Michael Markl
Enabling Ultra-Fast Cardiovascular Imaging Across Heterogeneous Clinical Environments with a Generalist Foundation Model and Multimodal Database
Dec 25, 2025Abstract:Multimodal cardiovascular magnetic resonance (CMR) imaging provides comprehensive and non-invasive insights into cardiovascular disease (CVD) diagnosis and underlying mechanisms. Despite decades of advancements, its widespread clinical adoption remains constrained by prolonged scan times and heterogeneity across medical environments. This underscores the urgent need for a generalist reconstruction foundation model for ultra-fast CMR imaging, one capable of adapting across diverse imaging scenarios and serving as the essential substrate for all downstream analyses. To enable this goal, we curate MMCMR-427K, the largest and most comprehensive multimodal CMR k-space database to date, comprising 427,465 multi-coil k-space data paired with structured metadata across 13 international centers, 12 CMR modalities, 15 scanners, and 17 CVD categories in populations across three continents. Building on this unprecedented resource, we introduce CardioMM, a generalist reconstruction foundation model capable of dynamically adapting to heterogeneous fast CMR imaging scenarios. CardioMM unifies semantic contextual understanding with physics-informed data consistency to deliver robust reconstructions across varied scanners, protocols, and patient presentations. Comprehensive evaluations demonstrate that CardioMM achieves state-of-the-art performance in the internal centers and exhibits strong zero-shot generalization to unseen external settings. Even at imaging acceleration up to 24x, CardioMM reliably preserves key cardiac phenotypes, quantitative myocardial biomarkers, and diagnostic image quality, enabling a substantial increase in CMR examination throughput without compromising clinical integrity. Together, our open-access MMCMR-427K database and CardioMM framework establish a scalable pathway toward high-throughput, high-quality, and clinically accessible cardiovascular imaging.
Towards Universal Learning-based Model for Cardiac Image Reconstruction: Summary of the CMRxRecon2024 Challenge
Mar 05, 2025Abstract:Cardiovascular magnetic resonance (CMR) offers diverse imaging contrasts for assessment of cardiac function and tissue characterization. However, acquiring each single CMR modality is often time-consuming, and comprehensive clinical protocols require multiple modalities with various sampling patterns, further extending the overall acquisition time and increasing susceptibility to motion artifacts. Existing deep learning-based reconstruction methods are often designed for specific acquisition parameters, which limits their ability to generalize across a variety of scan scenarios. As part of the CMRxRecon Series, the CMRxRecon2024 challenge provides diverse datasets encompassing multi-modality multi-view imaging with various sampling patterns, and a platform for the international community to develop and benchmark reconstruction solutions in two well-crafted tasks. Task 1 is a modality-universal setting, evaluating the out-of-distribution generalization of the reconstructed model, while Task 2 follows sampling-universal setting assessing the one-for-all adaptability of the universal model. Main contributions include providing the first and largest publicly available multi-modality, multi-view cardiac k-space dataset; developing a benchmarking platform that simulates clinical acceleration protocols, with a shared code library and tutorial for various k-t undersampling patterns and data processing; giving technical insights of enhanced data consistency based on physic-informed networks and adaptive prompt-learning embedding to be versatile to different clinical settings; additional finding on evaluation metrics to address the limitations of conventional ground-truth references in universal reconstruction tasks.
CMRxRecon2024: A Multi-Modality, Multi-View K-Space Dataset Boosting Universal Machine Learning for Accelerated Cardiac MRI
Jun 27, 2024



Abstract:Cardiac magnetic resonance imaging (MRI) has emerged as a clinically gold-standard technique for diagnosing cardiac diseases, thanks to its ability to provide diverse information with multiple modalities and anatomical views. Accelerated cardiac MRI is highly expected to achieve time-efficient and patient-friendly imaging, and then advanced image reconstruction approaches are required to recover high-quality, clinically interpretable images from undersampled measurements. However, the lack of publicly available cardiac MRI k-space dataset in terms of both quantity and diversity has severely hindered substantial technological progress, particularly for data-driven artificial intelligence. Here, we provide a standardized, diverse, and high-quality CMRxRecon2024 dataset to facilitate the technical development, fair evaluation, and clinical transfer of cardiac MRI reconstruction approaches, towards promoting the universal frameworks that enable fast and robust reconstructions across different cardiac MRI protocols in clinical practice. To the best of our knowledge, the CMRxRecon2024 dataset is the largest and most diverse publicly available cardiac k-space dataset. It is acquired from 330 healthy volunteers, covering commonly used modalities, anatomical views, and acquisition trajectories in clinical cardiac MRI workflows. Besides, an open platform with tutorials, benchmarks, and data processing tools is provided to facilitate data usage, advanced method development, and fair performance evaluation.
A deep learning approach to using wearable seismocardiography for diagnosing aortic valve stenosis and predicting aortic hemodynamics obtained by 4D flow MRI
Jan 05, 2023Abstract:In this paper, we explored the use of deep learning for the prediction of aortic flow metrics obtained using 4D flow MRI using wearable seismocardiography (SCG) devices. 4D flow MRI provides a comprehensive assessment of cardiovascular hemodynamics, but it is costly and time-consuming. We hypothesized that deep learning could be used to identify pathological changes in blood flow, such as elevated peak systolic velocity Vmax in patients with heart valve diseases, from SCG signals. We also investigated the ability of this deep learning technique to differentiate between patients diagnosed with aortic valve stenosis (AS), non-AS patients with a bicuspid aortic valve (BAV), non-AS patients with a mechanical aortic valve (MAV), and healthy subjects with a normal tricuspid aortic valve (TAV). In a study of 77 subjects who underwent same-day 4D flow MRI and SCG, we found that the Vmax values obtained using deep learning and SCGs were in good agreement with those obtained by 4D flow MRI. Additionally, subjects with TAV, BAV, MAV, and AS could be classified with ROC-AUC values of 92%, 95%, 81%, and 83%, respectively. This suggests that SCG obtained using low-cost wearable electronics may be used as a supplement to 4D flow MRI exams or as a screening tool for aortic valve disease.
MRI-MECH: Mechanics-informed MRI to estimate esophageal health
Sep 15, 2022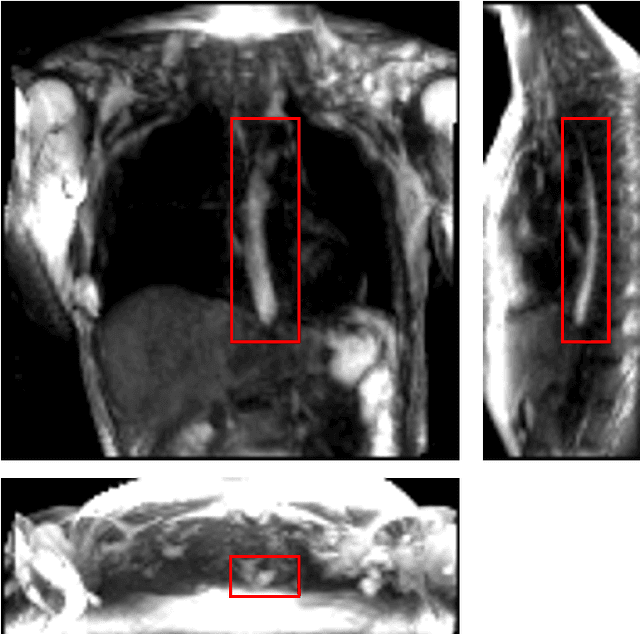
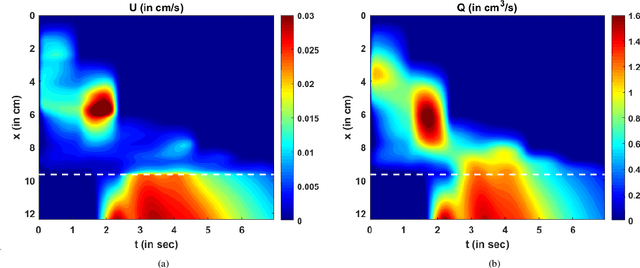
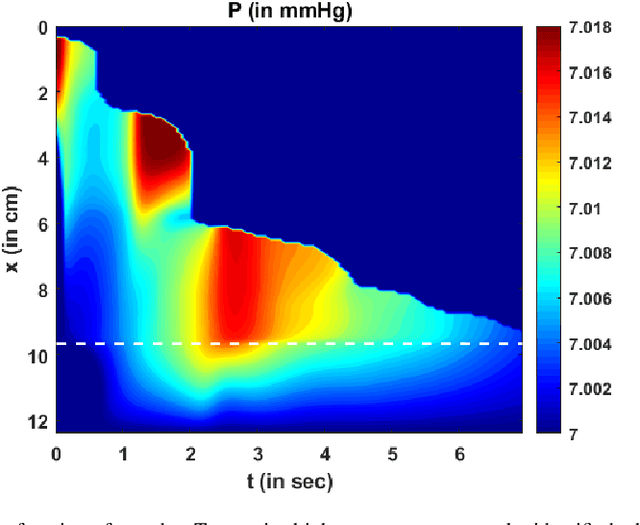
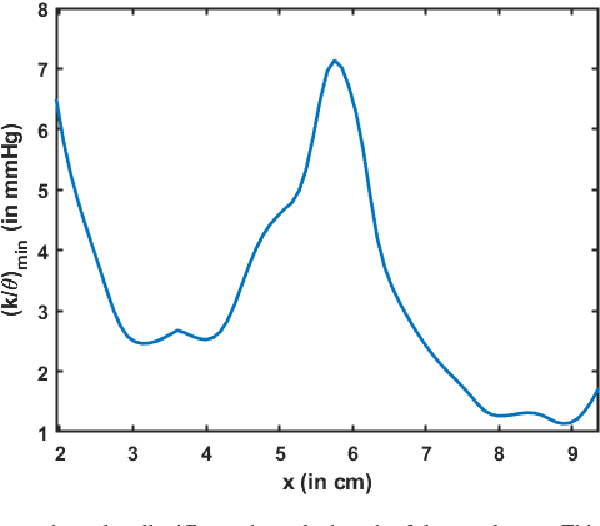
Abstract:Dynamic magnetic resonance imaging (MRI) is a popular medical imaging technique to generate image sequences of the flow of a contrast material inside tissues and organs. However, its application to imaging bolus movement through the esophagus has only been demonstrated in few feasibility studies and is relatively unexplored. In this work, we present a computational framework called mechanics-informed MRI (MRI-MECH) that enhances that capability thereby increasing the applicability of dynamic MRI for diagnosing esophageal disorders. Pineapple juice was used as the swallowed contrast material for the dynamic MRI and the MRI image sequence was used as input to the MRI-MECH. The MRI-MECH modeled the esophagus as a flexible one-dimensional tube and the elastic tube walls followed a linear tube law. Flow through the esophagus was then governed by one-dimensional mass and momentum conservation equations. These equations were solved using a physics-informed neural network (PINN). The PINN minimized the difference between the measurements from the MRI and model predictions ensuring that the physics of the fluid flow problem was always followed. MRI-MECH calculated the fluid velocity and pressure during esophageal transit and estimated the mechanical health of the esophagus by calculating wall stiffness and active relaxation. Additionally, MRI-MECH predicted missing information about the lower esophageal sphincter during the emptying process, demonstrating its applicability to scenarios with missing data or poor image resolution. In addition to potentially improving clinical decisions based on quantitative estimates of the mechanical health of the esophagus, MRI-MECH can also be enhanced for application to other medical imaging modalities to enhance their functionality as well.
Machine-Learned Prediction Equilibrium for Dynamic Traffic Assignment
Sep 14, 2021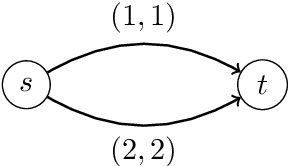
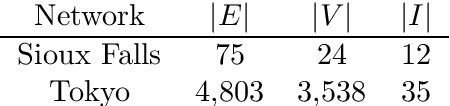
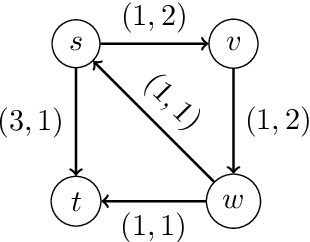
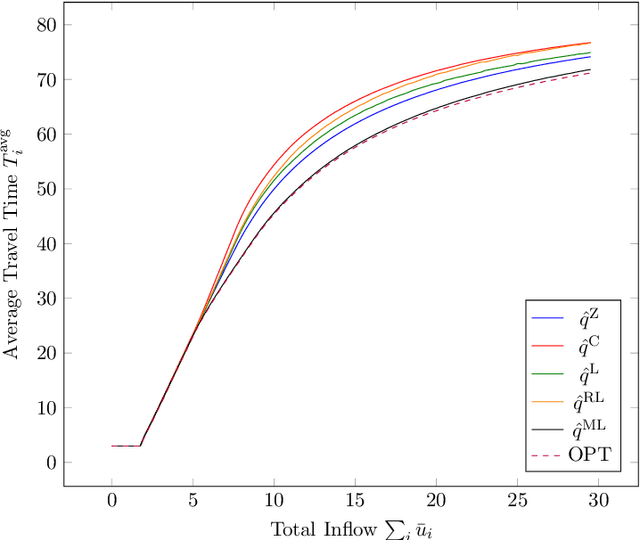
Abstract:We study a dynamic traffic assignment model, where agents base their instantaneous routing decisions on real-time delay predictions. We formulate a mathematically concise model and derive properties of the predictors that ensure a dynamic prediction equilibrium exists. We demonstrate the versatility of our framework by showing that it subsumes the well-known full information and instantaneous information models, in addition to admitting further realistic predictors as special cases. We complement our theoretical analysis by an experimental study, in which we systematically compare the induced average travel times of different predictors, including a machine-learning model trained on data gained from previously computed equilibrium flows, both on a synthetic and a real road network.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge