Xiahai Zhuang
Unsupervised Cardiac Segmentation Utilizing Synthesized Images from Anatomical Labels
Jan 15, 2023



Abstract:Cardiac segmentation is in great demand for clinical practice. Due to the enormous labor of manual delineation, unsupervised segmentation is desired. The ill-posed optimization problem of this task is inherently challenging, requiring well-designed constraints. In this work, we propose an unsupervised framework for multi-class segmentation with both intensity and shape constraints. Firstly, we extend a conventional non-convex energy function as an intensity constraint and implement it with U-Net. For shape constraint, synthetic images are generated from anatomical labels via image-to-image translation, as shape supervision for the segmentation network. Moreover, augmentation invariance is applied to facilitate the segmentation network to learn the latent features in terms of shape. We evaluated the proposed framework using the public datasets from MICCAI2019 MSCMR Challenge and achieved promising results on cardiac MRIs with Dice scores of 0.5737, 0.7796, and 0.6287 in Myo, LV, and RV, respectively.
Multi-Target Landmark Detection with Incomplete Images via Reinforcement Learning and Shape Prior
Jan 13, 2023



Abstract:Medical images are generally acquired with limited field-of-view (FOV), which could lead to incomplete regions of interest (ROI), and thus impose a great challenge on medical image analysis. This is particularly evident for the learning-based multi-target landmark detection, where algorithms could be misleading to learn primarily the variation of background due to the varying FOV, failing the detection of targets. Based on learning a navigation policy, instead of predicting targets directly, reinforcement learning (RL)-based methods have the potential totackle this challenge in an efficient manner. Inspired by this, in this work we propose a multi-agent RL framework for simultaneous multi-target landmark detection. This framework is aimed to learn from incomplete or (and) complete images to form an implicit knowledge of global structure, which is consolidated during the training stage for the detection of targets from either complete or incomplete test images. To further explicitly exploit the global structural information from incomplete images, we propose to embed a shape model into the RL process. With this prior knowledge, the proposed RL model can not only localize dozens of targetssimultaneously, but also work effectively and robustly in the presence of incomplete images. We validated the applicability and efficacy of the proposed method on various multi-target detection tasks with incomplete images from practical clinics, using body dual-energy X-ray absorptiometry (DXA), cardiac MRI and head CT datasets. Results showed that our method could predict whole set of landmarks with incomplete training images up to 80% missing proportion (average distance error 2.29 cm on body DXA), and could detect unseen landmarks in regions with missing image information outside FOV of target images (average distance error 6.84 mm on 3D half-head CT).
ZScribbleSeg: Zen and the Art of Scribble Supervised Medical Image Segmentation
Jan 12, 2023



Abstract:Curating a large scale fully-annotated dataset can be both labour-intensive and expertise-demanding, especially for medical images. To alleviate this problem, we propose to utilize solely scribble annotations for weakly supervised segmentation. Existing solutions mainly leverage selective losses computed solely on annotated areas and generate pseudo gold standard segmentation by propagating labels to adjacent areas. However, these methods could suffer from the inaccurate and sometimes unrealistic pseudo segmentation due to the insufficient supervision and incomplete shape features. Different from previous efforts, we first investigate the principle of ''good scribble annotations'', which leads to efficient scribble forms via supervision maximization and randomness simulation. Furthermore, we introduce regularization terms to encode the spatial relationship and shape prior, where a new formulation is developed to estimate the mixture ratios of label classes. These ratios are critical in identifying the unlabeled pixels for each class and correcting erroneous predictions, thus the accurate estimation lays the foundation for the incorporation of spatial prior. Finally, we integrate the efficient scribble supervision with the prior into a unified framework, denoted as ZScribbleSeg, and apply the method to multiple scenarios. Leveraging only scribble annotations, ZScribbleSeg set new state-of-the-arts on four segmentation tasks using ACDC, MSCMRseg, MyoPS and PPSS datasets.
MyoPS-Net: Myocardial Pathology Segmentation with Flexible Combination of Multi-Sequence CMR Images
Nov 06, 2022



Abstract:Myocardial pathology segmentation (MyoPS) can be a prerequisite for the accurate diagnosis and treatment planning of myocardial infarction. However, achieving this segmentation is challenging, mainly due to the inadequate and indistinct information from an image. In this work, we develop an end-to-end deep neural network, referred to as MyoPS-Net, to flexibly combine five-sequence cardiac magnetic resonance (CMR) images for MyoPS. To extract precise and adequate information, we design an effective yet flexible architecture to extract and fuse cross-modal features. This architecture can tackle different numbers of CMR images and complex combinations of modalities, with output branches targeting specific pathologies. To impose anatomical knowledge on the segmentation results, we first propose a module to regularize myocardium consistency and localize the pathologies, and then introduce an inclusiveness loss to utilize relations between myocardial scars and edema. We evaluated the proposed MyoPS-Net on two datasets, i.e., a private one consisting of 50 paired multi-sequence CMR images and a public one from MICCAI2020 MyoPS Challenge. Experimental results showed that MyoPS-Net could achieve state-of-the-art performance in various scenarios. Note that in practical clinics, the subjects may not have full sequences, such as missing LGE CMR or mapping CMR scans. We therefore conducted extensive experiments to investigate the performance of the proposed method in dealing with such complex combinations of different CMR sequences. Results proved the superiority and generalizability of MyoPS-Net, and more importantly, indicated a practical clinical application.
$\mathcal{X}$-Metric: An N-Dimensional Information-Theoretic Framework for Groupwise Registration and Deep Combined Computing
Nov 03, 2022



Abstract:This paper presents a generic probabilistic framework for estimating the statistical dependency and finding the anatomical correspondences among an arbitrary number of medical images. The method builds on a novel formulation of the $N$-dimensional joint intensity distribution by representing the common anatomy as latent variables and estimating the appearance model with nonparametric estimators. Through connection to maximum likelihood and the expectation-maximization algorithm, an information\hyp{}theoretic metric called $\mathcal{X}$-metric and a co-registration algorithm named $\mathcal{X}$-CoReg are induced, allowing groupwise registration of the $N$ observed images with computational complexity of $\mathcal{O}(N)$. Moreover, the method naturally extends for a weakly-supervised scenario where anatomical labels of certain images are provided. This leads to a combined\hyp{}computing framework implemented with deep learning, which performs registration and segmentation simultaneously and collaboratively in an end-to-end fashion. Extensive experiments were conducted to demonstrate the versatility and applicability of our model, including multimodal groupwise registration, motion correction for dynamic contrast enhanced magnetic resonance images, and deep combined computing for multimodal medical images. Results show the superiority of our method in various applications in terms of both accuracy and efficiency, highlighting the advantage of the proposed representation of the imaging process.
Multi-Modality Cardiac Image Computing: A Survey
Aug 26, 2022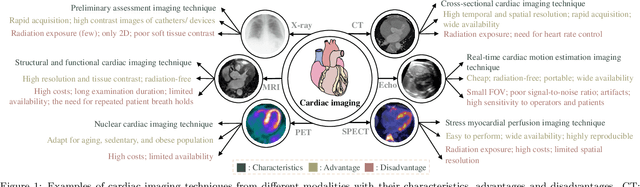

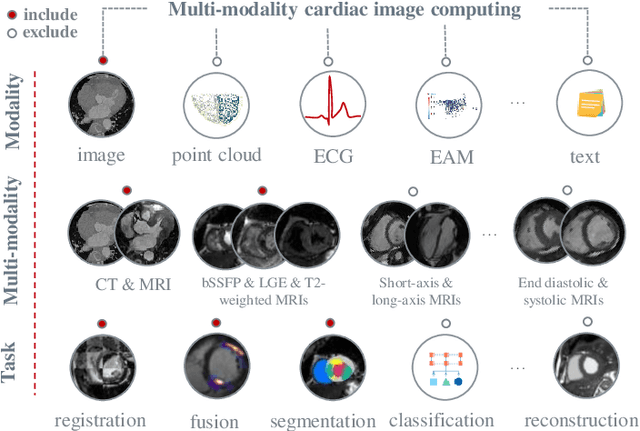

Abstract:Multi-modality cardiac imaging plays a key role in the management of patients with cardiovascular diseases. It allows a combination of complementary anatomical, morphological and functional information, increases diagnosis accuracy, and improves the efficacy of cardiovascular interventions and clinical outcomes. Fully-automated processing and quantitative analysis of multi-modality cardiac images could have a direct impact on clinical research and evidence-based patient management. However, these require overcoming significant challenges including inter-modality misalignment and finding optimal methods to integrate information from different modalities. This paper aims to provide a comprehensive review of multi-modality imaging in cardiology, the computing methods, the validation strategies, the related clinical workflows and future perspectives. For the computing methodologies, we have a favored focus on the three tasks, i.e., registration, fusion and segmentation, which generally involve multi-modality imaging data, \textit{either combining information from different modalities or transferring information across modalities}. The review highlights that multi-modality cardiac imaging data has the potential of wide applicability in the clinic, such as trans-aortic valve implantation guidance, myocardial viability assessment, and catheter ablation therapy and its patient selection. Nevertheless, many challenges remain unsolved, such as missing modality, combination of imaging and non-imaging data, and uniform analysis and representation of different modalities. There is also work to do in defining how the well-developed techniques fit in clinical workflows and how much additional and relevant information they introduce. These problems are likely to continue to be an active field of research and the questions to be answered in the future.
Deep Compatible Learning for Partially-Supervised Medical Image Segmentation
Jun 18, 2022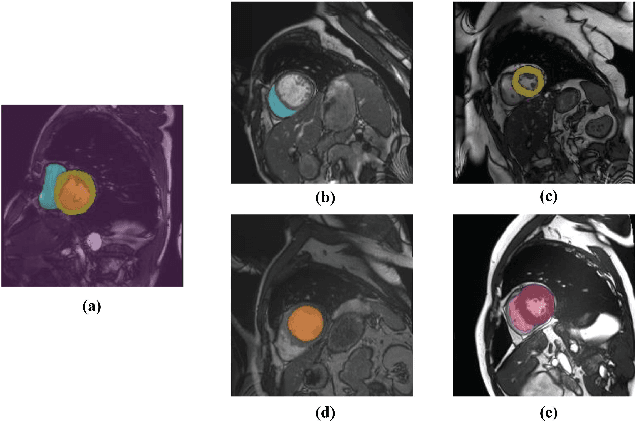
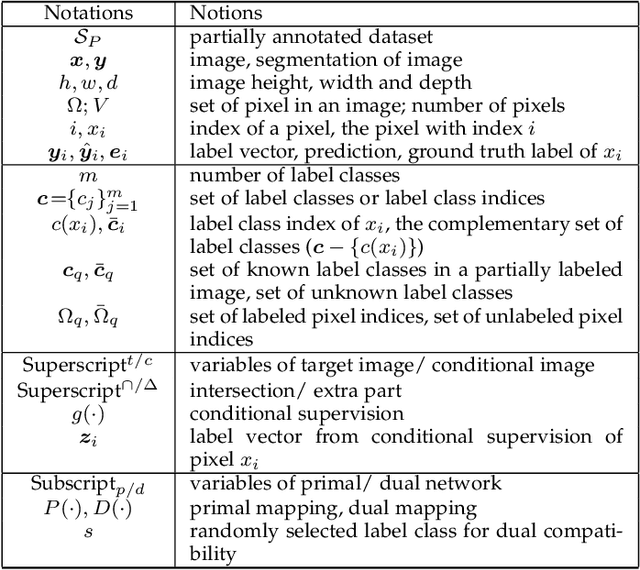

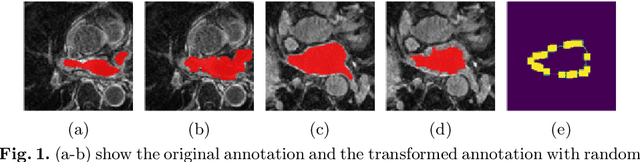
Abstract:Partially-supervised learning can be challenging for segmentation due to the lack of supervision for unlabeled structures, and the methods directly applying fully-supervised learning could lead to incompatibility, meaning ground truth is not in the solution set of the optimization problem given the loss function. To address the challenge, we propose a deep compatible learning (DCL) framework, which trains a single multi-label segmentation network using images with only partial structures annotated. We first formulate the partially-supervised segmentation as an optimization problem compatible with missing labels, and prove its compatibility. Then, we equip the model with a conditional segmentation strategy, to propagate labels from multiple partially-annotated images to the target. Additionally, we propose a dual learning strategy, which learns two opposite mappings of label propagation simultaneously, to provide substantial supervision for unlabeled structures. The two strategies are formulated into compatible forms, termed as conditional compatibility and dual compatibility, respectively. We show this framework is generally applicable for conventional loss functions. The approach attains significant performance improvement over existing methods, especially in the situation where only a small training dataset is available. Results on three segmentation tasks have shown that the proposed framework could achieve performance matching fully-supervised models.
Decoupling Predictions in Distributed Learning for Multi-Center Left Atrial MRI Segmentation
Jun 10, 2022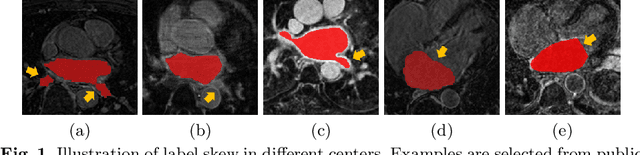
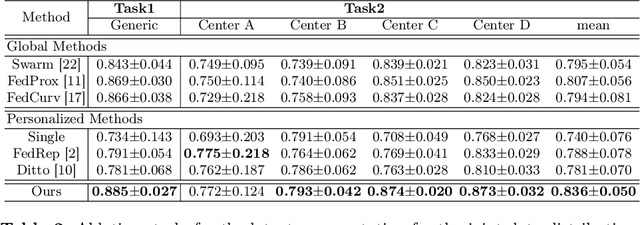
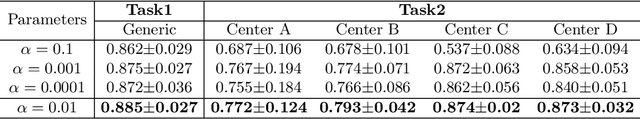
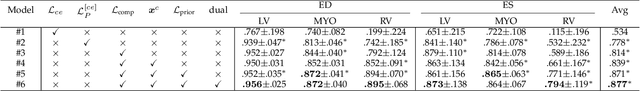
Abstract:Distributed learning has shown great potential in medical image analysis. It allows to use multi-center training data with privacy protection. However, data distributions in local centers can vary from each other due to different imaging vendors, and annotation protocols. Such variation degrades the performance of learning-based methods. To mitigate the influence, two groups of methods have been proposed for different aims, i.e., the global methods and the personalized methods. The former are aimed to improve the performance of a single global model for all test data from unseen centers (known as generic data); while the latter target multiple models for each center (denoted as local data). However, little has been researched to achieve both goals simultaneously. In this work, we propose a new framework of distributed learning that bridges the gap between two groups, and improves the performance for both generic and local data. Specifically, our method decouples the predictions for generic data and local data, via distribution-conditioned adaptation matrices. Results on multi-center left atrial (LA) MRI segmentation showed that our method demonstrated superior performance over existing methods on both generic and local data. Our code is available at https://github.com/key1589745/decouple_predict
Joint Modeling of Image and Label Statistics for Enhancing Model Generalizability of Medical Image Segmentation
Jun 09, 2022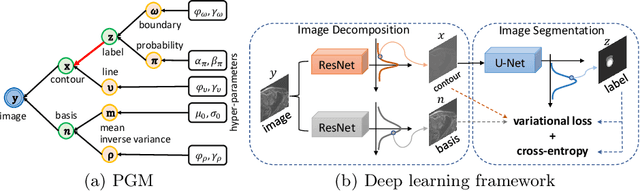
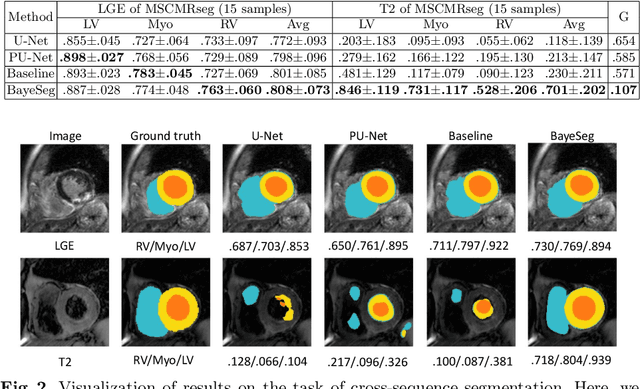
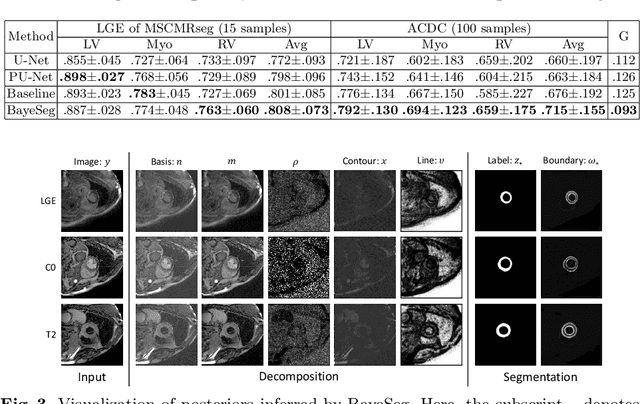
Abstract:Although supervised deep-learning has achieved promising performance in medical image segmentation, many methods cannot generalize well on unseen data, limiting their real-world applicability. To address this problem, we propose a deep learning-based Bayesian framework, which jointly models image and label statistics, utilizing the domain-irrelevant contour of a medical image for segmentation. Specifically, we first decompose an image into components of contour and basis. Then, we model the expected label as a variable only related to the contour. Finally, we develop a variational Bayesian framework to infer the posterior distributions of these variables, including the contour, the basis, and the label. The framework is implemented with neural networks, thus is referred to as deep Bayesian segmentation. Results on the task of cross-sequence cardiac MRI segmentation show that our method set a new state of the art for model generalizability. Particularly, the BayeSeg model trained with LGE MRI generalized well on T2 images and outperformed other models with great margins, i.e., over 0.47 in terms of average Dice. Our code is available at https://zmiclab.github.io/projects.html.
Bayesian intrinsic groupwise registration via explicit hierarchical disentanglement
Jun 06, 2022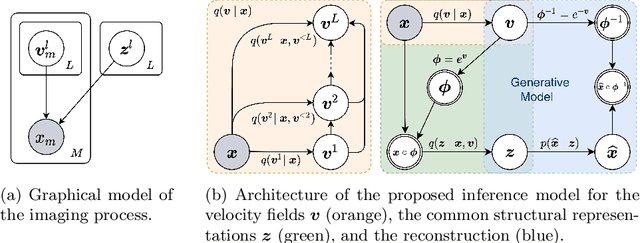
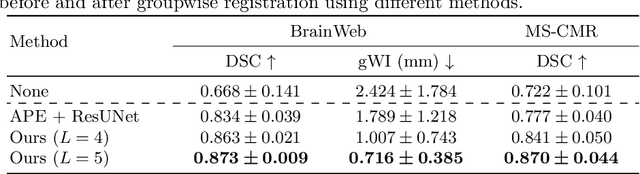
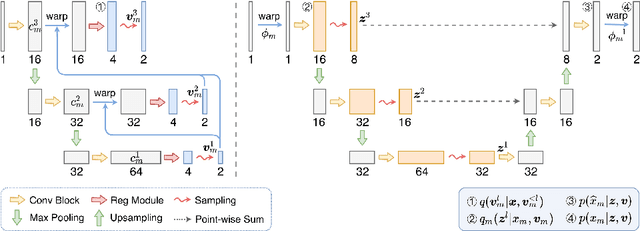
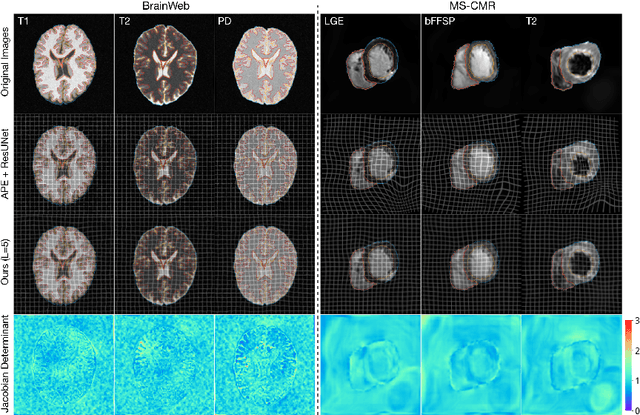
Abstract:Previous methods on multimodal groupwise registration typically require certain highly specialized similarity metrics with restrained applicability. In this work, we instead propose a general framework which formulates groupwise registration as a procedure of hierarchical Bayesian inference. Here, the imaging process of multimodal medical images, including shape transition and appearance variation, is characterized by a disentangled variational auto-encoder. To this end, we propose a novel variational posterior and network architecture that facilitate joint learning of the common structural representation and the desired spatial correspondences. The performance of the proposed model was validated on two publicly available multimodal datasets, i.e., BrainWeb and MS-CMR of the heart. Results have demonstrated the efficacy of our framework in realizing multimodal groupwise registration in an end-to-end fashion.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge