Matthew Muckley
V-JEPA 2: Self-Supervised Video Models Enable Understanding, Prediction and Planning
Jun 11, 2025Abstract:A major challenge for modern AI is to learn to understand the world and learn to act largely by observation. This paper explores a self-supervised approach that combines internet-scale video data with a small amount of interaction data (robot trajectories), to develop models capable of understanding, predicting, and planning in the physical world. We first pre-train an action-free joint-embedding-predictive architecture, V-JEPA 2, on a video and image dataset comprising over 1 million hours of internet video. V-JEPA 2 achieves strong performance on motion understanding (77.3 top-1 accuracy on Something-Something v2) and state-of-the-art performance on human action anticipation (39.7 recall-at-5 on Epic-Kitchens-100) surpassing previous task-specific models. Additionally, after aligning V-JEPA 2 with a large language model, we demonstrate state-of-the-art performance on multiple video question-answering tasks at the 8 billion parameter scale (e.g., 84.0 on PerceptionTest, 76.9 on TempCompass). Finally, we show how self-supervised learning can be applied to robotic planning tasks by post-training a latent action-conditioned world model, V-JEPA 2-AC, using less than 62 hours of unlabeled robot videos from the Droid dataset. We deploy V-JEPA 2-AC zero-shot on Franka arms in two different labs and enable picking and placing of objects using planning with image goals. Notably, this is achieved without collecting any data from the robots in these environments, and without any task-specific training or reward. This work demonstrates how self-supervised learning from web-scale data and a small amount of robot interaction data can yield a world model capable of planning in the physical world.
Qinco2: Vector Compression and Search with Improved Implicit Neural Codebooks
Jan 06, 2025



Abstract:Vector quantization is a fundamental technique for compression and large-scale nearest neighbor search. For high-accuracy operating points, multi-codebook quantization associates data vectors with one element from each of multiple codebooks. An example is residual quantization (RQ), which iteratively quantizes the residual error of previous steps. Dependencies between the different parts of the code are, however, ignored in RQ, which leads to suboptimal rate-distortion performance. QINCo recently addressed this inefficiency by using a neural network to determine the quantization codebook in RQ based on the vector reconstruction from previous steps. In this paper we introduce QINCo2 which extends and improves QINCo with (i) improved vector encoding using codeword pre-selection and beam-search, (ii) a fast approximate decoder leveraging codeword pairs to establish accurate short-lists for search, and (iii) an optimized training procedure and network architecture. We conduct experiments on four datasets to evaluate QINCo2 for vector compression and billion-scale nearest neighbor search. We obtain outstanding results in both settings, improving the state-of-the-art reconstruction MSE by 34% for 16-byte vector compression on BigANN, and search accuracy by 24% with 8-byte encodings on Deep1M.
On Improved Conditioning Mechanisms and Pre-training Strategies for Diffusion Models
Nov 05, 2024Abstract:Large-scale training of latent diffusion models (LDMs) has enabled unprecedented quality in image generation. However, the key components of the best performing LDM training recipes are oftentimes not available to the research community, preventing apple-to-apple comparisons and hindering the validation of progress in the field. In this work, we perform an in-depth study of LDM training recipes focusing on the performance of models and their training efficiency. To ensure apple-to-apple comparisons, we re-implement five previously published models with their corresponding recipes. Through our study, we explore the effects of (i)~the mechanisms used to condition the generative model on semantic information (e.g., text prompt) and control metadata (e.g., crop size, random flip flag, etc.) on the model performance, and (ii)~the transfer of the representations learned on smaller and lower-resolution datasets to larger ones on the training efficiency and model performance. We then propose a novel conditioning mechanism that disentangles semantic and control metadata conditionings and sets a new state-of-the-art in class-conditional generation on the ImageNet-1k dataset -- with FID improvements of 7% on 256 and 8% on 512 resolutions -- as well as text-to-image generation on the CC12M dataset -- with FID improvements of 8% on 256 and 23% on 512 resolution.
Exact Byte-Level Probabilities from Tokenized Language Models for FIM-Tasks and Model Ensembles
Oct 11, 2024



Abstract:Tokenization is associated with many poorly understood shortcomings in language models (LMs), yet remains an important component for long sequence scaling purposes. This work studies how tokenization impacts model performance by analyzing and comparing the stochastic behavior of tokenized models with their byte-level, or token-free, counterparts. We discover that, even when the two models are statistically equivalent, their predictive distributions over the next byte can be substantially different, a phenomenon we term as "tokenization bias''. To fully characterize this phenomenon, we introduce the Byte-Token Representation Lemma, a framework that establishes a mapping between the learned token distribution and its equivalent byte-level distribution. From this result, we develop a next-byte sampling algorithm that eliminates tokenization bias without requiring further training or optimization. In other words, this enables zero-shot conversion of tokenized LMs into statistically equivalent token-free ones. We demonstrate its broad applicability with two use cases: fill-in-the-middle (FIM) tasks and model ensembles. In FIM tasks where input prompts may terminate mid-token, leading to out-of-distribution tokenization, our method mitigates performance degradation and achieves an approximately 18% improvement in FIM coding benchmarks, consistently outperforming the standard token healing fix. For model ensembles where each model employs a distinct vocabulary, our approach enables seamless integration, resulting in improved performance (up to 3.7%) over individual models across various standard baselines in reasoning, knowledge, and coding.
Understanding and Mitigating Tokenization Bias in Language Models
Jun 24, 2024


Abstract:State-of-the-art language models are autoregressive and operate on subword units known as tokens. Specifically, one must encode the conditioning string into a list of tokens before passing to the language models for next-token prediction. We show that, for encoding schemes such as maximum prefix matching, tokenization induces a sampling bias that cannot be mitigated with more training or data. To counter this universal problem, we propose a novel algorithm to obtain unbiased estimates from a model that was trained on tokenized data. Our method does not require finetuning the model, and its complexity, defined as the number of model runs, scales linearly with the sequence length. As a consequence, we show that one can simulate token-free behavior from a tokenized language model. We empirically verify the correctness of our method through a Markov-chain setup, where it accurately recovers the transition probabilities, as opposed to the conventional method of directly prompting tokens into the language model.
Consistency-diversity-realism Pareto fronts of conditional image generative models
Jun 14, 2024



Abstract:Building world models that accurately and comprehensively represent the real world is the utmost aspiration for conditional image generative models as it would enable their use as world simulators. For these models to be successful world models, they should not only excel at image quality and prompt-image consistency but also ensure high representation diversity. However, current research in generative models mostly focuses on creative applications that are predominantly concerned with human preferences of image quality and aesthetics. We note that generative models have inference time mechanisms - or knobs - that allow the control of generation consistency, quality, and diversity. In this paper, we use state-of-the-art text-to-image and image-and-text-to-image models and their knobs to draw consistency-diversity-realism Pareto fronts that provide a holistic view on consistency-diversity-realism multi-objective. Our experiments suggest that realism and consistency can both be improved simultaneously; however there exists a clear tradeoff between realism/consistency and diversity. By looking at Pareto optimal points, we note that earlier models are better at representation diversity and worse in consistency/realism, and more recent models excel in consistency/realism while decreasing significantly the representation diversity. By computing Pareto fronts on a geodiverse dataset, we find that the first version of latent diffusion models tends to perform better than more recent models in all axes of evaluation, and there exist pronounced consistency-diversity-realism disparities between geographical regions. Overall, our analysis clearly shows that there is no best model and the choice of model should be determined by the downstream application. With this analysis, we invite the research community to consider Pareto fronts as an analytical tool to measure progress towards world models.
Advancing human-centric AI for robust X-ray analysis through holistic self-supervised learning
May 02, 2024



Abstract:AI Foundation models are gaining traction in various applications, including medical fields like radiology. However, medical foundation models are often tested on limited tasks, leaving their generalisability and biases unexplored. We present RayDINO, a large visual encoder trained by self-supervision on 873k chest X-rays. We compare RayDINO to previous state-of-the-art models across nine radiology tasks, from classification and dense segmentation to text generation, and provide an in depth analysis of population, age and sex biases of our model. Our findings suggest that self-supervision allows patient-centric AI proving useful in clinical workflows and interpreting X-rays holistically. With RayDINO and small task-specific adapters, we reach state-of-the-art results and improve generalization to unseen populations while mitigating bias, illustrating the true promise of foundation models: versatility and robustness.
Residual Quantization with Implicit Neural Codebooks
Jan 26, 2024



Abstract:Vector quantization is a fundamental operation for data compression and vector search. To obtain high accuracy, multi-codebook methods increase the rate by representing each vector using codewords across multiple codebooks. Residual quantization (RQ) is one such method, which increases accuracy by iteratively quantizing the error of the previous step. The error distribution is dependent on previously selected codewords. This dependency is, however, not accounted for in conventional RQ as it uses a generic codebook per quantization step. In this paper, we propose QINCo, a neural RQ variant which predicts specialized codebooks per vector using a neural network that is conditioned on the approximation of the vector from previous steps. Experiments show that QINCo outperforms state-of-the-art methods by a large margin on several datasets and code sizes. For example, QINCo achieves better nearest-neighbor search accuracy using 12 bytes codes than other methods using 16 bytes on the BigANN and Deep1B dataset.
Latent Discretization for Continuous-time Sequence Compression
Dec 28, 2022



Abstract:Neural compression offers a domain-agnostic approach to creating codecs for lossy or lossless compression via deep generative models. For sequence compression, however, most deep sequence models have costs that scale with the sequence length rather than the sequence complexity. In this work, we instead treat data sequences as observations from an underlying continuous-time process and learn how to efficiently discretize while retaining information about the full sequence. As a consequence of decoupling sequential information from its temporal discretization, our approach allows for greater compression rates and smaller computational complexity. Moreover, the continuous-time approach naturally allows us to decode at different time intervals. We empirically verify our approach on multiple domains involving compression of video and motion capture sequences, showing that our approaches can automatically achieve reductions in bit rates by learning how to discretize.
On learning adaptive acquisition policies for undersampled multi-coil MRI reconstruction
Mar 30, 2022
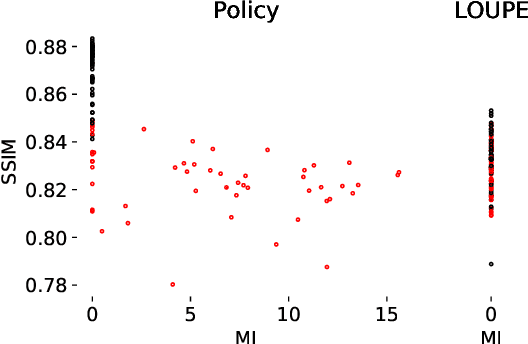
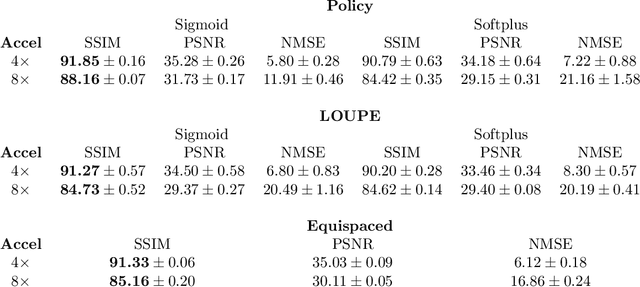
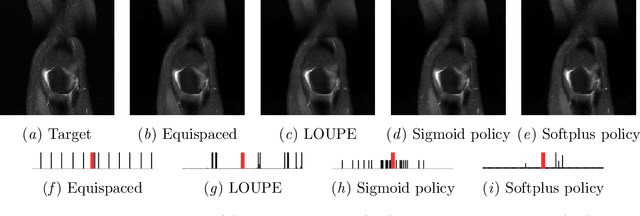
Abstract:Most current approaches to undersampled multi-coil MRI reconstruction focus on learning the reconstruction model for a fixed, equidistant acquisition trajectory. In this paper, we study the problem of joint learning of the reconstruction model together with acquisition policies. To this end, we extend the End-to-End Variational Network with learnable acquisition policies that can adapt to different data points. We validate our model on a coil-compressed version of the large scale undersampled multi-coil fastMRI dataset using two undersampling factors: $4\times$ and $8\times$. Our experiments show on-par performance with the learnable non-adaptive and handcrafted equidistant strategies at $4\times$, and an observed improvement of more than $2\%$ in SSIM at $8\times$ acceleration, suggesting that potentially-adaptive $k$-space acquisition trajectories can improve reconstructed image quality for larger acceleration factors. However, and perhaps surprisingly, our best performing policies learn to be explicitly non-adaptive.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge