Sanjay Kumar
FLUX: Data Worth Training On
Mar 14, 2026Abstract:Modern large language model training is no longer limited by data availability, but by the inability of existing preprocessing pipelines to simultaneously achieve massive scale and high data quality. Current approaches are forced to sacrifice one for the other: either aggressively filtering to improve quality at the cost of severe token loss, or retaining large volumes of data while introducing substantial noise. In this work, we introduce FLUX, a preprocessing pipeline specifically designed to break this long-standing trade-off by maximizing token retention while enforcing rigorous quality control. Models trained on FLUX-curated data consistently outperform prior methods. A 3B-parameter model trained on 60B tokens with FLUX achieves 32.14% MMLU accuracy, surpassing the previous state-of-the-art pipeline DCLM (31.98%) and significantly outperforming FineWeb (29.88%). FLUX achieves the same aggregate score as a model trained on DCLM data using only 39B tokens, resulting in a 34.4% reduction in training compute. At the data level, FLUX extracts 50B usable tokens from a single dump (CC-MAIN-2025-51), compared to 40B from DCLM (+25% retention). FLUX-Base yields 192B tokens, exceeding FineWeb's 170B while still maintaining superior quality. Overall, FLUX establishes a new state of the art in web-scale data preprocessing by demonstrating that high retention, strong quality control, and computational efficiency can be achieved simultaneously, redefining the limits of scalable dataset construction for modern language models.
Evaluating the Impact of Weather-Induced Sensor Occlusion on BEVFusion for 3D Object Detection
Nov 06, 2025Abstract:Accurate 3D object detection is essential for automated vehicles to navigate safely in complex real-world environments. Bird's Eye View (BEV) representations, which project multi-sensor data into a top-down spatial format, have emerged as a powerful approach for robust perception. Although BEV-based fusion architectures have demonstrated strong performance through multimodal integration, the effects of sensor occlusions, caused by environmental conditions such as fog, haze, or physical obstructions, on 3D detection accuracy remain underexplored. In this work, we investigate the impact of occlusions on both camera and Light Detection and Ranging (LiDAR) outputs using the BEVFusion architecture, evaluated on the nuScenes dataset. Detection performance is measured using mean Average Precision (mAP) and the nuScenes Detection Score (NDS). Our results show that moderate camera occlusions lead to a 41.3% drop in mAP (from 35.6% to 20.9%) when detection is based only on the camera. On the other hand, LiDAR sharply drops in performance only under heavy occlusion, with mAP falling by 47.3% (from 64.7% to 34.1%), with a severe impact on long-range detection. In fused settings, the effect depends on which sensor is occluded: occluding the camera leads to a minor 4.1% drop (from 68.5% to 65.7%), while occluding LiDAR results in a larger 26.8% drop (to 50.1%), revealing the model's stronger reliance on LiDAR for the task of 3D object detection. Our results highlight the need for future research into occlusion-aware evaluation methods and improved sensor fusion techniques that can maintain detection accuracy in the presence of partial sensor failure or degradation due to adverse environmental conditions.
GBSVR: Granular Ball Support Vector Regression
Mar 13, 2025



Abstract:Support Vector Regression (SVR) and its variants are widely used to handle regression tasks, however, since their solution involves solving an expensive quadratic programming problem, it limits its application, especially when dealing with large datasets. Additionally, SVR uses an epsilon-insensitive loss function which is sensitive to outliers and therefore can adversely affect its performance. We propose Granular Ball Support Vector Regression (GBSVR) to tackle problem of regression by using granular ball concept. These balls are useful in simplifying complex data spaces for machine learning tasks, however, to the best of our knowledge, they have not been sufficiently explored for regression problems. Granular balls group the data points into balls based on their proximity and reduce the computational cost in SVR by replacing the large number of data points with far fewer granular balls. This work also suggests a discretization method for continuous-valued attributes to facilitate the construction of granular balls. The effectiveness of the proposed approach is evaluated on several benchmark datasets and it outperforms existing state-of-the-art approaches
Minimizing Occlusion Effect on Multi-View Camera Perception in BEV with Multi-Sensor Fusion
Jan 10, 2025Abstract:Autonomous driving technology is rapidly evolving, offering the potential for safer and more efficient transportation. However, the performance of these systems can be significantly compromised by the occlusion on sensors due to environmental factors like dirt, dust, rain, and fog. These occlusions severely affect vision-based tasks such as object detection, vehicle segmentation, and lane recognition. In this paper, we investigate the impact of various kinds of occlusions on camera sensor by projecting their effects from multi-view camera images of the nuScenes dataset into the Bird's-Eye View (BEV) domain. This approach allows us to analyze how occlusions spatially distribute and influence vehicle segmentation accuracy within the BEV domain. Despite significant advances in sensor technology and multi-sensor fusion, a gap remains in the existing literature regarding the specific effects of camera occlusions on BEV-based perception systems. To address this gap, we use a multi-sensor fusion technique that integrates LiDAR and radar sensor data to mitigate the performance degradation caused by occluded cameras. Our findings demonstrate that this approach significantly enhances the accuracy and robustness of vehicle segmentation tasks, leading to more reliable autonomous driving systems.
Maximum Solar Energy Tracking Leverage High-DoF Robotics System with Deep Reinforcement Learning
Nov 21, 2024



Abstract:Solar trajectory monitoring is a pivotal challenge in solar energy systems, underpinning applications such as autonomous energy harvesting and environmental sensing. A prevalent failure mode in sustained solar tracking arises when the predictive algorithm erroneously diverges from the solar locus, erroneously anchoring to extraneous celestial or terrestrial features. This phenomenon is attributable to an inadequate assimilation of solar-specific objectness attributes within the tracking paradigm. To mitigate this deficiency inherent in extant methodologies, we introduce an innovative objectness regularization framework that compels tracking points to remain confined within the delineated boundaries of the solar entity. By encapsulating solar objectness indicators during the training phase, our approach obviates the necessity for explicit solar mask computation during operational deployment. Furthermore, we leverage the high-DoF robot arm to integrate our method to improve its robustness and flexibility in different outdoor environments.
Generative Artificial Intelligence: A Systematic Review and Applications
May 17, 2024



Abstract:In recent years, the study of artificial intelligence (AI) has undergone a paradigm shift. This has been propelled by the groundbreaking capabilities of generative models both in supervised and unsupervised learning scenarios. Generative AI has shown state-of-the-art performance in solving perplexing real-world conundrums in fields such as image translation, medical diagnostics, textual imagery fusion, natural language processing, and beyond. This paper documents the systematic review and analysis of recent advancements and techniques in Generative AI with a detailed discussion of their applications including application-specific models. Indeed, the major impact that generative AI has made to date, has been in language generation with the development of large language models, in the field of image translation and several other interdisciplinary applications of generative AI. Moreover, the primary contribution of this paper lies in its coherent synthesis of the latest advancements in these areas, seamlessly weaving together contemporary breakthroughs in the field. Particularly, how it shares an exploration of the future trajectory for generative AI. In conclusion, the paper ends with a discussion of Responsible AI principles, and the necessary ethical considerations for the sustainability and growth of these generative models.
Classification of White Blood Cells Using Machine and Deep Learning Models: A Systematic Review
Aug 21, 2023Abstract:Machine learning (ML) and deep learning (DL) models have been employed to significantly improve analyses of medical imagery, with these approaches used to enhance the accuracy of prediction and classification. Model predictions and classifications assist diagnoses of various cancers and tumors. This review presents an in-depth analysis of modern techniques applied within the domain of medical image analysis for white blood cell classification. The methodologies that use blood smear images, magnetic resonance imaging (MRI), X-rays, and similar medical imaging domains are identified and discussed, with a detailed analysis of ML/DL techniques applied to the classification of white blood cells (WBCs) representing the primary focus of the review. The data utilized in this research has been extracted from a collection of 136 primary papers that were published between the years 2006 and 2023. The most widely used techniques and best-performing white blood cell classification methods are identified. While the use of ML and DL for white blood cell classification has concurrently increased and improved in recent year, significant challenges remain - 1) Availability of appropriate datasets remain the primary challenge, and may be resolved using data augmentation techniques. 2) Medical training of researchers is recommended to improve current understanding of white blood cell structure and subsequent selection of appropriate classification models. 3) Advanced DL networks including Generative Adversarial Networks, R-CNN, Fast R-CNN, and faster R-CNN will likely be increasingly employed to supplement or replace current techniques.
Automatic Classification of Blood Cell Images Using Convolutional Neural Network
Aug 21, 2023Abstract:Human blood primarily comprises plasma, red blood cells, white blood cells, and platelets. It plays a vital role in transporting nutrients to different organs, where it stores essential health-related data about the human body. Blood cells are utilized to defend the body against diverse infections, including fungi, viruses, and bacteria. Hence, blood analysis can help physicians assess an individual's physiological condition. Blood cells have been sub-classified into eight groups: Neutrophils, eosinophils, basophils, lymphocytes, monocytes, immature granulocytes (promyelocytes, myelocytes, and metamyelocytes), erythroblasts, and platelets or thrombocytes on the basis of their nucleus, shape, and cytoplasm. Traditionally, pathologists and hematologists in laboratories have examined these blood cells using a microscope before manually classifying them. The manual approach is slower and more prone to human error. Therefore, it is essential to automate this process. In our paper, transfer learning with CNN pre-trained models. VGG16, VGG19, ResNet-50, ResNet-101, ResNet-152, InceptionV3, MobileNetV2, and DenseNet-20 applied to the PBC dataset's normal DIB. The overall accuracy achieved with these models lies between 91.375 and 94.72%. Hence, inspired by these pre-trained architectures, a model has been proposed to automatically classify the ten types of blood cells with increased accuracy. A novel CNN-based framework has been presented to improve accuracy. The proposed CNN model has been tested on the PBC dataset normal DIB. The outcomes of the experiments demonstrate that our CNN-based framework designed for blood cell classification attains an accuracy of 99.91% on the PBC dataset. Our proposed convolutional neural network model performs competitively when compared to earlier results reported in the literature.
Design and Evaluation of Reconfigurable Intelligent Surfaces in Real-World Environment
Sep 16, 2021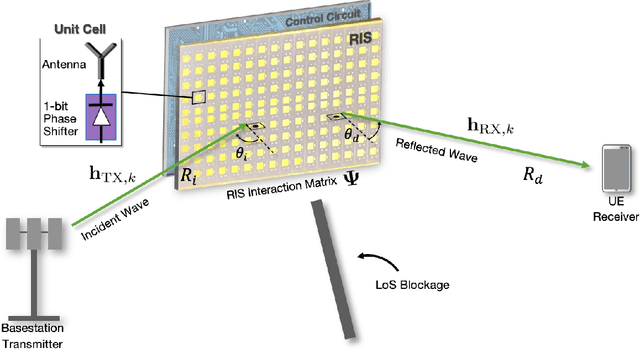
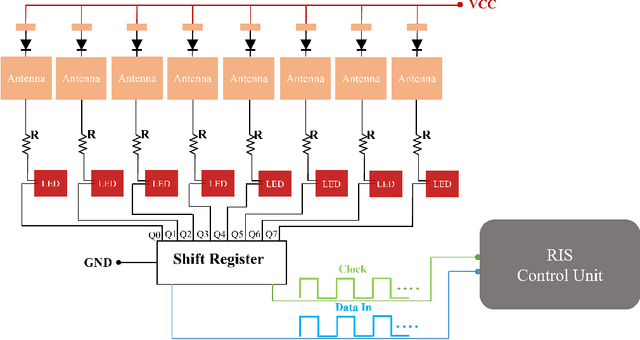

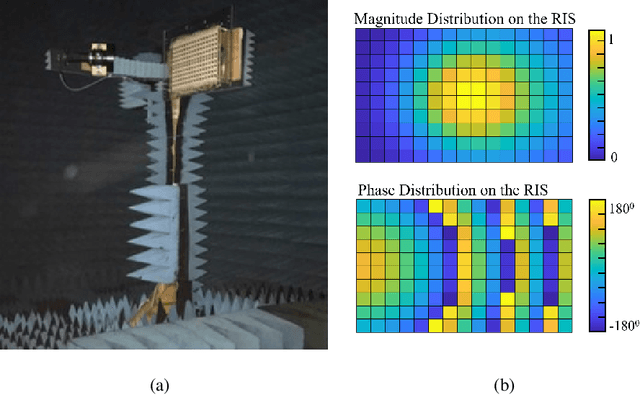
Abstract:Reconfigurable intelligent surfaces (RISs) have promising coverage and data rate gains for wireless communication systems in 5G and beyond. Prior work has mainly focused on analyzing the performance of these surfaces using computer simulations or lab-level prototypes. To draw accurate insights about the actual performance of these systems, this paper develops an RIS proof-of-concept prototype and extensively evaluates its potential gains in the field and under realistic wireless communication settings. In particular, a 160-element reconfigurable surface, operating at a 5.8GHz band, is first designed, fabricated, and accurately measured in the anechoic chamber. This surface is then integrated into a wireless communication system and the beamforming gains, path-loss, and coverage improvements are evaluated in realistic outdoor communication scenarios. When both the transmitter and receiver employ directional antennas and with 5m and 10m distances between the transmitter-RIS and RIS-receiver, the developed RIS achieves $15$-$20$dB gain in the signal-to-noise ratio (SNR) in a range of $\pm60^\circ$ beamforming angles. In terms of coverage, and considering a far-field experiment with a blockage between a base station and a grid of mobile users and with an average distance of $35m$ between base station (BS) and the user (through the RIS), the RIS provides an average SNR improvement of $6$dB (max $8$dB) within an area $> 75$m$^2$. Thanks to the scalable RIS design, these SNR gains can be directly increased with larger RIS areas. For example, a 1,600-element RIS with the same design is expected to provide around $26$dB SNR gain for a similar deployment. These results, among others, draw useful insights into the design and performance of RIS systems and provide an important proof for their potential gains in real-world far-field wireless communication environments.
Investigation of lightweight acoustic curtains for mid-to-high frequency noise insulations
Aug 16, 2021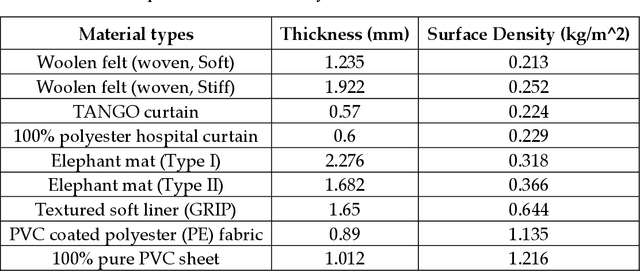

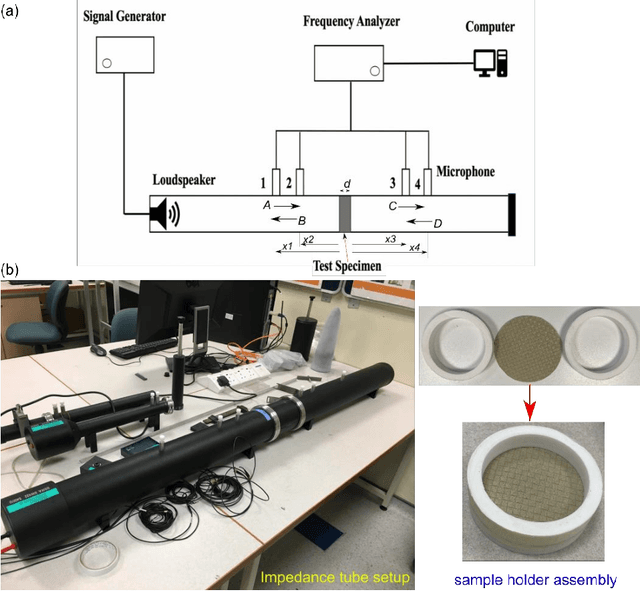
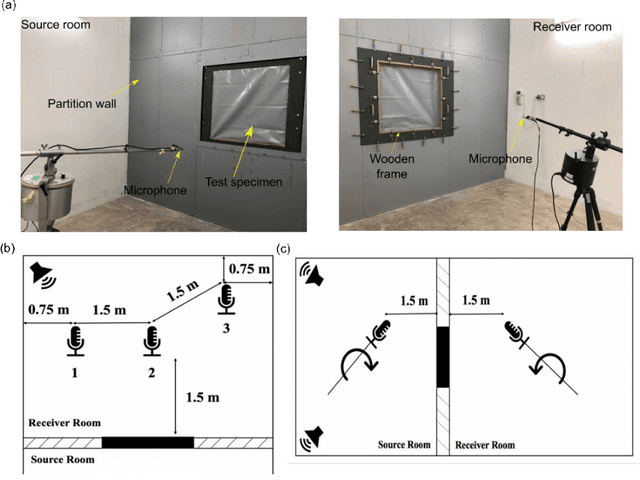
Abstract:The continuous surge of environmental noise levels has become a vital challenge for humanity. Earlier studies have reported that prolonged exposure to loud noise may cause auditory and non-auditory disorders. Therefore, there is a growing demand for suitable noise barriers. Herein, we have investigated several commercially available curtain fabrics' acoustic performance, potentially used for sound insulation purposes. Thorough experimental investigations have been performed on PVC coated polyester fabrics' acoustical performances and 100 % pure PVC sheets. The PVC-coated polyester fabric exhibited better sound insulation properties, particularly in the mid-to-high frequency range (600-1600 Hz) with a transmission loss of about 11 to 22 dB, while insertion loss of > 10 dB has been achieved. Also, the acoustic performance of multi-layer curtains has been investigated. These multi-layer curtains have shown superior acoustic properties to that of single-layer acoustic curtains.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge