Yuzhe Wang
Speaker Verification with Speech-Aware LLMs: Evaluation and Augmentation
Mar 11, 2026Abstract:Speech-aware large language models (LLMs) can accept speech inputs, yet their training objectives largely emphasize linguistic content or specific fields such as emotions or the speaker's gender, leaving it unclear whether they encode speaker identity. First, we propose a model-agnostic scoring protocol that produces continuous verification scores for both API-only and open-weight models, using confidence scores or log-likelihood ratios from the Yes/No token probabilities. Using this protocol, we benchmark recent speech-aware LLMs and observe weak speaker discrimination (EERs above 20% on VoxCeleb1). Second, we introduce a lightweight augmentation that equips an LLM with ASV capability by injecting frozen ECAPA-TDNN speaker embeddings through a learned projection and training only LoRA adapters. On TinyLLaMA-1.1B, the resulting ECAPA-LLM achieves 1.03% EER on VoxCeleb1-E, approaching a dedicated speaker verification system while preserving a natural-language interface.
CausalFlip: A Benchmark for LLM Causal Judgment Beyond Semantic Matching
Feb 23, 2026Abstract:As large language models (LLMs) witness increasing deployment in complex, high-stakes decision-making scenarios, it becomes imperative to ground their reasoning in causality rather than spurious correlations. However, strong performance on traditional reasoning benchmarks does not guarantee true causal reasoning ability of LLMs, as high accuracy may still arise from memorizing semantic patterns instead of analyzing the underlying true causal structures. To bridge this critical gap, we propose a new causal reasoning benchmark, CausalFlip, designed to encourage the development of new LLM paradigm or training algorithms that ground LLM reasoning in causality rather than semantic correlation. CausalFlip consists of causal judgment questions built over event triples that could form different confounder, chain, and collider relations. Based on this, for each event triple, we construct pairs of semantically similar questions that reuse the same events but yield opposite causal answers, where models that rely heavily on semantic matching are systematically driven toward incorrect predictions. To further probe models' reliance on semantic patterns, we introduce a noisy-prefix evaluation that prepends causally irrelevant text before intermediate causal reasoning steps without altering the underlying causal relations or the logic of the reasoning process. We evaluate LLMs under multiple training paradigms, including answer-only training, explicit Chain-of-Thought (CoT) supervision, and a proposed internalized causal reasoning approach that aims to mitigate explicit reliance on correlation in the reasoning process. Our results show that explicit CoT can still be misled by spurious semantic correlations, where internalizing reasoning steps yields substantially improved causal grounding, suggesting that it is promising to better elicit the latent causal reasoning capabilities of base LLMs.
Detecting Neurodegenerative Diseases using Frame-Level Handwriting Embeddings
Feb 10, 2025Abstract:In this study, we explored the use of spectrograms to represent handwriting signals for assessing neurodegenerative diseases, including 42 healthy controls (CTL), 35 subjects with Parkinson's Disease (PD), 21 with Alzheimer's Disease (AD), and 15 with Parkinson's Disease Mimics (PDM). We applied CNN and CNN-BLSTM models for binary classification using both multi-channel fixed-size and frame-based spectrograms. Our results showed that handwriting tasks and spectrogram channel combinations significantly impacted classification performance. The highest F1-score (89.8%) was achieved for AD vs. CTL, while PD vs. CTL reached 74.5%, and PD vs. PDM scored 77.97%. CNN consistently outperformed CNN-BLSTM. Different sliding window lengths were tested for constructing frame-based spectrograms. A 1-second window worked best for AD, longer windows improved PD classification, and window length had little effect on PD vs. PDM.
AnomalyNCD: Towards Novel Anomaly Class Discovery in Industrial Scenarios
Oct 18, 2024



Abstract:In the industrial scenario, anomaly detection could locate but cannot classify anomalies. To complete their capability, we study to automatically discover and recognize visual classes of industrial anomalies. In terms of multi-class anomaly classification, previous methods cluster anomalies represented by frozen pre-trained models but often fail due to poor discrimination. Novel class discovery (NCD) has the potential to tackle this. However, it struggles with non-prominent and semantically weak anomalies that challenge network learning focus. To address these, we introduce AnomalyNCD, a multi-class anomaly classification framework compatible with existing anomaly detection methods. This framework learns anomaly-specific features and classifies anomalies in a self-supervised manner. Initially, a technique called Main Element Binarization (MEBin) is first designed, which segments primary anomaly regions into masks to alleviate the impact of incorrect detections on learning. Subsequently, we employ mask-guided contrastive representation learning to improve feature discrimination, which focuses network attention on isolated anomalous regions and reduces the confusion of erroneous inputs through re-corrected pseudo labels. Finally, to enable flexible classification at both region and image levels during inference, we develop a region merging strategy that determines the overall image category based on the classified anomaly regions. Our method outperforms the state-of-the-art works on the MVTec AD and MTD datasets. Compared with the current methods, AnomalyNCD combined with zero-shot anomaly detection method achieves a 10.8% $F_1$ gain, 8.8% NMI gain, and 9.5% ARI gain on MVTec AD, 12.8% $F_1$ gain, 5.7% NMI gain, and 10.8% ARI gain on MTD. The source code is available at https://github.com/HUST-SLOW/AnomalyNCD.
A pruning method based on the dissimilarity of angle among channels and filters
Oct 29, 2022



Abstract:Convolutional Neural Network (CNN) is more and more widely used in various fileds, and its computation and memory-demand are also increasing significantly. In order to make it applicable to limited conditions such as embedded application, network compression comes out. Among them, researchers pay more attention to network pruning. In this paper, we encode the convolution network to obtain the similarity of different encoding nodes, and evaluate the connectivity-power among convolutional kernels on the basis of the similarity. Then impose different level of penalty according to different connectivity-power. Meanwhile, we propose Channel Pruning base on the Dissimilarity of Angle (DACP). Firstly, we train a sparse model by GL penalty, and impose an angle dissimilarity constraint on the channels and filters of convolutional network to obtain a more sparse structure. Eventually, the effectiveness of our method is demonstrated in the section of experiment. On CIFAR-10, we reduce 66.86% FLOPs on VGG-16 with 93.31% accuracy after pruning, where FLOPs represents the number of floating-point operations per second of the model. Moreover, on ResNet-32, we reduce FLOPs by 58.46%, which makes the accuracy after pruning reach 91.76%.
MechRetro is a chemical-mechanism-driven graph learning framework for interpretable retrosynthesis prediction and pathway planning
Oct 06, 2022
Abstract:Leveraging artificial intelligence for automatic retrosynthesis speeds up organic pathway planning in digital laboratories. However, existing deep learning approaches are unexplainable, like "black box" with few insights, notably limiting their applications in real retrosynthesis scenarios. Here, we propose MechRetro, a chemical-mechanism-driven graph learning framework for interpretable retrosynthetic prediction and pathway planning, which learns several retrosynthetic actions to simulate a reverse reaction via elaborate self-adaptive joint learning. By integrating chemical knowledge as prior information, we design a novel Graph Transformer architecture to adaptively learn discriminative and chemically meaningful molecule representations, highlighting the strong capacity in molecule feature representation learning. We demonstrate that MechRetro outperforms the state-of-the-art approaches for retrosynthetic prediction with a large margin on large-scale benchmark datasets. Extending MechRetro to the multi-step retrosynthesis analysis, we identify efficient synthetic routes via an interpretable reasoning mechanism, leading to a better understanding in the realm of knowledgeable synthetic chemists. We also showcase that MechRetro discovers a novel pathway for protokylol, along with energy scores for uncertainty assessment, broadening the applicability for practical scenarios. Overall, we expect MechRetro to provide meaningful insights for high-throughput automated organic synthesis in drug discovery.
Multi-Phase Cross-modal Learning for Noninvasive Gene Mutation Prediction in Hepatocellular Carcinoma
May 08, 2020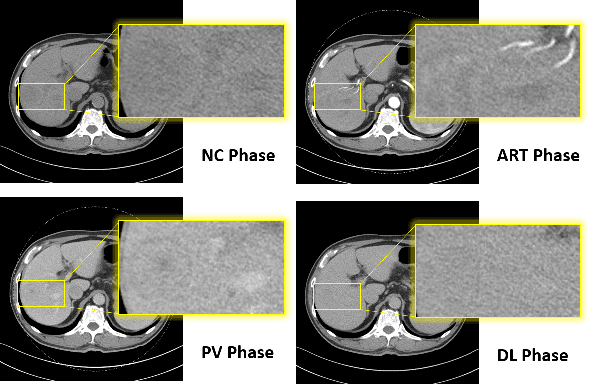
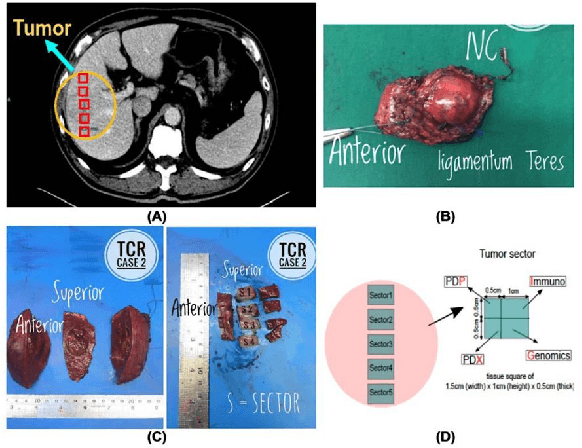
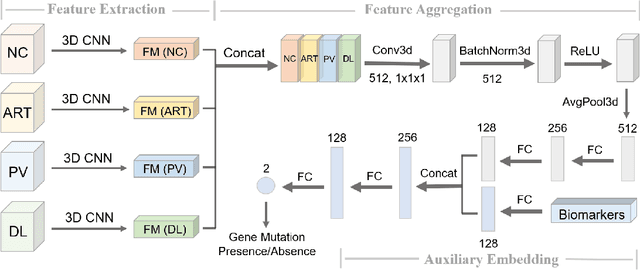
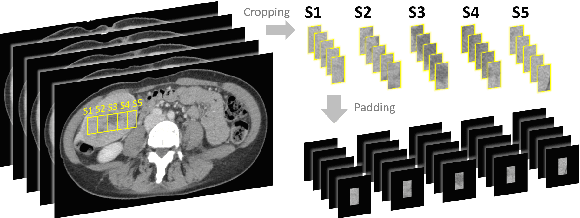
Abstract:Hepatocellular carcinoma (HCC) is the most common type of primary liver cancer and the fourth most common cause of cancer-related death worldwide. Understanding the underlying gene mutations in HCC provides great prognostic value for treatment planning and targeted therapy. Radiogenomics has revealed an association between non-invasive imaging features and molecular genomics. However, imaging feature identification is laborious and error-prone. In this paper, we propose an end-to-end deep learning framework for mutation prediction in APOB, COL11A1 and ATRX genes using multiphasic CT scans. Considering intra-tumour heterogeneity (ITH) in HCC, multi-region sampling technology is implemented to generate the dataset for experiments. Experimental results demonstrate the effectiveness of the proposed model.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge