Maria Robu
Confidence-aware Monocular Depth Estimation for Minimally Invasive Surgery
Mar 03, 2026Abstract:Purpose: Monocular depth estimation (MDE) is vital for scene understanding in minimally invasive surgery (MIS). However, endoscopic video sequences are often contaminated by smoke, specular reflections, blur, and occlusions, limiting the accuracy of MDE models. In addition, current MDE models do not output depth confidence, which could be a valuable tool for improving their clinical reliability. Methods: We propose a novel confidence-aware MDE framework featuring three significant contributions: (i) Calibrated confidence targets: an ensemble of fine-tuned stereo matching models is used to capture disparity variance into pixel-wise confidence probabilities; (ii) Confidence-aware loss: Baseline MDE models are optimized with confidence-aware loss functions, utilizing pixel-wise confidence probabilities such that reliable pixels dominate training; and (iii) Inference-time confidence: a confidence estimation head is proposed with two convolution layers to predict per-pixel confidence at inference, enabling assessment of depth reliability. Results: Comprehensive experimental validation across internal and public datasets demonstrates that our framework improves depth estimation accuracy and can robustly quantify the prediction's confidence. On the internal clinical endoscopic dataset (StereoKP), we improve dense depth estimation accuracy by ~8% as compared to the baseline model. Conclusion: Our confidence-aware framework enables improved accuracy of MDE models in MIS, addressing challenges posed by noise and artifacts in pre-clinical and clinical data, and allows MDE models to provide confidence maps that may be used to improve their reliability for clinical applications.
CholecTriplet2021: A benchmark challenge for surgical action triplet recognition
Apr 10, 2022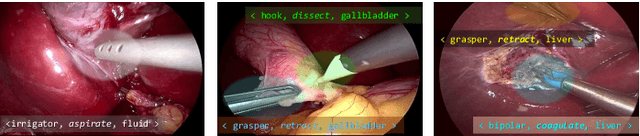
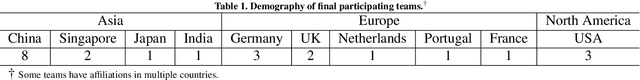
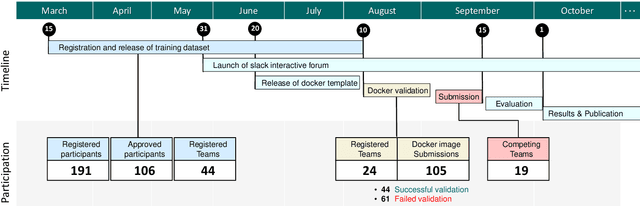
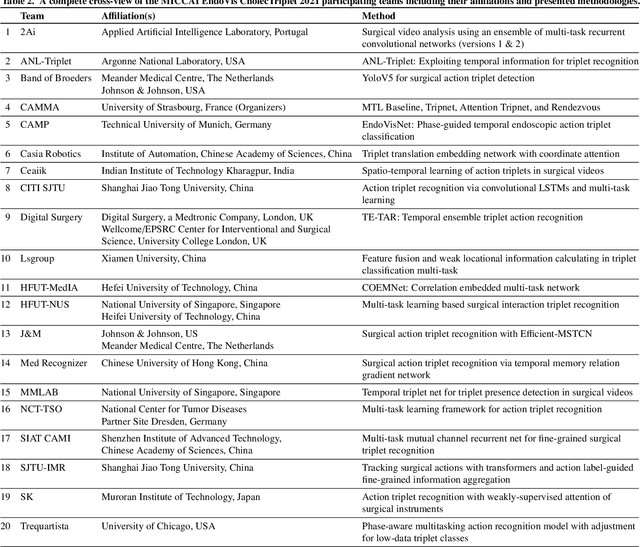
Abstract:Context-aware decision support in the operating room can foster surgical safety and efficiency by leveraging real-time feedback from surgical workflow analysis. Most existing works recognize surgical activities at a coarse-grained level, such as phases, steps or events, leaving out fine-grained interaction details about the surgical activity; yet those are needed for more helpful AI assistance in the operating room. Recognizing surgical actions as triplets of <instrument, verb, target> combination delivers comprehensive details about the activities taking place in surgical videos. This paper presents CholecTriplet2021: an endoscopic vision challenge organized at MICCAI 2021 for the recognition of surgical action triplets in laparoscopic videos. The challenge granted private access to the large-scale CholecT50 dataset, which is annotated with action triplet information. In this paper, we present the challenge setup and assessment of the state-of-the-art deep learning methods proposed by the participants during the challenge. A total of 4 baseline methods from the challenge organizers and 19 new deep learning algorithms by competing teams are presented to recognize surgical action triplets directly from surgical videos, achieving mean average precision (mAP) ranging from 4.2% to 38.1%. This study also analyzes the significance of the results obtained by the presented approaches, performs a thorough methodological comparison between them, in-depth result analysis, and proposes a novel ensemble method for enhanced recognition. Our analysis shows that surgical workflow analysis is not yet solved, and also highlights interesting directions for future research on fine-grained surgical activity recognition which is of utmost importance for the development of AI in surgery.
Fast Estimation of Haemoglobin Concentration in Tissue Via Wavelet Decomposition
Jun 22, 2017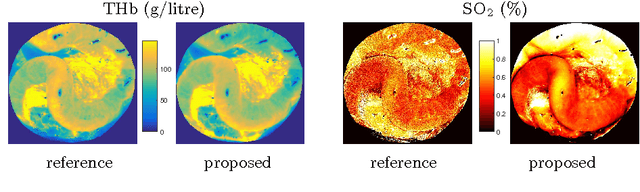
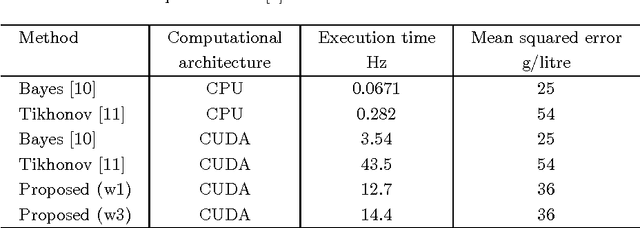
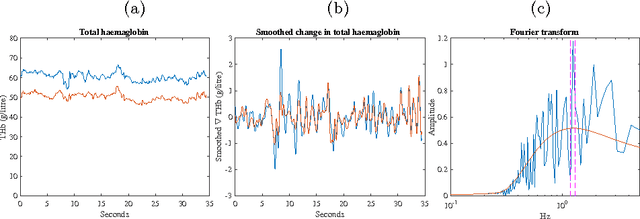
Abstract:Tissue oxygenation and perfusion can be an indicator for organ viability during minimally invasive surgery, for example allowing real-time assessment of tissue perfusion and oxygen saturation. Multispectral imaging is an optical modality that can inspect tissue perfusion in wide field images without contact. In this paper, we present a novel, fast method for using RGB images for MSI, which while limiting the spectral resolution of the modality allows normal laparoscopic systems to be used. We exploit the discrete Haar decomposition to separate individual video frames into low pass and directional coefficients and we utilise a different multispectral estimation technique on each. The increase in speed is achieved by using fast Tikhonov regularisation on the directional coefficients and more accurate Bayesian estimation on the low pass component. The pipeline is implemented using a graphics processing unit (GPU) architecture and achieves a frame rate of approximately 15Hz. We validate the method on animal models and on human data captured using a da Vinci stereo laparoscope.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge