Rickmer Braren
Department of Diagnostic and Interventional Radiology, School of Medicine, Klinikum rechts der Isar, Technical University of Munich, Germany
3D Arterial Segmentation via Single 2D Projections and Depth Supervision in Contrast-Enhanced CT Images
Sep 15, 2023Abstract:Automated segmentation of the blood vessels in 3D volumes is an essential step for the quantitative diagnosis and treatment of many vascular diseases. 3D vessel segmentation is being actively investigated in existing works, mostly in deep learning approaches. However, training 3D deep networks requires large amounts of manual 3D annotations from experts, which are laborious to obtain. This is especially the case for 3D vessel segmentation, as vessels are sparse yet spread out over many slices and disconnected when visualized in 2D slices. In this work, we propose a novel method to segment the 3D peripancreatic arteries solely from one annotated 2D projection per training image with depth supervision. We perform extensive experiments on the segmentation of peripancreatic arteries on 3D contrast-enhanced CT images and demonstrate how well we capture the rich depth information from 2D projections. We demonstrate that by annotating a single, randomly chosen projection for each training sample, we obtain comparable performance to annotating multiple 2D projections, thereby reducing the annotation effort. Furthermore, by mapping the 2D labels to the 3D space using depth information and incorporating this into training, we almost close the performance gap between 3D supervision and 2D supervision. Our code is available at: https://github.com/alinafdima/3Dseg-mip-depth.
Constructing Population-Specific Atlases from Whole Body MRI: Application to the UKBB
Aug 28, 2023Abstract:Population atlases are commonly utilised in medical imaging to facilitate the investigation of variability across populations. Such atlases enable the mapping of medical images into a common coordinate system, promoting comparability and enabling the study of inter-subject differences. Constructing such atlases becomes particularly challenging when working with highly heterogeneous datasets, such as whole-body images, where subjects show significant anatomical variations. In this work, we propose a pipeline for generating a standardised whole-body atlas for a highly heterogeneous population by partitioning the population into meaningful subgroups. We create six whole-body atlases that represent a healthy population average using magnetic resonance (MR) images from the UK Biobank dataset. We furthermore unbias them, and this way obtain a realistic representation of the population. In addition to the anatomical atlases, we generate probabilistic atlases that capture the distributions of abdominal fat and five abdominal organs across the population. We demonstrate different applications of these atlases, using the differences between subjects with medical conditions such as diabetes and cardiovascular diseases and healthy subjects from the atlas space. With this work, we make the constructed anatomical and label atlases publically available and anticipate them to support medical research conducted on whole-body MR images.
Atlas-Based Interpretable Age Prediction
Jul 14, 2023



Abstract:Age prediction is an important part of medical assessments and research. It can aid in detecting diseases as well as abnormal ageing by highlighting the discrepancy between chronological and biological age. To gain a comprehensive understanding of age-related changes observed in various body parts, we investigate them on a larger scale by using whole-body images. We utilise the Grad-CAM interpretability method to determine the body areas most predictive of a person's age. We expand our analysis beyond individual subjects by employing registration techniques to generate population-wide interpretability maps. Furthermore, we set state-of-the-art whole-body age prediction with a model that achieves a mean absolute error of 2.76 years. Our findings reveal three primary areas of interest: the spine, the autochthonous back muscles, and the cardiac region, which exhibits the highest importance.
Body Fat Estimation from Surface Meshes using Graph Neural Networks
Jul 13, 2023



Abstract:Body fat volume and distribution can be a strong indication for a person's overall health and the risk for developing diseases like type 2 diabetes and cardiovascular diseases. Frequently used measures for fat estimation are the body mass index (BMI), waist circumference, or the waist-hip-ratio. However, those are rather imprecise measures that do not allow for a discrimination between different types of fat or between fat and muscle tissue. The estimation of visceral (VAT) and abdominal subcutaneous (ASAT) adipose tissue volume has shown to be a more accurate measure for named risk factors. In this work, we show that triangulated body surface meshes can be used to accurately predict VAT and ASAT volumes using graph neural networks. Our methods achieve high performance while reducing training time and required resources compared to state-of-the-art convolutional neural networks in this area. We furthermore envision this method to be applicable to cheaper and easily accessible medical surface scans instead of expensive medical images.
Explainable 2D Vision Models for 3D Medical Data
Jul 13, 2023



Abstract:Training Artificial Intelligence (AI) models on three-dimensional image data presents unique challenges compared to the two-dimensional case: Firstly, the computational resources are significantly higher, and secondly, the availability of large pretraining datasets is often limited, impeding training success. In this study, we propose a simple approach of adapting 2D networks with an intermediate feature representation for processing 3D volumes. Our method involves sequentially applying these networks to slices of a 3D volume from all orientations. Subsequently, a feature reduction module combines the extracted slice features into a single representation, which is then used for classification. We evaluate our approach on medical classification benchmarks and a real-world clinical dataset, demonstrating comparable results to existing methods. Furthermore, by employing attention pooling as a feature reduction module we obtain weighted importance values for each slice during the forward pass. We show that slices deemed important by our approach allow the inspection of the basis of a model's prediction.
Xplainer: From X-Ray Observations to Explainable Zero-Shot Diagnosis
Mar 27, 2023Abstract:Automated diagnosis prediction from medical images is a valuable resource to support clinical decision-making. However, such systems usually need to be trained on large amounts of annotated data, which often is scarce in the medical domain. Zero-shot methods address this challenge by allowing a flexible adaption to new settings with different clinical findings without relying on labeled data. Further, to integrate automated diagnosis in the clinical workflow, methods should be transparent and explainable, increasing medical professionals' trust and facilitating correctness verification. In this work, we introduce Xplainer, a novel framework for explainable zero-shot diagnosis in the clinical setting. Xplainer adapts the classification-by-description approach of contrastive vision-language models to the multi-label medical diagnosis task. Specifically, instead of directly predicting a diagnosis, we prompt the model to classify the existence of descriptive observations, which a radiologist would look for on an X-Ray scan, and use the descriptor probabilities to estimate the likelihood of a diagnosis. Our model is explainable by design, as the final diagnosis prediction is directly based on the prediction of the underlying descriptors. We evaluate Xplainer on two chest X-ray datasets, CheXpert and ChestX-ray14, and demonstrate its effectiveness in improving the performance and explainability of zero-shot diagnosis. Our results suggest that Xplainer provides a more detailed understanding of the decision-making process and can be a valuable tool for clinical diagnosis.
Private, fair and accurate: Training large-scale, privacy-preserving AI models in radiology
Feb 03, 2023Abstract:Artificial intelligence (AI) models are increasingly used in the medical domain. However, as medical data is highly sensitive, special precautions to ensure the protection of said data are required. The gold standard for privacy preservation is the introduction of differential privacy (DP) to model training. However, prior work has shown that DP has negative implications on model accuracy and fairness. Therefore, the purpose of this study is to demonstrate that the privacy-preserving training of AI models for chest radiograph diagnosis is possible with high accuracy and fairness compared to non-private training. N=193,311 high quality clinical chest radiographs were retrospectively collected and manually labeled by experienced radiologists, who assigned one or more of the following diagnoses: cardiomegaly, congestion, pleural effusion, pneumonic infiltration and atelectasis, to each side (where applicable). The non-private AI models were compared with privacy-preserving (DP) models with respect to privacy-utility trade-offs (measured as area under the receiver-operator-characteristic curve (AUROC)), and privacy-fairness trade-offs (measured as Pearson-R or Statistical Parity Difference). The non-private AI model achieved an average AUROC score of 0.90 over all labels, whereas the DP AI model with a privacy budget of epsilon=7.89 resulted in an AUROC of 0.87, i.e., a mere 2.6% performance decrease compared to non-private training. The privacy-preserving training of diagnostic AI models can achieve high performance with a small penalty on model accuracy and does not amplify discrimination against age, sex or co-morbidity. We thus encourage practitioners to integrate state-of-the-art privacy-preserving techniques into medical AI model development.
Current State of Community-Driven Radiological AI Deployment in Medical Imaging
Dec 29, 2022



Abstract:Artificial Intelligence (AI) has become commonplace to solve routine everyday tasks. Because of the exponential growth in medical imaging data volume and complexity, the workload on radiologists is steadily increasing. We project that the gap between the number of imaging exams and the number of expert radiologist readers required to cover this increase will continue to expand, consequently introducing a demand for AI-based tools that improve the efficiency with which radiologists can comfortably interpret these exams. AI has been shown to improve efficiency in medical-image generation, processing, and interpretation, and a variety of such AI models have been developed across research labs worldwide. However, very few of these, if any, find their way into routine clinical use, a discrepancy that reflects the divide between AI research and successful AI translation. To address the barrier to clinical deployment, we have formed MONAI Consortium, an open-source community which is building standards for AI deployment in healthcare institutions, and developing tools and infrastructure to facilitate their implementation. This report represents several years of weekly discussions and hands-on problem solving experience by groups of industry experts and clinicians in the MONAI Consortium. We identify barriers between AI-model development in research labs and subsequent clinical deployment and propose solutions. Our report provides guidance on processes which take an imaging AI model from development to clinical implementation in a healthcare institution. We discuss various AI integration points in a clinical Radiology workflow. We also present a taxonomy of Radiology AI use-cases. Through this report, we intend to educate the stakeholders in healthcare and AI (AI researchers, radiologists, imaging informaticists, and regulators) about cross-disciplinary challenges and possible solutions.
Exploiting segmentation labels and representation learning to forecast therapy response of PDAC patients
Nov 08, 2022


Abstract:The prediction of pancreatic ductal adenocarcinoma therapy response is a clinically challenging and important task in this high-mortality tumour entity. The training of neural networks able to tackle this challenge is impeded by a lack of large datasets and the difficult anatomical localisation of the pancreas. Here, we propose a hybrid deep neural network pipeline to predict tumour response to initial chemotherapy which is based on the Response Evaluation Criteria in Solid Tumors (RECIST) score, a standardised method for cancer response evaluation by clinicians as well as tumour markers, and clinical evaluation of the patients. We leverage a combination of representation transfer from segmentation to classification, as well as localisation and representation learning. Our approach yields a remarkably data-efficient method able to predict treatment response with a ROC-AUC of 63.7% using only 477 datasets in total.
Longitudinal Self-Supervision for COVID-19 Pathology Quantification
Mar 21, 2022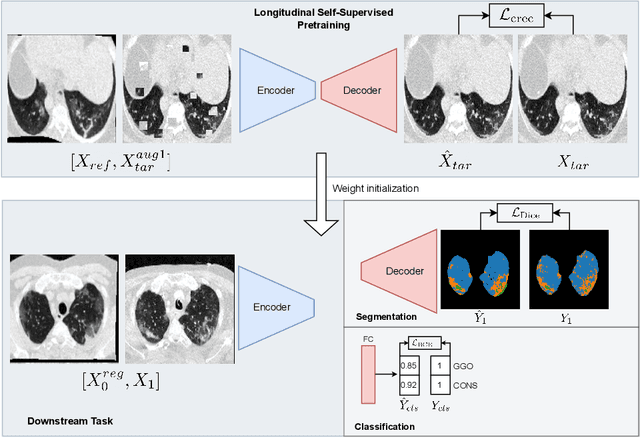
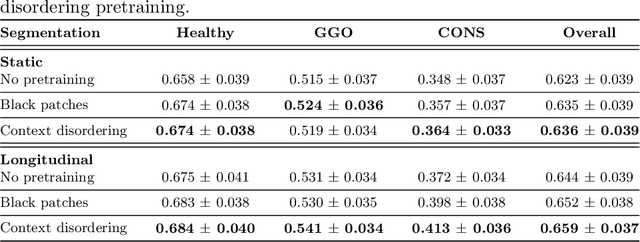
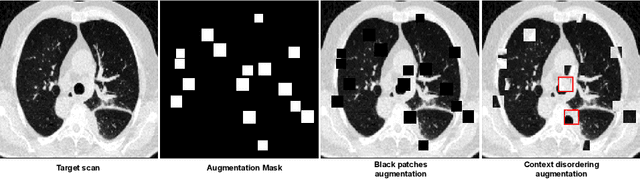

Abstract:Quantifying COVID-19 infection over time is an important task to manage the hospitalization of patients during a global pandemic. Recently, deep learning-based approaches have been proposed to help radiologists automatically quantify COVID-19 pathologies on longitudinal CT scans. However, the learning process of deep learning methods demands extensive training data to learn the complex characteristics of infected regions over longitudinal scans. It is challenging to collect a large-scale dataset, especially for longitudinal training. In this study, we want to address this problem by proposing a new self-supervised learning method to effectively train longitudinal networks for the quantification of COVID-19 infections. For this purpose, longitudinal self-supervision schemes are explored on clinical longitudinal COVID-19 CT scans. Experimental results show that the proposed method is effective, helping the model better exploit the semantics of longitudinal data and improve two COVID-19 quantification tasks.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge