains for different architectures.
U-GAT: Multimodal Graph Attention Network for COVID-19 Outcome Prediction
Jul 29, 2021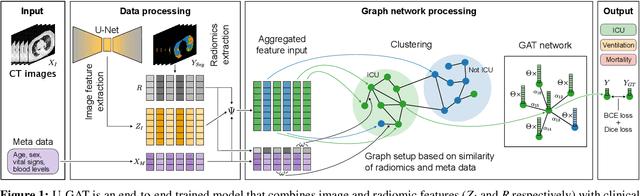
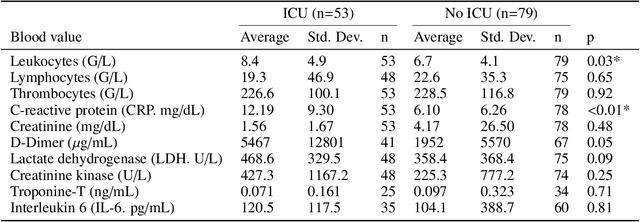
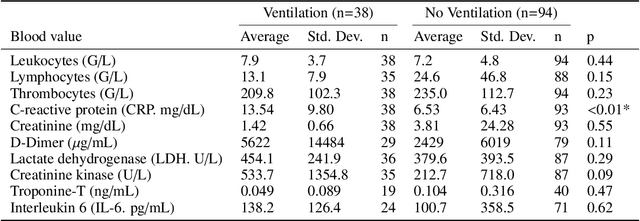
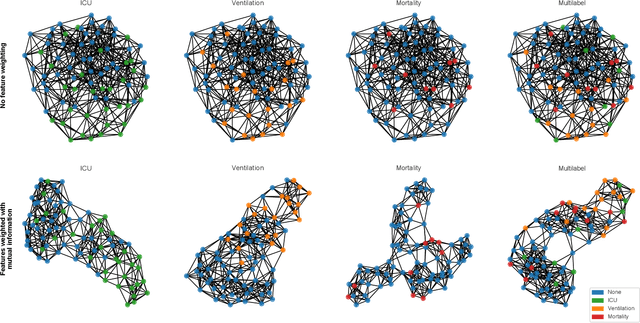
Abstract:During the first wave of COVID-19, hospitals were overwhelmed with the high number of admitted patients. An accurate prediction of the most likely individual disease progression can improve the planning of limited resources and finding the optimal treatment for patients. However, when dealing with a newly emerging disease such as COVID-19, the impact of patient- and disease-specific factors (e.g. body weight or known co-morbidities) on the immediate course of disease is by and large unknown. In the case of COVID-19, the need for intensive care unit (ICU) admission of pneumonia patients is often determined only by acute indicators such as vital signs (e.g. breathing rate, blood oxygen levels), whereas statistical analysis and decision support systems that integrate all of the available data could enable an earlier prognosis. To this end, we propose a holistic graph-based approach combining both imaging and non-imaging information. Specifically, we introduce a multimodal similarity metric to build a population graph for clustering patients and an image-based end-to-end Graph Attention Network to process this graph and predict the COVID-19 patient outcomes: admission to ICU, need for ventilation and mortality. Additionally, the network segments chest CT images as an auxiliary task and extracts image features and radiomics for feature fusion with the available metadata. Results on a dataset collected in Klinikum rechts der Isar in Munich, Germany show that our approach outperforms single modality and non-graph baselines. Moreover, our clustering and graph attention allow for increased understanding of the patient relationships within the population graph and provide insight into the network's decision-making process.
Longitudinal Quantitative Assessment of COVID-19 Infection Progression from Chest CTs
Mar 12, 2021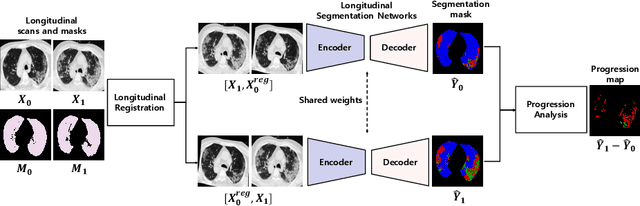
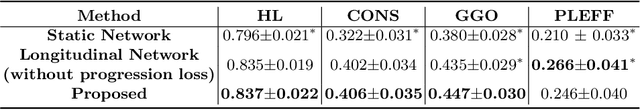

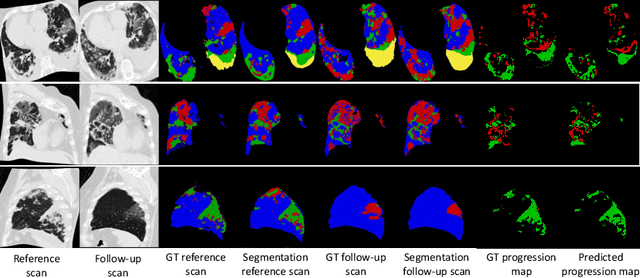
Abstract:Chest computed tomography (CT) has played an essential diagnostic role in assessing patients with COVID-19 by showing disease-specific image features such as ground-glass opacity and consolidation. Image segmentation methods have proven to help quantify the disease burden and even help predict the outcome. The availability of longitudinal CT series may also result in an efficient and effective method to reliably assess the progression of COVID-19, monitor the healing process and the response to different therapeutic strategies. In this paper, we propose a new framework to identify infection at a voxel level (identification of healthy lung, consolidation, and ground-glass opacity) and visualize the progression of COVID-19 using sequential low-dose non-contrast CT scans. In particular, we devise a longitudinal segmentation network that utilizes the reference scan information to improve the performance of disease identification. Experimental results on a clinical longitudinal dataset collected in our institution show the effectiveness of the proposed method compared to the static deep neural networks for disease quantification.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge