Caixia Sun
GeoTikzBridge: Advancing Multimodal Code Generation for Geometric Perception and Reasoning
Mar 24, 2026Abstract:Multimodal Large Language Models (MLLMs) have recently demonstrated remarkable perceptual and reasoning abilities. However, they struggle to perceive fine-grained geometric structures, constraining their ability of geometric understanding and visual reasoning. To address this, we propose GeoTikzBridge, a framework that enhances local geometric perception and visual reasoning through tikz-based code generation. Within this framework, we build two models supported by two complementary datasets. The GeoTikzBridge-Base model is trained on GeoTikz-Base dataset, the largest image-to-tikz dataset to date with 2.5M pairs (16 $\times$ larger than existing open-sourced datasets). This process is achieved via iterative data expansion and a localized geometric transformation strategy. Subsequently, GeoTikzBridge-Instruct is fine-tuned on GeoTikz-Instruct dataset which is the first instruction-augmented tikz dataset supporting visual reasoning. Extensive experimental results demonstrate that our models achieve state-of-the-art performance among open-sourced MLLMs. Furthermore, GeoTikzBridge models can serve as plug-and-play reasoning modules for any MLLM(LLM), enhancing reasoning performance in geometric problem-solving. Datasets and codes are publicly available at: https://github.com/sjy-1995/GeoTikzBridge-Advancing-Multimodal-Code-Generation-for-Geometric-Perception-and-Reasoning.
Convolutional Restricted Boltzmann Machine Based-Radiomics for Prediction of Pathological Complete Response to Neoadjuvant Chemotherapy in Breast Cancer
May 23, 2019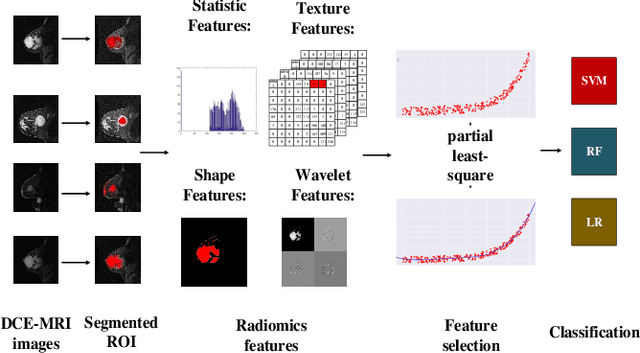
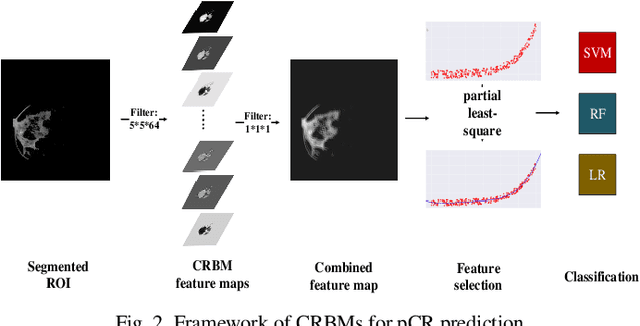
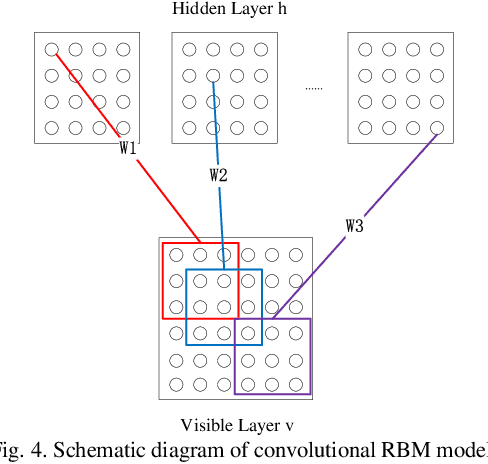
Abstract:We proposed a novel convolutional restricted Boltzmann machine CRBM-based radiomic method for predicting pathologic complete response (pCR) to neoadjuvant chemotherapy treatment (NACT) in breast cancer. The method consists of extracting semantic features from CRBM network, and pCR prediction. It was evaluated on the dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) data of 57 patients and using the area under the receiver operating characteristic curve (AUC). Traditional radiomics features and the semantic features learned from CRBM network were extracted from the images acquired before and after the administration of NACT. After the feature selection, the support vector machine (SVM), logistic regression (LR) and random forest (RF) were trained to predict the pCR status. Compared to traditional radiomic methods, the proposed CRBM-based radiomic method yielded an AUC of 0.92 for the prediction with the images acquired before and after NACT, and an AUC of 0.87 for the pretreatment prediction, which was increased by about 38%. The results showed that the CRBM-based radiomic method provided a potential means for accurately predicting the pCR to NACT in breast cancer before the treatment, which is very useful for making more appropriate and personalized treatment regimens.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge