Georges El Fakhri
Self-semantic contour adaptation for cross modality brain tumor segmentation
Jan 13, 2022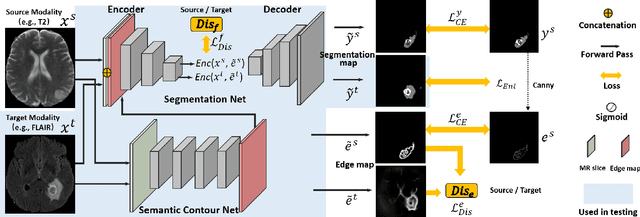
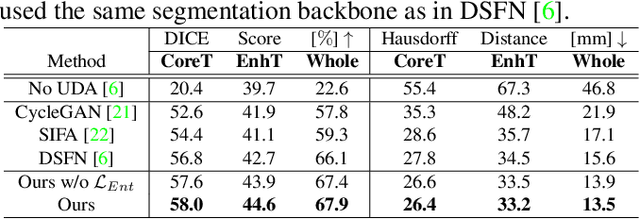
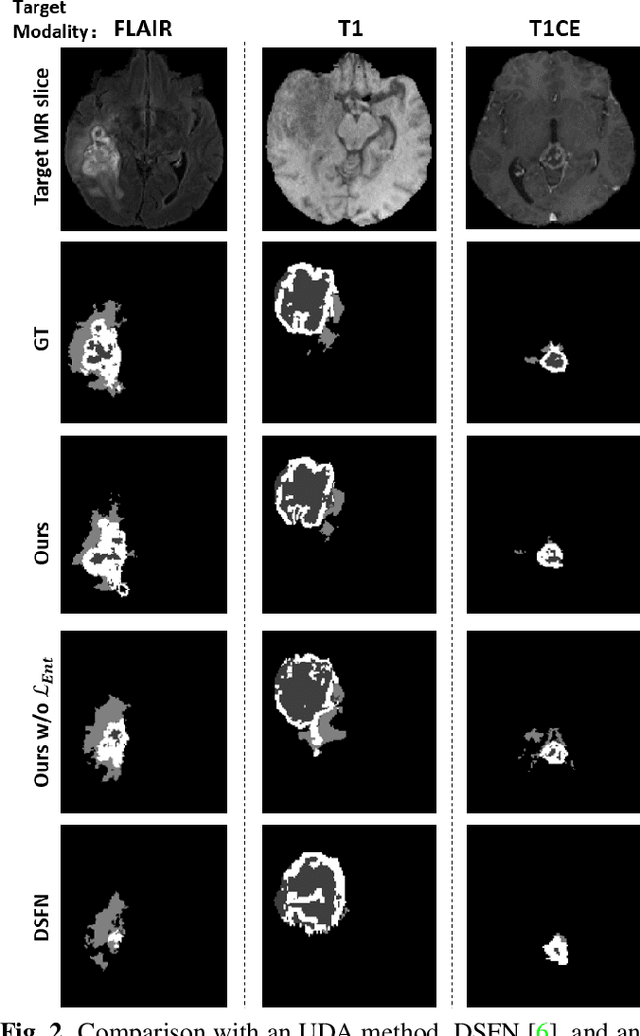
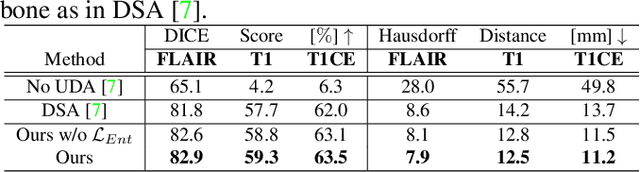
Abstract:Unsupervised domain adaptation (UDA) between two significantly disparate domains to learn high-level semantic alignment is a crucial yet challenging task.~To this end, in this work, we propose exploiting low-level edge information to facilitate the adaptation as a precursor task, which has a small cross-domain gap, compared with semantic segmentation.~The precise contour then provides spatial information to guide the semantic adaptation. More specifically, we propose a multi-task framework to learn a contouring adaptation network along with a semantic segmentation adaptation network, which takes both magnetic resonance imaging (MRI) slice and its initial edge map as input.~These two networks are jointly trained with source domain labels, and the feature and edge map level adversarial learning is carried out for cross-domain alignment. In addition, self-entropy minimization is incorporated to further enhance segmentation performance. We evaluated our framework on the BraTS2018 database for cross-modality segmentation of brain tumors, showing the validity and superiority of our approach, compared with competing methods.
Adversarial Unsupervised Domain Adaptation with Conditional and Label Shift: Infer, Align and Iterate
Aug 02, 2021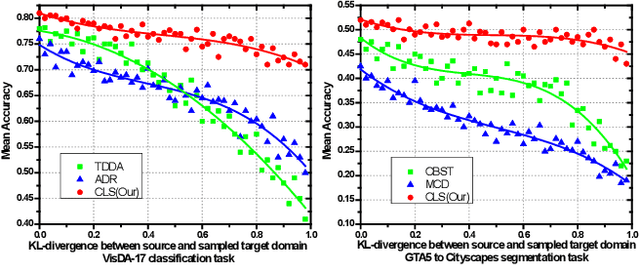
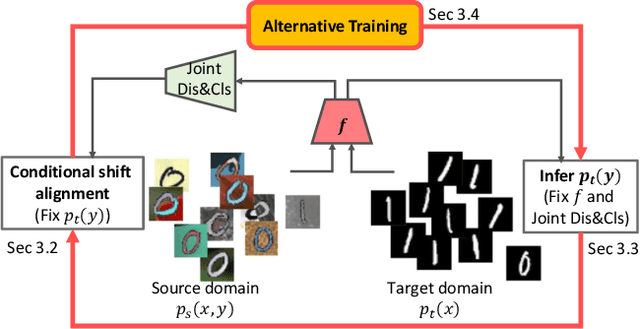
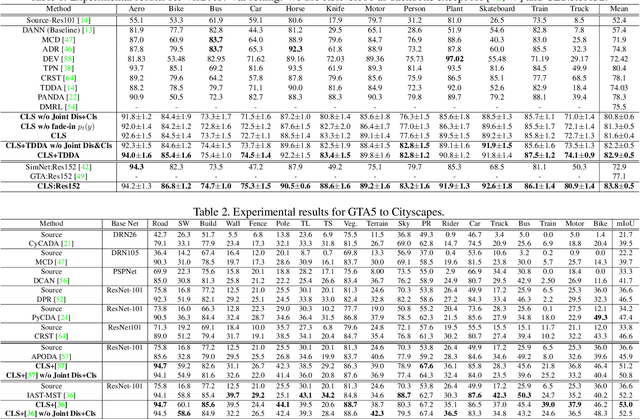
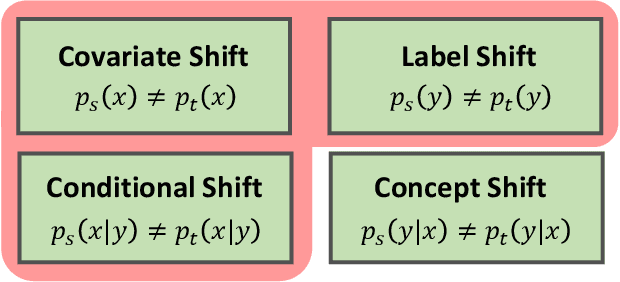
Abstract:In this work, we propose an adversarial unsupervised domain adaptation (UDA) approach with the inherent conditional and label shifts, in which we aim to align the distributions w.r.t. both $p(x|y)$ and $p(y)$. Since the label is inaccessible in the target domain, the conventional adversarial UDA assumes $p(y)$ is invariant across domains, and relies on aligning $p(x)$ as an alternative to the $p(x|y)$ alignment. To address this, we provide a thorough theoretical and empirical analysis of the conventional adversarial UDA methods under both conditional and label shifts, and propose a novel and practical alternative optimization scheme for adversarial UDA. Specifically, we infer the marginal $p(y)$ and align $p(x|y)$ iteratively in the training, and precisely align the posterior $p(y|x)$ in testing. Our experimental results demonstrate its effectiveness on both classification and segmentation UDA, and partial UDA.
Segmentation of Cardiac Structures via Successive Subspace Learning with Saab Transform from Cine MRI
Jul 22, 2021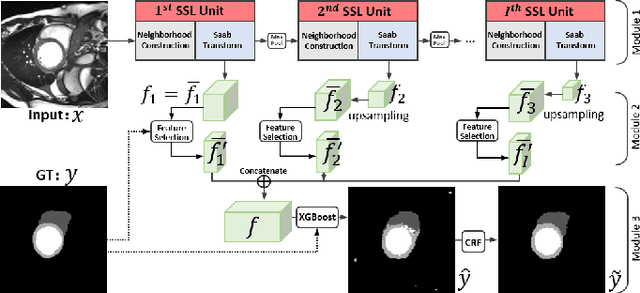

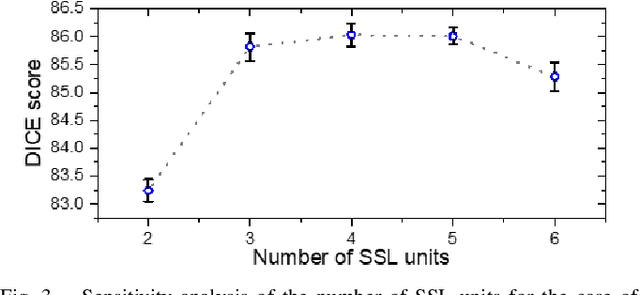

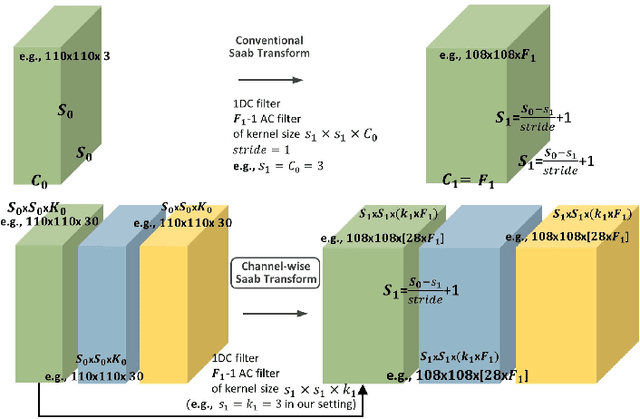
Abstract:Assessment of cardiovascular disease (CVD) with cine magnetic resonance imaging (MRI) has been used to non-invasively evaluate detailed cardiac structure and function. Accurate segmentation of cardiac structures from cine MRI is a crucial step for early diagnosis and prognosis of CVD, and has been greatly improved with convolutional neural networks (CNN). There, however, are a number of limitations identified in CNN models, such as limited interpretability and high complexity, thus limiting their use in clinical practice. In this work, to address the limitations, we propose a lightweight and interpretable machine learning model, successive subspace learning with the subspace approximation with adjusted bias (Saab) transform, for accurate and efficient segmentation from cine MRI. Specifically, our segmentation framework is comprised of the following steps: (1) sequential expansion of near-to-far neighborhood at different resolutions; (2) channel-wise subspace approximation using the Saab transform for unsupervised dimension reduction; (3) class-wise entropy guided feature selection for supervised dimension reduction; (4) concatenation of features and pixel-wise classification with gradient boost; and (5) conditional random field for post-processing. Experimental results on the ACDC 2017 segmentation database, showed that our framework performed better than state-of-the-art U-Net models with 200$\times$ fewer parameters in delineating the left ventricle, right ventricle, and myocardium, thus showing its potential to be used in clinical practice.
Generative Self-training for Cross-domain Unsupervised Tagged-to-Cine MRI Synthesis
Jun 23, 2021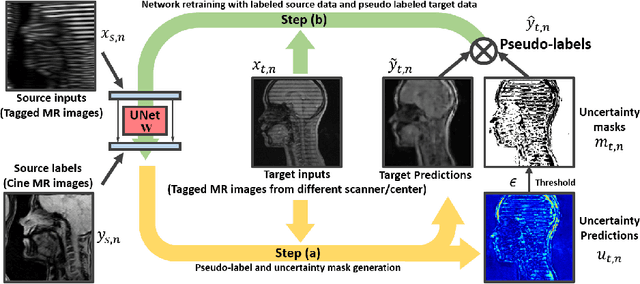
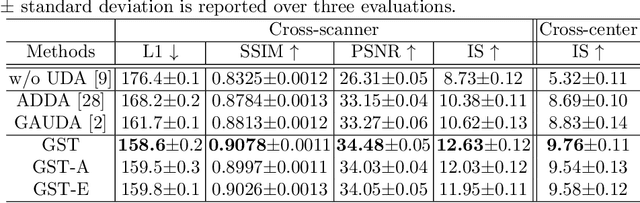
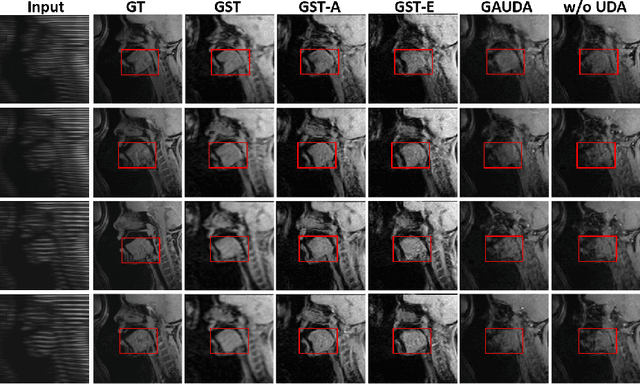
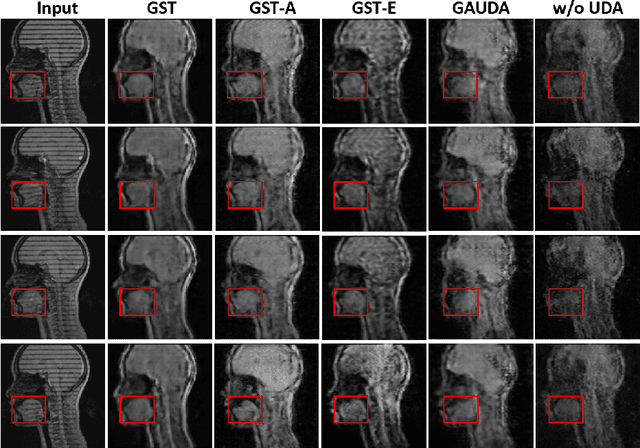
Abstract:Self-training based unsupervised domain adaptation (UDA) has shown great potential to address the problem of domain shift, when applying a trained deep learning model in a source domain to unlabeled target domains. However, while the self-training UDA has demonstrated its effectiveness on discriminative tasks, such as classification and segmentation, via the reliable pseudo-label selection based on the softmax discrete histogram, the self-training UDA for generative tasks, such as image synthesis, is not fully investigated. In this work, we propose a novel generative self-training (GST) UDA framework with continuous value prediction and regression objective for cross-domain image synthesis. Specifically, we propose to filter the pseudo-label with an uncertainty mask, and quantify the predictive confidence of generated images with practical variational Bayes learning. The fast test-time adaptation is achieved by a round-based alternative optimization scheme. We validated our framework on the tagged-to-cine magnetic resonance imaging (MRI) synthesis problem, where datasets in the source and target domains were acquired from different scanners or centers. Extensive validations were carried out to verify our framework against popular adversarial training UDA methods. Results show that our GST, with tagged MRI of test subjects in new target domains, improved the synthesis quality by a large margin, compared with the adversarial training UDA methods.
Adapting Off-the-Shelf Source Segmenter for Target Medical Image Segmentation
Jun 23, 2021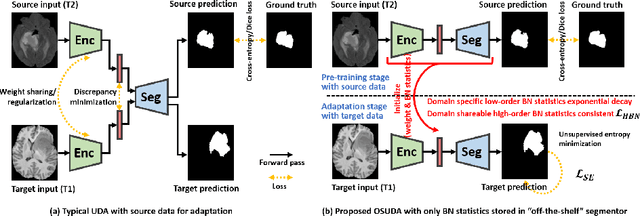
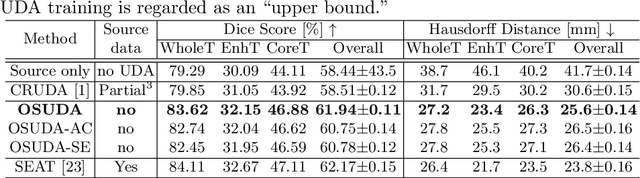
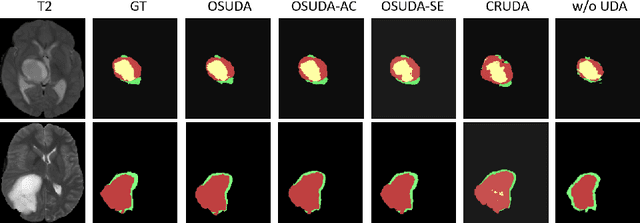
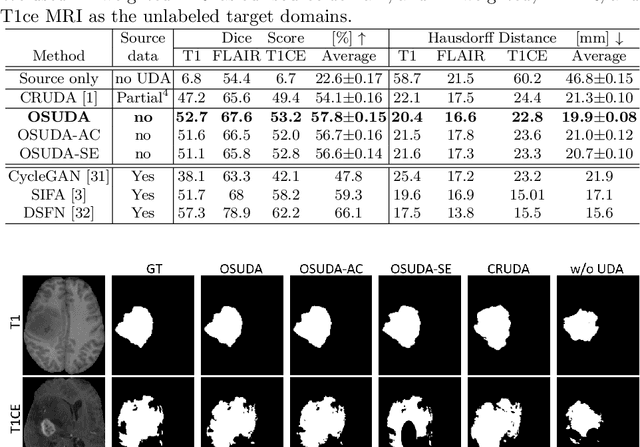
Abstract:Unsupervised domain adaptation (UDA) aims to transfer knowledge learned from a labeled source domain to an unlabeled and unseen target domain, which is usually trained on data from both domains. Access to the source domain data at the adaptation stage, however, is often limited, due to data storage or privacy issues. To alleviate this, in this work, we target source free UDA for segmentation, and propose to adapt an ``off-the-shelf" segmentation model pre-trained in the source domain to the target domain, with an adaptive batch-wise normalization statistics adaptation framework. Specifically, the domain-specific low-order batch statistics, i.e., mean and variance, are gradually adapted with an exponential momentum decay scheme, while the consistency of domain shareable high-order batch statistics, i.e., scaling and shifting parameters, is explicitly enforced by our optimization objective. The transferability of each channel is adaptively measured first from which to balance the contribution of each channel. Moreover, the proposed source free UDA framework is orthogonal to unsupervised learning methods, e.g., self-entropy minimization, which can thus be simply added on top of our framework. Extensive experiments on the BraTS 2018 database show that our source free UDA framework outperformed existing source-relaxed UDA methods for the cross-subtype UDA segmentation task and yielded comparable results for the cross-modality UDA segmentation task, compared with a supervised UDA methods with the source data.
Dual-cycle Constrained Bijective VAE-GAN For Tagged-to-Cine Magnetic Resonance Image Synthesis
Jan 14, 2021
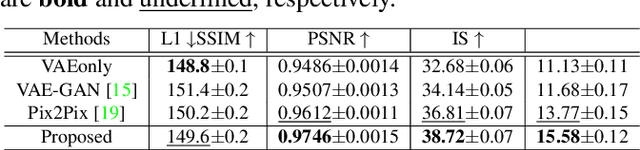

Abstract:Tagged magnetic resonance imaging (MRI) is a widely used imaging technique for measuring tissue deformation in moving organs. Due to tagged MRI's intrinsic low anatomical resolution, another matching set of cine MRI with higher resolution is sometimes acquired in the same scanning session to facilitate tissue segmentation, thus adding extra time and cost. To mitigate this, in this work, we propose a novel dual-cycle constrained bijective VAE-GAN approach to carry out tagged-to-cine MR image synthesis. Our method is based on a variational autoencoder backbone with cycle reconstruction constrained adversarial training to yield accurate and realistic cine MR images given tagged MR images. Our framework has been trained, validated, and tested using 1,768, 416, and 1,560 subject-independent paired slices of tagged and cine MRI from twenty healthy subjects, respectively, demonstrating superior performance over the comparison methods. Our method can potentially be used to reduce the extra acquisition time and cost, while maintaining the same workflow for further motion analyses.
A Unified Conditional Disentanglement Framework for Multimodal Brain MR Image Translation
Jan 14, 2021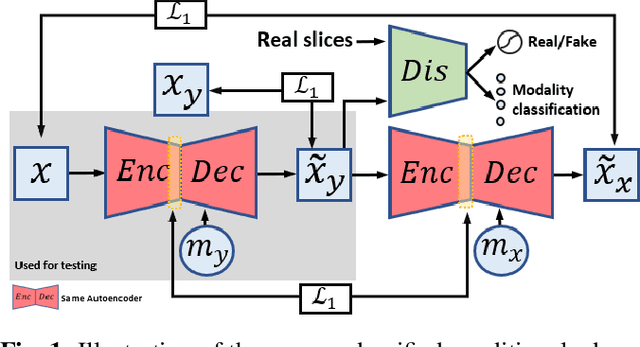
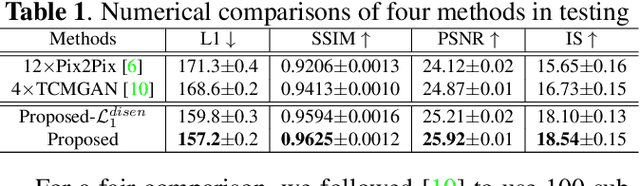
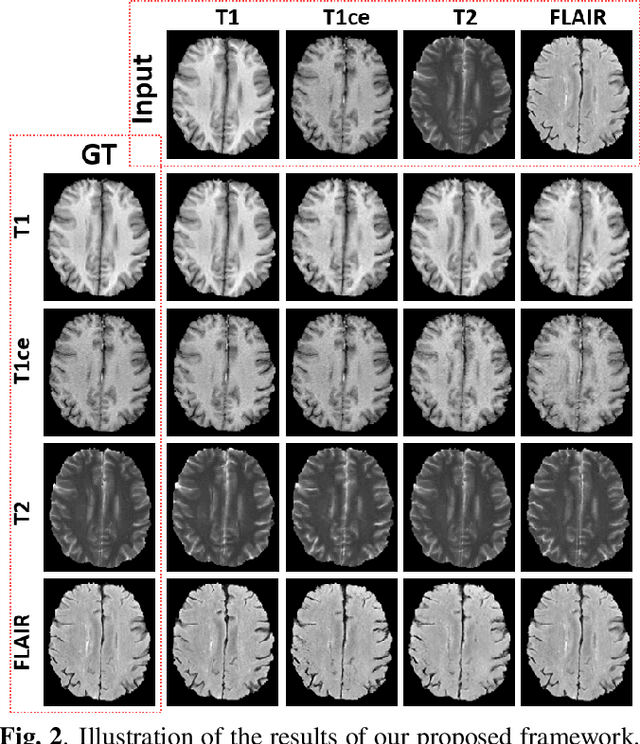
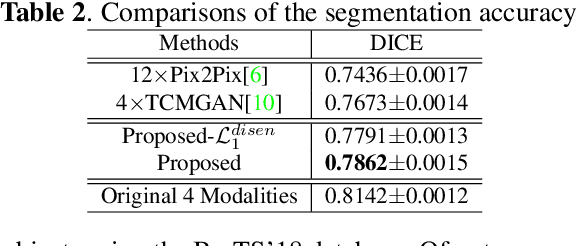
Abstract:Multimodal MRI provides complementary and clinically relevant information to probe tissue condition and to characterize various diseases. However, it is often difficult to acquire sufficiently many modalities from the same subject due to limitations in study plans, while quantitative analysis is still demanded. In this work, we propose a unified conditional disentanglement framework to synthesize any arbitrary modality from an input modality. Our framework hinges on a cycle-constrained conditional adversarial training approach, where it can extract a modality-invariant anatomical feature with a modality-agnostic encoder and generate a target modality with a conditioned decoder. We validate our framework on four MRI modalities, including T1-weighted, T1 contrast enhanced, T2-weighted, and FLAIR MRI, from the BraTS'18 database, showing superior performance on synthesis quality over the comparison methods. In addition, we report results from experiments on a tumor segmentation task carried out with synthesized data.
VoxelHop: Successive Subspace Learning for ALS Disease Classification Using Structural MRI
Jan 13, 2021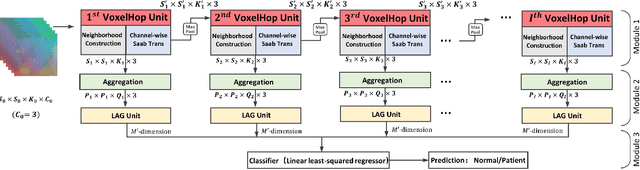
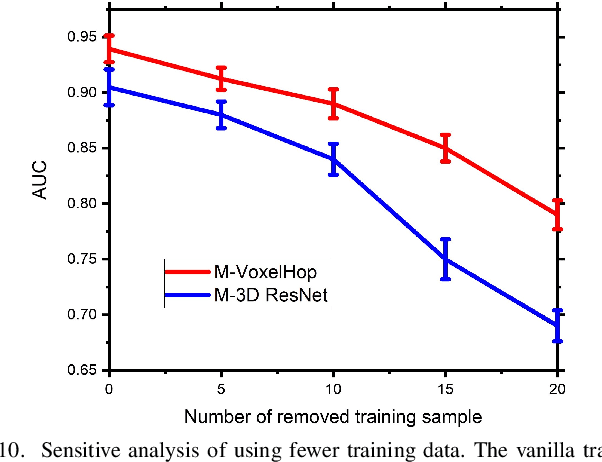
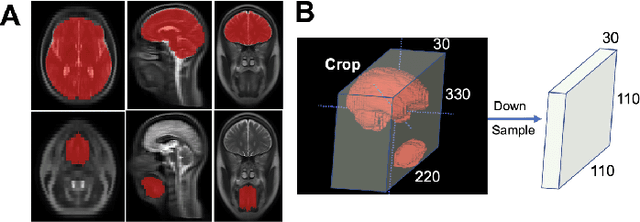
Abstract:Deep learning has great potential for accurate detection and classification of diseases with medical imaging data, but the performance is often limited by the number of training datasets and memory requirements. In addition, many deep learning models are considered a "black-box," thereby often limiting their adoption in clinical applications. To address this, we present a successive subspace learning model, termed VoxelHop, for accurate classification of Amyotrophic Lateral Sclerosis (ALS) using T2-weighted structural MRI data. Compared with popular convolutional neural network (CNN) architectures, VoxelHop has modular and transparent structures with fewer parameters without any backpropagation, so it is well-suited to small dataset size and 3D imaging data. Our VoxelHop has four key components, including (1) sequential expansion of near-to-far neighborhood for multi-channel 3D data; (2) subspace approximation for unsupervised dimension reduction; (3) label-assisted regression for supervised dimension reduction; and (4) concatenation of features and classification between controls and patients. Our experimental results demonstrate that our framework using a total of 20 controls and 26 patients achieves an accuracy of 93.48$\%$ and an AUC score of 0.9394 in differentiating patients from controls, even with a relatively small number of datasets, showing its robustness and effectiveness. Our thorough evaluations also show its validity and superiority to the state-of-the-art 3D CNN classification methods. Our framework can easily be generalized to other classification tasks using different imaging modalities.
Subtype-aware Unsupervised Domain Adaptation for Medical Diagnosis
Jan 11, 2021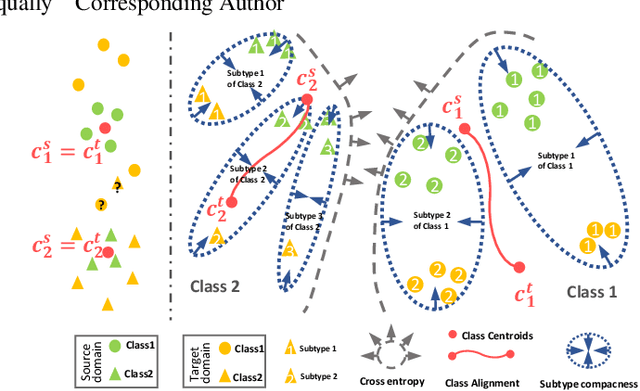
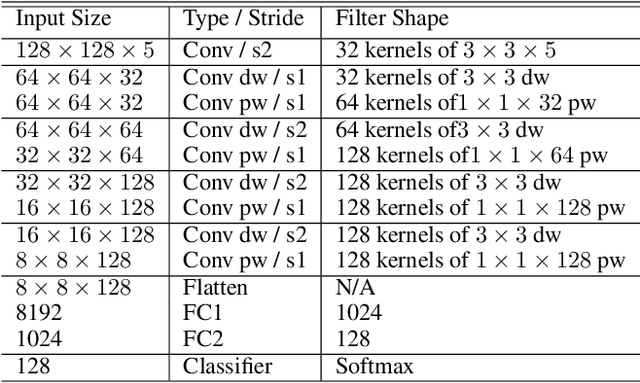
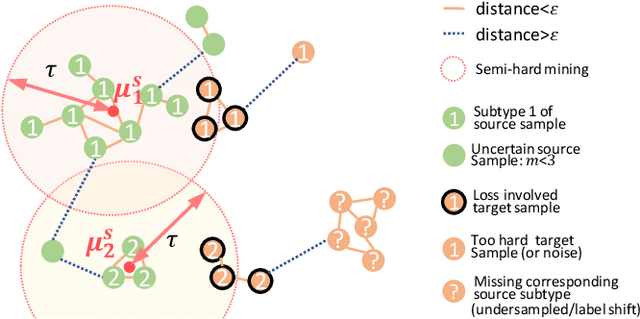
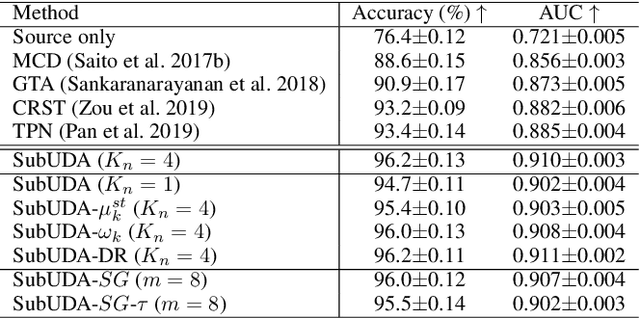
Abstract:Recent advances in unsupervised domain adaptation (UDA) show that transferable prototypical learning presents a powerful means for class conditional alignment, which encourages the closeness of cross-domain class centroids. However, the cross-domain inner-class compactness and the underlying fine-grained subtype structure remained largely underexplored. In this work, we propose to adaptively carry out the fine-grained subtype-aware alignment by explicitly enforcing the class-wise separation and subtype-wise compactness with intermediate pseudo labels. Our key insight is that the unlabeled subtypes of a class can be divergent to one another with different conditional and label shifts, while inheriting the local proximity within a subtype. The cases of with or without the prior information on subtype numbers are investigated to discover the underlying subtype structure in an online fashion. The proposed subtype-aware dynamic UDA achieves promising results on medical diagnosis tasks.
A Deep Joint Sparse Non-negative Matrix Factorization Framework for Identifying the Common and Subject-specific Functional Units of Tongue Motion During Speech
Jul 09, 2020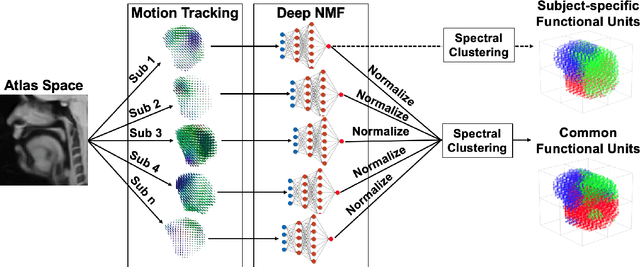
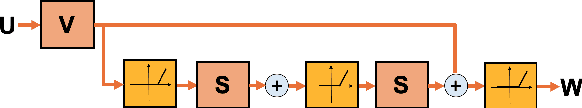
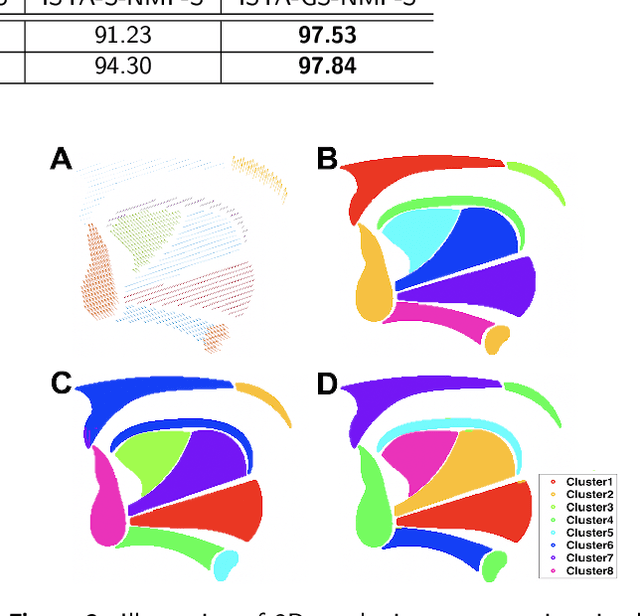
Abstract:Intelligible speech is produced by creating varying internal local muscle groupings---i.e., functional units---that are generated in a systematic and coordinated manner. There are two major challenges in characterizing and analyzing functional units. First, due to the complex and convoluted nature of tongue structure and function, it is of great importance to develop a method that can accurately decode complex muscle coordination patterns during speech. Second, it is challenging to keep identified functional units across subjects comparable due to their substantial variability. In this work, to address these challenges, we develop a new deep learning framework to identify common and subject-specific functional units of tongue motion during speech. Our framework hinges on joint deep graph-regularized sparse non-negative matrix factorization (NMF) using motion quantities derived from displacements by tagged Magnetic Resonance Imaging. More specifically, we transform NMF with sparse and manifold regularizations into modular architectures akin to deep neural networks by means of unfolding the Iterative Shrinkage-Thresholding Algorithm to learn interpretable building blocks and associated weighting map. We then apply spectral clustering to common and subject-specific functional units. Experiments carried out with simulated datasets show that the proposed method surpasses the comparison methods. Experiments carried out with in vivo tongue motion datasets show that the proposed method can determine the common and subject-specific functional units with increased interpretability and decreased size variability.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge