Yuli Jiang
Decision Support System for Chronic Diseases Based on Drug-Drug Interactions
Mar 04, 2023



Abstract:Many patients with chronic diseases resort to multiple medications to relieve various symptoms, which raises concerns about the safety of multiple medication use, as severe drug-drug antagonism can lead to serious adverse effects or even death. This paper presents a Decision Support System, called DSSDDI, based on drug-drug interactions to support doctors prescribing decisions. DSSDDI contains three modules, Drug-Drug Interaction (DDI) module, Medical Decision (MD) module and Medical Support (MS) module. The DDI module learns safer and more effective drug representations from the drug-drug interactions. To capture the potential causal relationship between DDI and medication use, the MD module considers the representations of patients and drugs as context, DDI and patients' similarity as treatment, and medication use as outcome to construct counterfactual links for the representation learning. Furthermore, the MS module provides drug candidates to doctors with explanations. Experiments on the chronic data collected from the Hong Kong Chronic Disease Study Project and a public diagnostic data MIMIC-III demonstrate that DSSDDI can be a reliable reference for doctors in terms of safety and efficiency of clinical diagnosis, with significant improvements compared to baseline methods.
QD-GCN: Query-Driven Graph Convolutional Networks for Attributed Community Search
Apr 08, 2021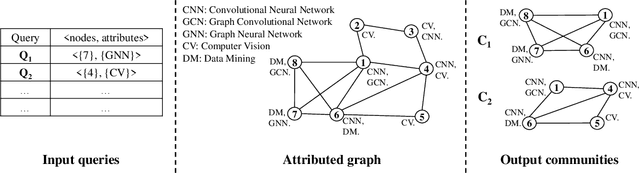
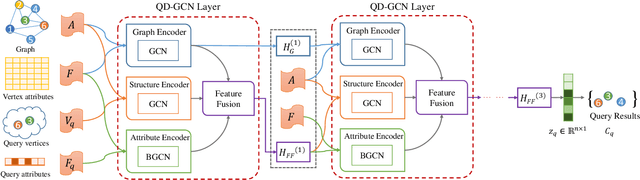
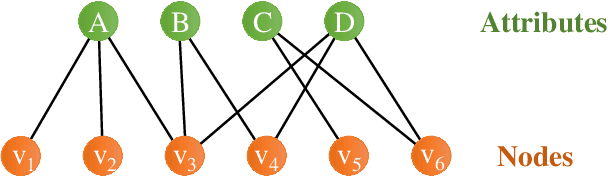
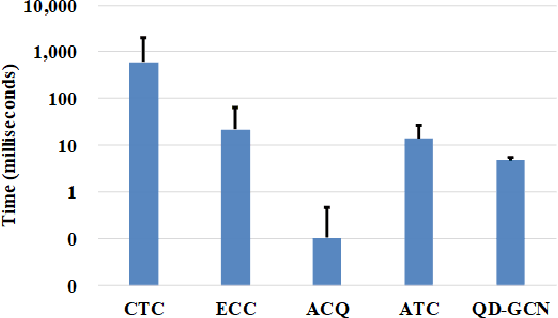
Abstract:Recently, attributed community search, a related but different problem to community detection and graph clustering, has been widely studied in the literature. Compared with the community detection that finds all existing static communities from a graph, the attributed community search (ACS) is more challenging since it aims to find dynamic communities with both cohesive structures and homogeneous node attributes given arbitrary queries. To solve the ACS problem, the most popular paradigm is to simplify the problem as two sub-problems, that is, structural matching and attribute filtering and deal with them separately. However, in real-world graphs, the community structure and the node attributes are actually correlated to each other. In this vein, current studies cannot capture these correlations which are vital for the ACS problem. In this paper, we propose Query-Driven Graph Convolutional Networks (QD-GCN), an end-to-end framework that unifies the community structure as well as node attribute to solve the ACS problem. In particular, QD-GCN leverages the Graph Convolutional Networks, which is a powerful tool to encode the graph topology and node attributes concurrently, as the backbones to extract the query-dependent community information from the original graph. By utilizing this query-dependent community information, QD-GCN is able to predict the target community given any queries. Experiments on real-world graphs with ground-truth communities demonstrate that QD-GCN outperforms existing attributed community search algorithms in terms of both efficiency and effectiveness.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge