Iman Azimi
Personalized Digital Health Modeling with Adaptive Support Users
May 03, 2026Abstract:Personalized models are essential in digital health because individuals exhibit substantial physiological and behavioral heterogeneity. Yet personalization is limited by scarce and noisy user-specific data. Most existing methods rely on population pretraining or data from similar users only, which can lead to biased transfer and weak generalization. We propose a unified personalization framework that trains a personal model using adaptively weighted support users, including both similar and dissimilar individuals. The objective integrates personal loss, similarity-weighted transfer from similar users, and contrastive regularization from dissimilar users to suppress misleading correlations. An iterative optimization algorithm jointly updates model parameters and user similarity weights. Experiments on six tasks across four real-world digital health datasets show consistent improvements over population and personalized baselines. The method achieves up to 10% lower RMSE on large-scale datasets and approximately 25% lower RMSE in low-data settings. The learned adaptive weights improve data efficiency and provide interpretable guidance for targeted data selection.
Multimodal Sleep Stage and Sleep Apnea Classification Using Vision Transformer: A Multitask Explainable Learning Approach
Feb 18, 2025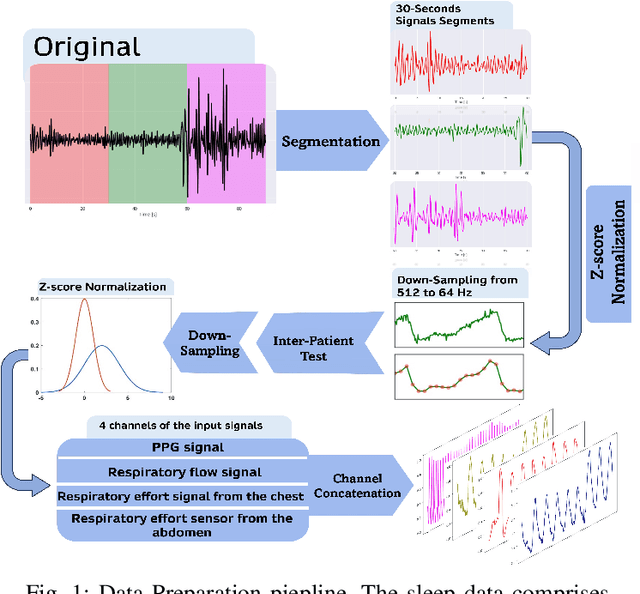
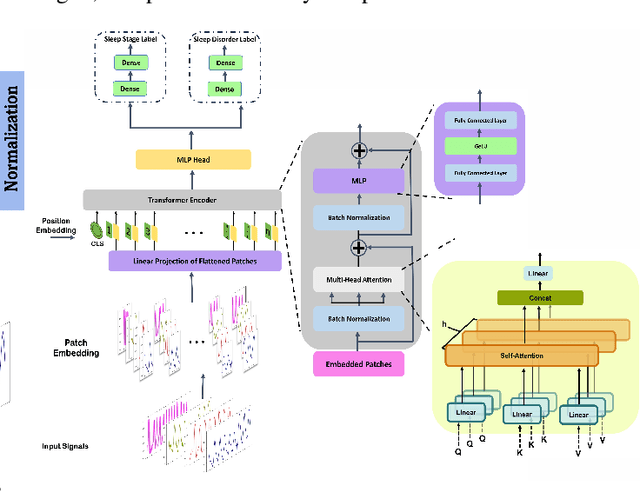

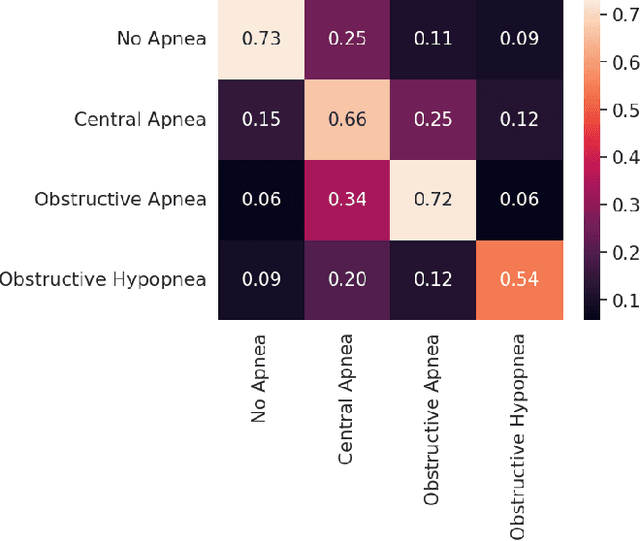
Abstract:Sleep is an essential component of human physiology, contributing significantly to overall health and quality of life. Accurate sleep staging and disorder detection are crucial for assessing sleep quality. Studies in the literature have proposed PSG-based approaches and machine-learning methods utilizing single-modality signals. However, existing methods often lack multimodal, multilabel frameworks and address sleep stages and disorders classification separately. In this paper, we propose a 1D-Vision Transformer for simultaneous classification of sleep stages and sleep disorders. Our method exploits the sleep disorders' correlation with specific sleep stage patterns and performs a simultaneous identification of a sleep stage and sleep disorder. The model is trained and tested using multimodal-multilabel sensory data (including photoplethysmogram, respiratory flow, and respiratory effort signals). The proposed method shows an overall accuracy (cohen's Kappa) of 78% (0.66) for five-stage sleep classification and 74% (0.58) for sleep apnea classification. Moreover, we analyzed the encoder attention weights to clarify our models' predictions and investigate the influence different features have on the models' outputs. The result shows that identified patterns, such as respiratory troughs and peaks, make a higher contribution to the final classification process.
An LLM-Powered Agent for Physiological Data Analysis: A Case Study on PPG-based Heart Rate Estimation
Feb 18, 2025Abstract:Large language models (LLMs) are revolutionizing healthcare by improving diagnosis, patient care, and decision support through interactive communication. More recently, they have been applied to analyzing physiological time-series like wearable data for health insight extraction. Existing methods embed raw numerical sequences directly into prompts, which exceeds token limits and increases computational costs. Additionally, some studies integrated features extracted from time-series in textual prompts or applied multimodal approaches. However, these methods often produce generic and unreliable outputs due to LLMs' limited analytical rigor and inefficiency in interpreting continuous waveforms. In this paper, we develop an LLM-powered agent for physiological time-series analysis aimed to bridge the gap in integrating LLMs with well-established analytical tools. Built on the OpenCHA, an open-source LLM-powered framework, our agent features an orchestrator that integrates user interaction, data sources, and analytical tools to generate accurate health insights. To evaluate its effectiveness, we implement a case study on heart rate (HR) estimation from Photoplethysmogram (PPG) signals using a dataset of PPG and Electrocardiogram (ECG) recordings in a remote health monitoring study. The agent's performance is benchmarked against OpenAI GPT-4o-mini and GPT-4o, with ECG serving as the gold standard for HR estimation. Results demonstrate that our agent significantly outperforms benchmark models by achieving lower error rates and more reliable HR estimations. The agent implementation is publicly available on GitHub.
Accuracy and Consistency of LLMs in the Registered Dietitian Exam: The Impact of Prompt Engineering and Knowledge Retrieval
Aug 06, 2024



Abstract:Large language models (LLMs) are fundamentally transforming human-facing applications in the health and well-being domains: boosting patient engagement, accelerating clinical decision-making, and facilitating medical education. Although state-of-the-art LLMs have shown superior performance in several conversational applications, evaluations within nutrition and diet applications are still insufficient. In this paper, we propose to employ the Registered Dietitian (RD) exam to conduct a standard and comprehensive evaluation of state-of-the-art LLMs, GPT-4o, Claude 3.5 Sonnet, and Gemini 1.5 Pro, assessing both accuracy and consistency in nutrition queries. Our evaluation includes 1050 RD exam questions encompassing several nutrition topics and proficiency levels. In addition, for the first time, we examine the impact of Zero-Shot (ZS), Chain of Thought (CoT), Chain of Thought with Self Consistency (CoT-SC), and Retrieval Augmented Prompting (RAP) on both accuracy and consistency of the responses. Our findings revealed that while these LLMs obtained acceptable overall performance, their results varied considerably with different prompts and question domains. GPT-4o with CoT-SC prompting outperformed the other approaches, whereas Gemini 1.5 Pro with ZS recorded the highest consistency. For GPT-4o and Claude 3.5, CoT improved the accuracy, and CoT-SC improved both accuracy and consistency. RAP was particularly effective for GPT-4o to answer Expert level questions. Consequently, choosing the appropriate LLM and prompting technique, tailored to the proficiency level and specific domain, can mitigate errors and potential risks in diet and nutrition chatbots.
Building Trust in Mental Health Chatbots: Safety Metrics and LLM-Based Evaluation Tools
Aug 03, 2024Abstract:Objective: This study aims to develop and validate an evaluation framework to ensure the safety and reliability of mental health chatbots, which are increasingly popular due to their accessibility, human-like interactions, and context-aware support. Materials and Methods: We created an evaluation framework with 100 benchmark questions and ideal responses, and five guideline questions for chatbot responses. This framework, validated by mental health experts, was tested on a GPT-3.5-turbo-based chatbot. Automated evaluation methods explored included large language model (LLM)-based scoring, an agentic approach using real-time data, and embedding models to compare chatbot responses against ground truth standards. Results: The results highlight the importance of guidelines and ground truth for improving LLM evaluation accuracy. The agentic method, dynamically accessing reliable information, demonstrated the best alignment with human assessments. Adherence to a standardized, expert-validated framework significantly enhanced chatbot response safety and reliability. Discussion: Our findings emphasize the need for comprehensive, expert-tailored safety evaluation metrics for mental health chatbots. While LLMs have significant potential, careful implementation is necessary to mitigate risks. The superior performance of the agentic approach underscores the importance of real-time data access in enhancing chatbot reliability. Conclusion: The study validated an evaluation framework for mental health chatbots, proving its effectiveness in improving safety and reliability. Future work should extend evaluations to accuracy, bias, empathy, and privacy to ensure holistic assessment and responsible integration into healthcare. Standardized evaluations will build trust among users and professionals, facilitating broader adoption and improved mental health support through technology.
Graph-Augmented LLMs for Personalized Health Insights: A Case Study in Sleep Analysis
Jun 25, 2024

Abstract:Health monitoring systems have revolutionized modern healthcare by enabling the continuous capture of physiological and behavioral data, essential for preventive measures and early health intervention. While integrating this data with Large Language Models (LLMs) has shown promise in delivering interactive health advice, traditional methods like Retrieval-Augmented Generation (RAG) and fine-tuning often fail to fully utilize the complex, multi-dimensional, and temporally relevant data from wearable devices. These conventional approaches typically provide limited actionable and personalized health insights due to their inadequate capacity to dynamically integrate and interpret diverse health data streams. In response, this paper introduces a graph-augmented LLM framework designed to significantly enhance the personalization and clarity of health insights. Utilizing a hierarchical graph structure, the framework captures inter and intra-patient relationships, enriching LLM prompts with dynamic feature importance scores derived from a Random Forest Model. The effectiveness of this approach is demonstrated through a sleep analysis case study involving 20 college students during the COVID-19 lockdown, highlighting the potential of our model to generate actionable and personalized health insights efficiently. We leverage another LLM to evaluate the insights for relevance, comprehensiveness, actionability, and personalization, addressing the critical need for models that process and interpret complex health data effectively. Our findings show that augmenting prompts with our framework yields significant improvements in all 4 criteria. Through our framework, we can elicit well-crafted, more thoughtful responses tailored to a specific patient.
Empathy Through Multimodality in Conversational Interfaces
May 08, 2024Abstract:Agents represent one of the most emerging applications of Large Language Models (LLMs) and Generative AI, with their effectiveness hinging on multimodal capabilities to navigate complex user environments. Conversational Health Agents (CHAs), a prime example of this, are redefining healthcare by offering nuanced support that transcends textual analysis to incorporate emotional intelligence. This paper introduces an LLM-based CHA engineered for rich, multimodal dialogue-especially in the realm of mental health support. It adeptly interprets and responds to users' emotional states by analyzing multimodal cues, thus delivering contextually aware and empathetically resonant verbal responses. Our implementation leverages the versatile openCHA framework, and our comprehensive evaluation involves neutral prompts expressed in diverse emotional tones: sadness, anger, and joy. We evaluate the consistency and repeatability of the planning capability of the proposed CHA. Furthermore, human evaluators critique the CHA's empathic delivery, with findings revealing a striking concordance between the CHA's outputs and evaluators' assessments. These results affirm the indispensable role of vocal (soon multimodal) emotion recognition in strengthening the empathetic connection built by CHAs, cementing their place at the forefront of interactive, compassionate digital health solutions.
ALCM: Autonomous LLM-Augmented Causal Discovery Framework
May 02, 2024



Abstract:To perform effective causal inference in high-dimensional datasets, initiating the process with causal discovery is imperative, wherein a causal graph is generated based on observational data. However, obtaining a complete and accurate causal graph poses a formidable challenge, recognized as an NP-hard problem. Recently, the advent of Large Language Models (LLMs) has ushered in a new era, indicating their emergent capabilities and widespread applicability in facilitating causal reasoning across diverse domains, such as medicine, finance, and science. The expansive knowledge base of LLMs holds the potential to elevate the field of causal reasoning by offering interpretability, making inferences, generalizability, and uncovering novel causal structures. In this paper, we introduce a new framework, named Autonomous LLM-Augmented Causal Discovery Framework (ALCM), to synergize data-driven causal discovery algorithms and LLMs, automating the generation of a more resilient, accurate, and explicable causal graph. The ALCM consists of three integral components: causal structure learning, causal wrapper, and LLM-driven causal refiner. These components autonomously collaborate within a dynamic environment to address causal discovery questions and deliver plausible causal graphs. We evaluate the ALCM framework by implementing two demonstrations on seven well-known datasets. Experimental results demonstrate that ALCM outperforms existing LLM methods and conventional data-driven causal reasoning mechanisms. This study not only shows the effectiveness of the ALCM but also underscores new research directions in leveraging the causal reasoning capabilities of LLMs.
Integrating Wearable Sensor Data and Self-reported Diaries for Personalized Affect Forecasting
Mar 23, 2024



Abstract:Emotional states, as indicators of affect, are pivotal to overall health, making their accurate prediction before onset crucial. Current studies are primarily centered on immediate short-term affect detection using data from wearable and mobile devices. These studies typically focus on objective sensory measures, often neglecting other forms of self-reported information like diaries and notes. In this paper, we propose a multimodal deep learning model for affect status forecasting. This model combines a transformer encoder with a pre-trained language model, facilitating the integrated analysis of objective metrics and self-reported diaries. To validate our model, we conduct a longitudinal study, enrolling college students and monitoring them over a year, to collect an extensive dataset including physiological, environmental, sleep, metabolic, and physical activity parameters, alongside open-ended textual diaries provided by the participants. Our results demonstrate that the proposed model achieves predictive accuracy of 82.50% for positive affect and 82.76% for negative affect, a full week in advance. The effectiveness of our model is further elevated by its explainability.
Knowledge-Infused LLM-Powered Conversational Health Agent: A Case Study for Diabetes Patients
Feb 28, 2024Abstract:Effective diabetes management is crucial for maintaining health in diabetic patients. Large Language Models (LLMs) have opened new avenues for diabetes management, facilitating their efficacy. However, current LLM-based approaches are limited by their dependence on general sources and lack of integration with domain-specific knowledge, leading to inaccurate responses. In this paper, we propose a knowledge-infused LLM-powered conversational health agent (CHA) for diabetic patients. We customize and leverage the open-source openCHA framework, enhancing our CHA with external knowledge and analytical capabilities. This integration involves two key components: 1) incorporating the American Diabetes Association dietary guidelines and the Nutritionix information and 2) deploying analytical tools that enable nutritional intake calculation and comparison with the guidelines. We compare the proposed CHA with GPT4. Our evaluation includes 100 diabetes-related questions on daily meal choices and assessing the potential risks associated with the suggested diet. Our findings show that the proposed agent demonstrates superior performance in generating responses to manage essential nutrients.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge