Jiyuan Hu
TRACE: High-Fidelity 3D Scene Editing via Tangible Reconstruction and Geometry-Aligned Contextual Video Masking
Apr 01, 2026Abstract:We present TRACE, a mesh-guided 3DGS editing framework that achieves automated, high-fidelity scene transformation. By anchoring video diffusion with explicit 3D geometry, TRACE uniquely enables fine-grained, part-level manipulatio--such as local pose shifting or component replacemen--while preserving the structural integrity of the central subject, a capability largely absent in existing editing methods. Our approach comprises three key stages: (1) Multi-view 3D-Anchor Synthesis, which leverages a sparse-view editor trained on our MV-TRACE datase--the first multi-view consistent dataset dedicated to scene-coherent object addition and modificatio--to generate spatially consistent 3D-anchors; (2) Tangible Geometry Anchoring (TGA), which ensures precise spatial synchronization between inserted meshes and the 3DGS scene via two-phase registration; and (3) Contextual Video Masking (CVM), which integrates 3D projections into an autoregressive video pipeline to achieve temporally stable, physically-grounded rendering. Extensive experiments demonstrate that TRACE consistently outperforms existing methods especially in editing versatility and structural integrity.
Quark Medical Alignment: A Holistic Multi-Dimensional Alignment and Collaborative Optimization Paradigm
Feb 12, 2026Abstract:While reinforcement learning for large language model alignment has progressed rapidly in recent years, transferring these paradigms to high-stakes medical question answering reveals a fundamental paradigm mismatch. Reinforcement Learning from Human Feedback relies on preference annotations that are prohibitively expensive and often fail to reflect the absolute correctness of medical facts. Reinforcement Learning from Verifiable Rewards lacks effective automatic verifiers and struggles to handle complex clinical contexts. Meanwhile, medical alignment requires the simultaneous optimization of correctness, safety, and compliance, yet multi-objective heterogeneous reward signals are prone to scale mismatch and optimization conflicts.To address these challenges, we propose a robust medical alignment paradigm. We first construct a holistic multi-dimensional medical alignment matrix that decomposes alignment objectives into four categories: fundamental capabilities, expert knowledge, online feedback, and format specifications. Within each category, we establish a closed loop of where observable metrics inform attributable diagnosis, which in turn drives optimizable rewards, thereby providing fine-grained, high-resolution supervision signals for subsequent iterative optimization. To resolve gradient domination and optimization instability problem caused by heterogeneous signals, we further propose a unified optimization mechanism. This mechanism employs Reference-Frozen Normalization to align reward scales and implements a Tri-Factor Adaptive Dynamic Weighting strategy to achieve collaborative optimization that is weakness-oriented, risk-prioritized, and redundancy-reducing. Experimental results demonstrate the effectiveness of our proposed paradigm in real-world medical scenario evaluations, establishing a new paradigm for complex alignment in vertical domains.
A Lossless Intra Reference Block Recompression Scheme for Bandwidth Reduction in HEVC-IBC
Apr 05, 2021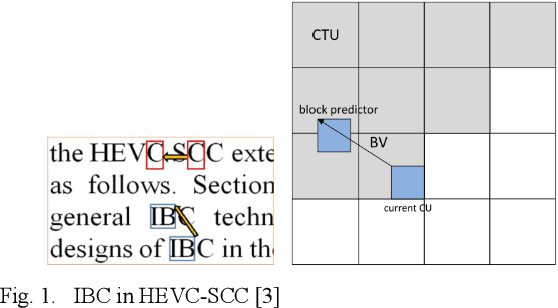
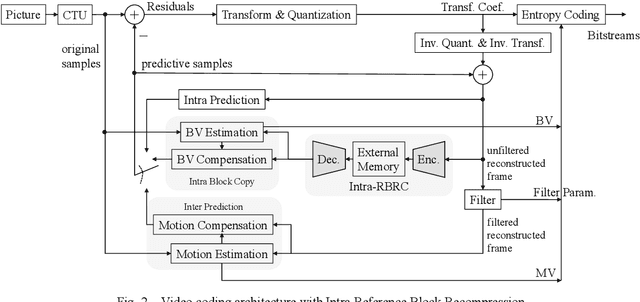
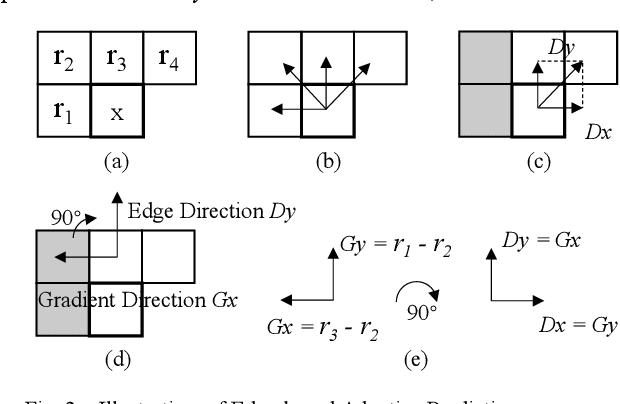
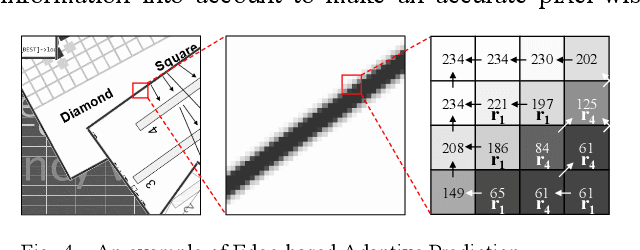
Abstract:The reference frame memory accesses in inter prediction result in high DRAM bandwidth requirement and power consumption. This problem is more intensive by the adoption of intra block copy (IBC), a new coding tool in the screen content coding (SCC) extension to High Efficiency Video Coding (HEVC). In this paper, we propose a lossless recompression scheme that compresses the reference blocks in intra prediction, i.e., intra block copy, before storing them into DRAM to alleviate this problem. The proposal performs pixel-wise texture analysis with an edge-based adaptive prediction method yet no signaling for direction in bitstreams, thus achieves a high gain for compression. Experimental results demonstrate that the proposed scheme shows a 72% data reduction rate on average, which solves the memory bandwidth problem.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge