cancer detection
Cancer detection using Artificial Intelligence (AI) involves leveraging advanced machine learning algorithms and techniques to identify and diagnose cancer from various medical data sources. The goal is to enhance early detection, improve diagnostic accuracy, and potentially reduce the need for invasive procedures.
Papers and Code
Prostate Cancer Screening with Artificial Intelligence-Enhanced Micro-Ultrasound: A Comparative Study with Traditional Methods
May 27, 2025Background and objective: Micro-ultrasound (micro-US) is a novel imaging modality with diagnostic accuracy comparable to MRI for detecting clinically significant prostate cancer (csPCa). We investigated whether artificial intelligence (AI) interpretation of micro-US can outperform clinical screening methods using PSA and digital rectal examination (DRE). Methods: We retrospectively studied 145 men who underwent micro-US guided biopsy (79 with csPCa, 66 without). A self-supervised convolutional autoencoder was used to extract deep image features from 2D micro-US slices. Random forest classifiers were trained using five-fold cross-validation to predict csPCa at the slice level. Patients were classified as csPCa-positive if 88 or more consecutive slices were predicted positive. Model performance was compared with a classifier using PSA, DRE, prostate volume, and age. Key findings and limitations: The AI-based micro-US model and clinical screening model achieved AUROCs of 0.871 and 0.753, respectively. At a fixed threshold, the micro-US model achieved 92.5% sensitivity and 68.1% specificity, while the clinical model showed 96.2% sensitivity but only 27.3% specificity. Limitations include a retrospective single-center design and lack of external validation. Conclusions and clinical implications: AI-interpreted micro-US improves specificity while maintaining high sensitivity for csPCa detection. This method may reduce unnecessary biopsies and serve as a low-cost alternative to PSA-based screening. Patient summary: We developed an AI system to analyze prostate micro-ultrasound images. It outperformed PSA and DRE in detecting aggressive cancer and may help avoid unnecessary biopsies.
Dynamic Contextual Attention Network: Transforming Spatial Representations into Adaptive Insights for Endoscopic Polyp Diagnosis
Apr 28, 2025



Colorectal polyps are key indicators for early detection of colorectal cancer. However, traditional endoscopic imaging often struggles with accurate polyp localization and lacks comprehensive contextual awareness, which can limit the explainability of diagnoses. To address these issues, we propose the Dynamic Contextual Attention Network (DCAN). This novel approach transforms spatial representations into adaptive contextual insights, using an attention mechanism that enhances focus on critical polyp regions without explicit localization modules. By integrating contextual awareness into the classification process, DCAN improves decision interpretability and overall diagnostic performance. This advancement in imaging could lead to more reliable colorectal cancer detection, enabling better patient outcomes.
Mitigating Catastrophic Forgetting in the Incremental Learning of Medical Images
Apr 28, 2025This paper proposes an Incremental Learning (IL) approach to enhance the accuracy and efficiency of deep learning models in analyzing T2-weighted (T2w) MRI medical images prostate cancer detection using the PI-CAI dataset. We used multiple health centers' artificial intelligence and radiology data, focused on different tasks that looked at prostate cancer detection using MRI (PI-CAI). We utilized Knowledge Distillation (KD), as it employs generated images from past tasks to guide the training of models for subsequent tasks. The approach yielded improved performance and faster convergence of the models. To demonstrate the versatility and robustness of our approach, we evaluated it on the PI-CAI dataset, a diverse set of medical imaging modalities including OCT and PathMNIST, and the benchmark continual learning dataset CIFAR-10. Our results indicate that KD can be a promising technique for IL in medical image analysis in which data is sourced from individual health centers and the storage of large datasets is not feasible. By using generated images from prior tasks, our method enables the model to retain and apply previously acquired knowledge without direct access to the original data.
Advanced cervical cancer classification: enhancing pap smear images with hybrid PMD Filter-CLAHE
Jun 18, 2025Cervical cancer remains a significant health problem, especially in developing countries. Early detection is critical for effective treatment. Convolutional neural networks (CNN) have shown promise in automated cervical cancer screening, but their performance depends on Pap smear image quality. This study investigates the impact of various image preprocessing techniques on CNN performance for cervical cancer classification using the SIPaKMeD dataset. Three preprocessing techniques were evaluated: perona-malik diffusion (PMD) filter for noise reduction, contrast-limited adaptive histogram equalization (CLAHE) for image contrast enhancement, and the proposed hybrid PMD filter-CLAHE approach. The enhanced image datasets were evaluated on pretrained models, such as ResNet-34, ResNet-50, SqueezeNet-1.0, MobileNet-V2, EfficientNet-B0, EfficientNet-B1, DenseNet-121, and DenseNet-201. The results show that hybrid preprocessing PMD filter-CLAHE can improve the Pap smear image quality and CNN architecture performance compared to the original images. The maximum metric improvements are 13.62% for accuracy, 10.04% for precision, 13.08% for recall, and 14.34% for F1-score. The proposed hybrid PMD filter-CLAHE technique offers a new perspective in improving cervical cancer classification performance using CNN architectures.
Vision-Language Model-Based Semantic-Guided Imaging Biomarker for Early Lung Cancer Detection
Apr 30, 2025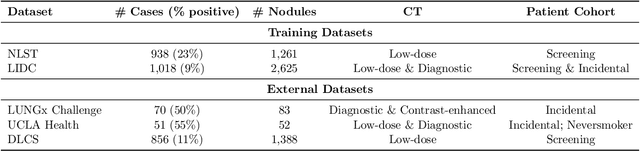
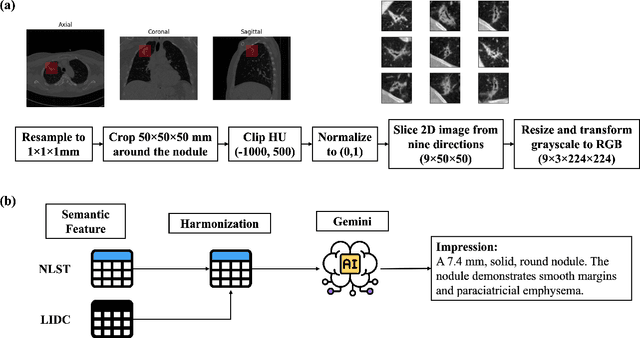
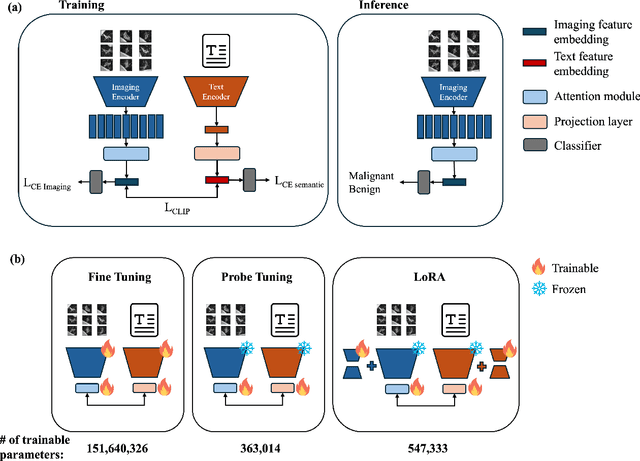
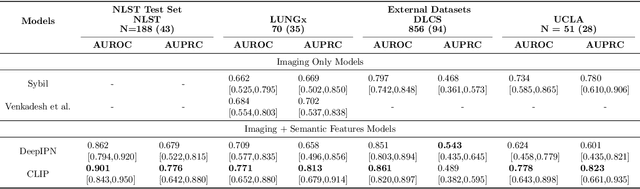
Objective: A number of machine learning models have utilized semantic features, deep features, or both to assess lung nodule malignancy. However, their reliance on manual annotation during inference, limited interpretability, and sensitivity to imaging variations hinder their application in real-world clinical settings. Thus, this research aims to integrate semantic features derived from radiologists' assessments of nodules, allowing the model to learn clinically relevant, robust, and explainable features for predicting lung cancer. Methods: We obtained 938 low-dose CT scans from the National Lung Screening Trial with 1,246 nodules and semantic features. The Lung Image Database Consortium dataset contains 1,018 CT scans, with 2,625 lesions annotated for nodule characteristics. Three external datasets were obtained from UCLA Health, the LUNGx Challenge, and the Duke Lung Cancer Screening. We finetuned a pretrained Contrastive Language-Image Pretraining model with a parameter-efficient fine-tuning approach to align imaging and semantic features and predict the one-year lung cancer diagnosis. Results: We evaluated the performance of the one-year diagnosis of lung cancer with AUROC and AUPRC and compared it to three state-of-the-art models. Our model demonstrated an AUROC of 0.90 and AUPRC of 0.78, outperforming baseline state-of-the-art models on external datasets. Using CLIP, we also obtained predictions on semantic features, such as nodule margin (AUROC: 0.81), nodule consistency (0.81), and pleural attachment (0.84), that can be used to explain model predictions. Conclusion: Our approach accurately classifies lung nodules as benign or malignant, providing explainable outputs, aiding clinicians in comprehending the underlying meaning of model predictions. This approach also prevents the model from learning shortcuts and generalizes across clinical settings.
Lightweight Relational Embedding in Task-Interpolated Few-Shot Networks for Enhanced Gastrointestinal Disease Classification
May 30, 2025Traditional diagnostic methods like colonoscopy are invasive yet critical tools necessary for accurately diagnosing colorectal cancer (CRC). Detection of CRC at early stages is crucial for increasing patient survival rates. However, colonoscopy is dependent on obtaining adequate and high-quality endoscopic images. Prolonged invasive procedures are inherently risky for patients, while suboptimal or insufficient images hamper diagnostic accuracy. These images, typically derived from video frames, often exhibit similar patterns, posing challenges in discrimination. To overcome these challenges, we propose a novel Deep Learning network built on a Few-Shot Learning architecture, which includes a tailored feature extractor, task interpolation, relational embedding, and a bi-level routing attention mechanism. The Few-Shot Learning paradigm enables our model to rapidly adapt to unseen fine-grained endoscopic image patterns, and the task interpolation augments the insufficient images artificially from varied instrument viewpoints. Our relational embedding approach discerns critical intra-image features and captures inter-image transitions between consecutive endoscopic frames, overcoming the limitations of Convolutional Neural Networks (CNNs). The integration of a light-weight attention mechanism ensures a concentrated analysis of pertinent image regions. By training on diverse datasets, the model's generalizability and robustness are notably improved for handling endoscopic images. Evaluated on Kvasir dataset, our model demonstrated superior performance, achieving an accuracy of 90.1\%, precision of 0.845, recall of 0.942, and an F1 score of 0.891. This surpasses current state-of-the-art methods, presenting a promising solution to the challenges of invasive colonoscopy by optimizing CRC detection through advanced image analysis.
* 6 pages, 15 figures
MAMBO: High-Resolution Generative Approach for Mammography Images
Jun 10, 2025Mammography is the gold standard for the detection and diagnosis of breast cancer. This procedure can be significantly enhanced with Artificial Intelligence (AI)-based software, which assists radiologists in identifying abnormalities. However, training AI systems requires large and diverse datasets, which are often difficult to obtain due to privacy and ethical constraints. To address this issue, the paper introduces MAMmography ensemBle mOdel (MAMBO), a novel patch-based diffusion approach designed to generate full-resolution mammograms. Diffusion models have shown breakthrough results in realistic image generation, yet few studies have focused on mammograms, and none have successfully generated high-resolution outputs required to capture fine-grained features of small lesions. To achieve this, MAMBO integrates separate diffusion models to capture both local and global (image-level) contexts. The contextual information is then fed into the final patch-based model, significantly aiding the noise removal process. This thoughtful design enables MAMBO to generate highly realistic mammograms of up to 3840x3840 pixels. Importantly, this approach can be used to enhance the training of classification models and extended to anomaly detection. Experiments, both numerical and radiologist validation, assess MAMBO's capabilities in image generation, super-resolution, and anomaly detection, highlighting its potential to enhance mammography analysis for more accurate diagnoses and earlier lesion detection.
White Light Specular Reflection Data Augmentation for Deep Learning Polyp Detection
May 08, 2025Colorectal cancer is one of the deadliest cancers today, but it can be prevented through early detection of malignant polyps in the colon, primarily via colonoscopies. While this method has saved many lives, human error remains a significant challenge, as missing a polyp could have fatal consequences for the patient. Deep learning (DL) polyp detectors offer a promising solution. However, existing DL polyp detectors often mistake white light reflections from the endoscope for polyps, which can lead to false positives.To address this challenge, in this paper, we propose a novel data augmentation approach that artificially adds more white light reflections to create harder training scenarios. Specifically, we first generate a bank of artificial lights using the training dataset. Then we find the regions of the training images that we should not add these artificial lights on. Finally, we propose a sliding window method to add the artificial light to the areas that fit of the training images, resulting in augmented images. By providing the model with more opportunities to make mistakes, we hypothesize that it will also have more chances to learn from those mistakes, ultimately improving its performance in polyp detection. Experimental results demonstrate the effectiveness of our new data augmentation method.
BreastDCEDL: Curating a Comprehensive DCE-MRI Dataset and developing a Transformer Implementation for Breast Cancer Treatment Response Prediction
Jun 13, 2025Breast cancer remains a leading cause of cancer-related mortality worldwide, making early detection and accurate treatment response monitoring critical priorities. We present BreastDCEDL, a curated, deep learning-ready dataset comprising pre-treatment 3D Dynamic Contrast-Enhanced MRI (DCE-MRI) scans from 2,070 breast cancer patients drawn from the I-SPY1, I-SPY2, and Duke cohorts, all sourced from The Cancer Imaging Archive. The raw DICOM imaging data were rigorously converted into standardized 3D NIfTI volumes with preserved signal integrity, accompanied by unified tumor annotations and harmonized clinical metadata including pathologic complete response (pCR), hormone receptor (HR), and HER2 status. Although DCE-MRI provides essential diagnostic information and deep learning offers tremendous potential for analyzing such complex data, progress has been limited by lack of accessible, public, multicenter datasets. BreastDCEDL addresses this gap by enabling development of advanced models, including state-of-the-art transformer architectures that require substantial training data. To demonstrate its capacity for robust modeling, we developed the first transformer-based model for breast DCE-MRI, leveraging Vision Transformer (ViT) architecture trained on RGB-fused images from three contrast phases (pre-contrast, early post-contrast, and late post-contrast). Our ViT model achieved state-of-the-art pCR prediction performance in HR+/HER2- patients (AUC 0.94, accuracy 0.93). BreastDCEDL includes predefined benchmark splits, offering a framework for reproducible research and enabling clinically meaningful modeling in breast cancer imaging.
Towards Facilitated Fairness Assessment of AI-based Skin Lesion Classifiers Through GenAI-based Image Synthesis
Jul 23, 2025Recent advancements in Deep Learning and its application on the edge hold great potential for the revolution of routine screenings for skin cancers like Melanoma. Along with the anticipated benefits of this technology, potential dangers arise from unforseen and inherent biases. Thus, assessing and improving the fairness of such systems is of utmost importance. A key challenge in fairness assessment is to ensure that the evaluation dataset is sufficiently representative of different Personal Identifiable Information (PII) (sex, age, and race) and other minority groups. Against the backdrop of this challenge, this study leverages the state-of-the-art Generative AI (GenAI) LightningDiT model to assess the fairness of publicly available melanoma classifiers. The results suggest that fairness assessment using highly realistic synthetic data is a promising direction. Yet, our findings indicate that verifying fairness becomes difficult when the melanoma-detection model used for evaluation is trained on data that differ from the dataset underpinning the synthetic images. Nonetheless, we propose that our approach offers a valuable new avenue for employing synthetic data to gauge and enhance fairness in medical-imaging GenAI systems.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge