Mahshad Lotfinia
Safety and accuracy follow different scaling laws in clinical large language models
May 05, 2026Abstract:Clinical LLMs are often scaled by increasing model size, context length, retrieval complexity, or inference-time compute, with the implicit expectation that higher accuracy implies safer behavior. This assumption is incomplete in medicine, where a few confident, high-risk, or evidence-contradicting errors can matter more than average benchmark performance. We introduce SaFE-Scale, a framework for measuring how clinical LLM safety changes across model scale, evidence quality, retrieval strategy, context exposure, and inference-time compute. To instantiate this framework, we introduce RadSaFE-200, a Radiology Safety-Focused Evaluation benchmark of 200 multiple-choice questions with clinician-defined clean evidence, conflict evidence, and option-level labels for high-risk error, unsafe answer, and evidence contradiction. We evaluated 34 locally deployed LLMs across six deployment conditions: closed-book prompting (zero-shot), clean evidence, conflict evidence, standard RAG, agentic RAG, and max-context prompting. Clean evidence produced the strongest improvement, increasing mean accuracy from 73.5% to 94.1%, while reducing high-risk error from 12.0% to 2.6%, contradiction from 12.7% to 2.3%, and dangerous overconfidence from 8.0% to 1.6%. Standard RAG and agentic RAG did not reproduce this safety profile: agentic RAG improved accuracy over standard RAG and reduced contradiction, but high-risk error and dangerous overconfidence remained elevated. Max-context prompting increased latency without closing the safety gap, and additional inference-time compute produced only limited gains. Worst-case analysis showed that clinically consequential errors concentrated in a small subset of questions. Clinical LLM safety is therefore not a passive consequence of scaling, but a deployment property shaped by evidence quality, retrieval design, context construction, and collective failure behavior.
Case-Grounded Evidence Verification: A Framework for Constructing Evidence-Sensitive Supervision
Apr 10, 2026Abstract:Evidence-grounded reasoning requires more than attaching retrieved text to a prediction: a model should make decisions that depend on whether the provided evidence supports the target claim. In practice, this often fails because supervision is weak, evidence is only loosely tied to the claim, and evaluation does not test evidence dependence directly. We introduce case-grounded evidence verification, a general framework in which a model receives a local case context, external evidence, and a structured claim, and must decide whether the evidence supports the claim for that case. Our key contribution is a supervision construction procedure that generates explicit support examples together with semantically controlled non-support examples, including counterfactual wrong-state and topic-related negatives, without manual evidence annotation. We instantiate the framework in radiology and train a standard verifier on the resulting support task. The learned verifier substantially outperforms both case-only and evidence-only baselines, remains strong under correct evidence, and collapses when evidence is removed or swapped, indicating genuine evidence dependence. This behavior transfers across unseen evidence articles and an external case distribution, though performance degrades under evidence-source shift and remains sensitive to backbone choice. Overall, the results suggest that a major bottleneck in evidence grounding is not only model capacity, but the lack of supervision that encodes the causal role of evidence.
The role of self-supervised pretraining in differentially private medical image analysis
Jan 27, 2026Abstract:Differential privacy (DP) provides formal protection for sensitive data but typically incurs substantial losses in diagnostic performance. Model initialization has emerged as a critical factor in mitigating this degradation, yet the role of modern self-supervised learning under full-model DP remains poorly understood. Here, we present a large-scale evaluation of initialization strategies for differentially private medical image analysis, using chest radiograph classification as a representative benchmark with more than 800,000 images. Using state-of-the-art ConvNeXt models trained with DP-SGD across realistic privacy regimes, we compare non-domain-specific supervised ImageNet initialization, non-domain-specific self-supervised DINOv3 initialization, and domain-specific supervised pretraining on MIMIC-CXR, the largest publicly available chest radiograph dataset. Evaluations are conducted across five external datasets spanning diverse institutions and acquisition settings. We show that DINOv3 initialization consistently improves diagnostic utility relative to ImageNet initialization under DP, but remains inferior to domain-specific supervised pretraining, which achieves performance closest to non-private baselines. We further demonstrate that initialization choice strongly influences demographic fairness, cross-dataset generalization, and robustness to data scale and model capacity under privacy constraints. The results establish initialization strategy as a central determinant of utility, fairness, and generalization in differentially private medical imaging.
Agentic large language models improve retrieval-based radiology question answering
Aug 01, 2025



Abstract:Clinical decision-making in radiology increasingly benefits from artificial intelligence (AI), particularly through large language models (LLMs). However, traditional retrieval-augmented generation (RAG) systems for radiology question answering (QA) typically rely on single-step retrieval, limiting their ability to handle complex clinical reasoning tasks. Here we propose an agentic RAG framework enabling LLMs to autonomously decompose radiology questions, iteratively retrieve targeted clinical evidence from Radiopaedia, and dynamically synthesize evidence-based responses. We evaluated 24 LLMs spanning diverse architectures, parameter scales (0.5B to >670B), and training paradigms (general-purpose, reasoning-optimized, clinically fine-tuned), using 104 expert-curated radiology questions from previously established RSNA-RadioQA and ExtendedQA datasets. Agentic retrieval significantly improved mean diagnostic accuracy over zero-shot prompting (73% vs. 64%; P<0.001) and conventional online RAG (73% vs. 68%; P<0.001). The greatest gains occurred in mid-sized models (e.g., Mistral Large improved from 72% to 81%) and small-scale models (e.g., Qwen 2.5-7B improved from 55% to 71%), while very large models (>200B parameters) demonstrated minimal changes (<2% improvement). Additionally, agentic retrieval reduced hallucinations (mean 9.4%) and retrieved clinically relevant context in 46% of cases, substantially aiding factual grounding. Even clinically fine-tuned models exhibited meaningful improvements (e.g., MedGemma-27B improved from 71% to 81%), indicating complementary roles of retrieval and fine-tuning. These results highlight the potential of agentic frameworks to enhance factuality and diagnostic accuracy in radiology QA, particularly among mid-sized LLMs, warranting future studies to validate their clinical utility.
Perceptual Implications of Automatic Anonymization in Pathological Speech
May 01, 2025Abstract:Automatic anonymization techniques are essential for ethical sharing of pathological speech data, yet their perceptual consequences remain understudied. This study presents the first comprehensive human-centered analysis of anonymized pathological speech, using a structured perceptual protocol involving ten native and non-native German listeners with diverse linguistic, clinical, and technical backgrounds. Listeners evaluated anonymized-original utterance pairs from 180 speakers spanning Cleft Lip and Palate, Dysarthria, Dysglossia, Dysphonia, and age-matched healthy controls. Speech was anonymized using state-of-the-art automatic methods (equal error rates in the range of 30-40%). Listeners completed Turing-style discrimination and quality rating tasks under zero-shot (single-exposure) and few-shot (repeated-exposure) conditions. Discrimination accuracy was high overall (91% zero-shot; 93% few-shot), but varied by disorder (repeated-measures ANOVA: p=0.007), ranging from 96% (Dysarthria) to 86% (Dysphonia). Anonymization consistently reduced perceived quality (from 83% to 59%, p<0.001), with pathology-specific degradation patterns (one-way ANOVA: p=0.005). Native listeners rated original speech slightly higher than non-native listeners (Delta=4%, p=0.199), but this difference nearly disappeared after anonymization (Delta=1%, p=0.724). No significant gender-based bias was observed. Critically, human perceptual outcomes did not correlate with automatic privacy or clinical utility metrics. These results underscore the need for listener-informed, disorder- and context-specific anonymization strategies that preserve privacy while maintaining interpretability, communicative functions, and diagnostic utility, especially for vulnerable populations such as children.
Boosting multi-demographic federated learning for chest x-ray analysis using general-purpose self-supervised representations
Apr 11, 2025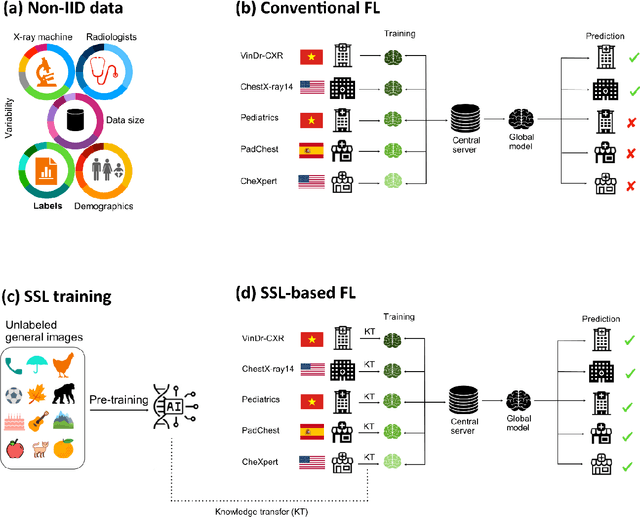



Abstract:Reliable artificial intelligence (AI) models for medical image analysis often depend on large and diverse labeled datasets. Federated learning (FL) offers a decentralized and privacy-preserving approach to training but struggles in highly non-independent and identically distributed (non-IID) settings, where institutions with more representative data may experience degraded performance. Moreover, existing large-scale FL studies have been limited to adult datasets, neglecting the unique challenges posed by pediatric data, which introduces additional non-IID variability. To address these limitations, we analyzed n=398,523 adult chest radiographs from diverse institutions across multiple countries and n=9,125 pediatric images, leveraging transfer learning from general-purpose self-supervised image representations to classify pneumonia and cases with no abnormality. Using state-of-the-art vision transformers, we found that FL improved performance only for smaller adult datasets (P<0.001) but degraded performance for larger datasets (P<0.064) and pediatric cases (P=0.242). However, equipping FL with self-supervised weights significantly enhanced outcomes across pediatric cases (P=0.031) and most adult datasets (P<0.008), except the largest dataset (P=0.052). These findings underscore the potential of easily deployable general-purpose self-supervised image representations to address non-IID challenges in clinical FL applications and highlight their promise for enhancing patient outcomes and advancing pediatric healthcare, where data scarcity and variability remain persistent obstacles.
Differential privacy for protecting patient data in speech disorder detection using deep learning
Sep 27, 2024



Abstract:Speech pathology has impacts on communication abilities and quality of life. While deep learning-based models have shown potential in diagnosing these disorders, the use of sensitive data raises critical privacy concerns. Although differential privacy (DP) has been explored in the medical imaging domain, its application in pathological speech analysis remains largely unexplored despite the equally critical privacy concerns. This study is the first to investigate DP's impact on pathological speech data, focusing on the trade-offs between privacy, diagnostic accuracy, and fairness. Using a large, real-world dataset of 200 hours of recordings from 2,839 German-speaking participants, we observed a maximum accuracy reduction of 3.85% when training with DP with a privacy budget, denoted by {\epsilon}, of 7.51. To generalize our findings, we validated our approach on a smaller dataset of Spanish-speaking Parkinson's disease patients, demonstrating that careful pretraining on large-scale task-specific datasets can maintain or even improve model accuracy under DP constraints. We also conducted a comprehensive fairness analysis, revealing that reasonable privacy levels (2<{\epsilon}<10) do not introduce significant gender bias, though age-related disparities may require further attention. Our results suggest that DP can effectively balance privacy and utility in speech disorder detection, but also highlight the unique challenges in the speech domain, particularly regarding the privacy-fairness trade-off. This provides a foundation for future work to refine DP methodologies and address fairness across diverse patient groups in real-world deployments.
RadioRAG: Factual Large Language Models for Enhanced Diagnostics in Radiology Using Dynamic Retrieval Augmented Generation
Jul 22, 2024



Abstract:Large language models (LLMs) have advanced the field of artificial intelligence (AI) in medicine. However LLMs often generate outdated or inaccurate information based on static training datasets. Retrieval augmented generation (RAG) mitigates this by integrating outside data sources. While previous RAG systems used pre-assembled, fixed databases with limited flexibility, we have developed Radiology RAG (RadioRAG) as an end-to-end framework that retrieves data from authoritative radiologic online sources in real-time. RadioRAG is evaluated using a dedicated radiologic question-and-answer dataset (RadioQA). We evaluate the diagnostic accuracy of various LLMs when answering radiology-specific questions with and without access to additional online information via RAG. Using 80 questions from RSNA Case Collection across radiologic subspecialties and 24 additional expert-curated questions, for which the correct gold-standard answers were available, LLMs (GPT-3.5-turbo, GPT-4, Mistral-7B, Mixtral-8x7B, and Llama3 [8B and 70B]) were prompted with and without RadioRAG. RadioRAG retrieved context-specific information from www.radiopaedia.org in real-time and incorporated them into its reply. RadioRAG consistently improved diagnostic accuracy across all LLMs, with relative improvements ranging from 2% to 54%. It matched or exceeded question answering without RAG across radiologic subspecialties, particularly in breast imaging and emergency radiology. However, degree of improvement varied among models; GPT-3.5-turbo and Mixtral-8x7B-instruct-v0.1 saw notable gains, while Mistral-7B-instruct-v0.2 showed no improvement, highlighting variability in its effectiveness. LLMs benefit when provided access to domain-specific data beyond their training data. For radiology, RadioRAG establishes a robust framework that substantially improves diagnostic accuracy and factuality in radiological question answering.
Empowering Clinicians and Democratizing Data Science: Large Language Models Automate Machine Learning for Clinical Studies
Aug 29, 2023Abstract:A knowledge gap persists between Machine Learning (ML) developers (e.g., data scientists) and practitioners (e.g., clinicians), hampering the full utilization of ML for clinical data analysis. We investigated the potential of the chatGPT Advanced Data Analysis (ADA), an extension of GPT-4, to bridge this gap and perform ML analyses efficiently. Real-world clinical datasets and study details from large trials across various medical specialties were presented to chatGPT ADA without specific guidance. ChatGPT ADA autonomously developed state-of-the-art ML models based on the original study's training data to predict clinical outcomes such as cancer development, cancer progression, disease complications, or biomarkers such as pathogenic gene sequences. Strikingly, these ML models matched or outperformed their published counterparts. We conclude that chatGPT ADA offers a promising avenue to democratize ML in medicine, making advanced analytics accessible to non-ML experts and promoting broader applications in medical research and practice.
Preserving privacy in domain transfer of medical AI models comes at no performance costs: The integral role of differential privacy
Jun 10, 2023



Abstract:Developing robust and effective artificial intelligence (AI) models in medicine requires access to large amounts of patient data. The use of AI models solely trained on large multi-institutional datasets can help with this, yet the imperative to ensure data privacy remains, particularly as membership inference risks breaching patient confidentiality. As a proposed remedy, we advocate for the integration of differential privacy (DP). We specifically investigate the performance of models trained with DP as compared to models trained without DP on data from institutions that the model had not seen during its training (i.e., external validation) - the situation that is reflective of the clinical use of AI models. By leveraging more than 590,000 chest radiographs from five institutions, we evaluated the efficacy of DP-enhanced domain transfer (DP-DT) in diagnosing cardiomegaly, pleural effusion, pneumonia, atelectasis, and in identifying healthy subjects. We juxtaposed DP-DT with non-DP-DT and examined diagnostic accuracy and demographic fairness using the area under the receiver operating characteristic curve (AUC) as the main metric, as well as accuracy, sensitivity, and specificity. Our results show that DP-DT, even with exceptionally high privacy levels (epsilon around 1), performs comparably to non-DP-DT (P>0.119 across all domains). Furthermore, DP-DT led to marginal AUC differences - less than 1% - for nearly all subgroups, relative to non-DP-DT. Despite consistent evidence suggesting that DP models induce significant performance degradation for on-domain applications, we show that off-domain performance is almost not affected. Therefore, we ardently advocate for the adoption of DP in training diagnostic medical AI models, given its minimal impact on performance.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge