Florian Dubost
for the ALFA study
Automated Detection of Patients in Hospital Video Recordings
Nov 28, 2021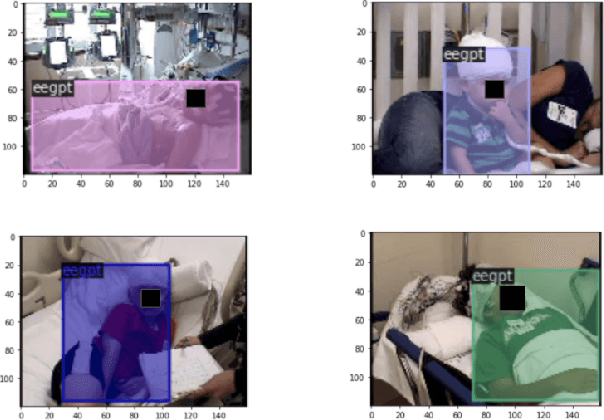
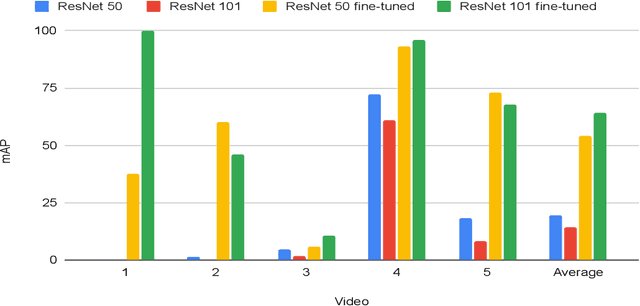
Abstract:In a clinical setting, epilepsy patients are monitored via video electroencephalogram (EEG) tests. A video EEG records what the patient experiences on videotape while an EEG device records their brainwaves. Currently, there are no existing automated methods for tracking the patient's location during a seizure, and video recordings of hospital patients are substantially different from publicly available video benchmark datasets. For example, the camera angle can be unusual, and patients can be partially covered with bedding sheets and electrode sets. Being able to track a patient in real-time with video EEG would be a promising innovation towards improving the quality of healthcare. Specifically, an automated patient detection system could supplement clinical oversight and reduce the resource-intensive efforts of nurses and doctors who need to continuously monitor patients. We evaluate an ImageNet pre-trained Mask R-CNN, a standard deep learning model for object detection, on the task of patient detection using our own curated dataset of 45 videos of hospital patients. The dataset was aggregated and curated for this work. We show that without fine-tuning, ImageNet pre-trained Mask R-CNN models perform poorly on such data. By fine-tuning the models with a subset of our dataset, we observe a substantial improvement in patient detection performance, with a mean average precision of 0.64. We show that the results vary substantially depending on the video clip.
Automated Segmentation and Volume Measurement of Intracranial Carotid Artery Calcification on Non-Contrast CT
Jul 20, 2021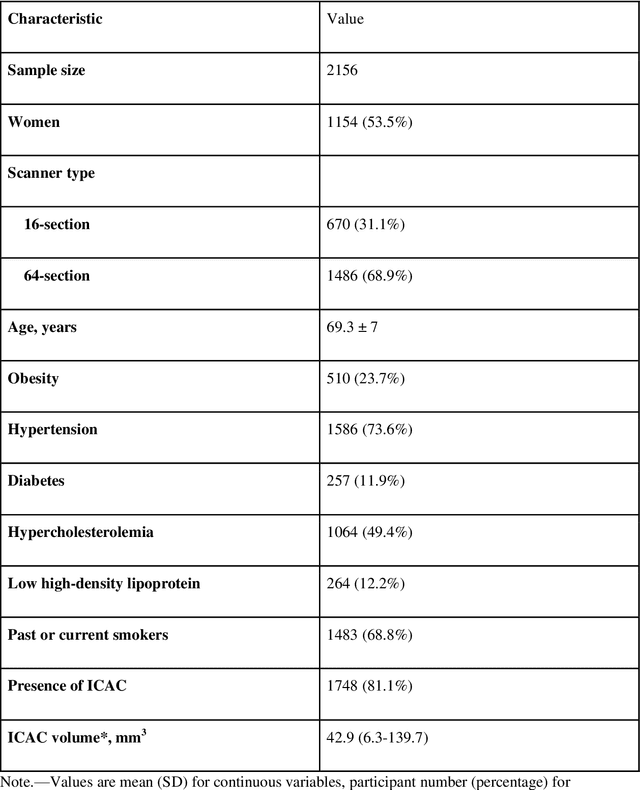
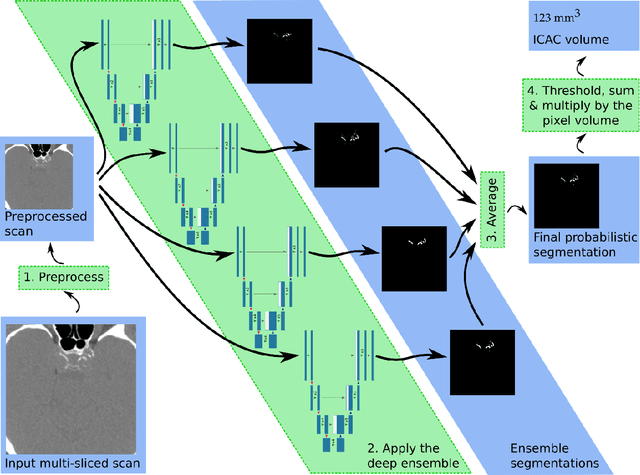
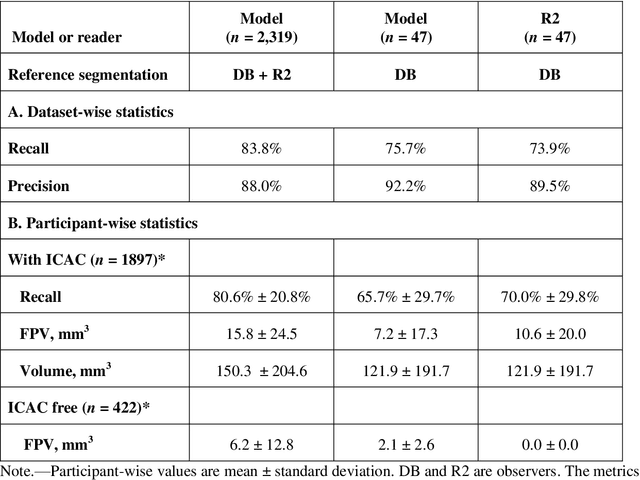
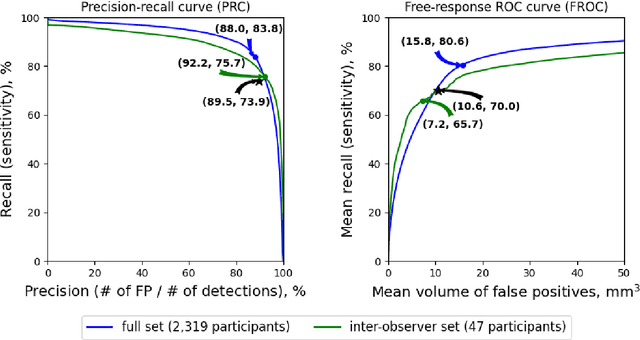
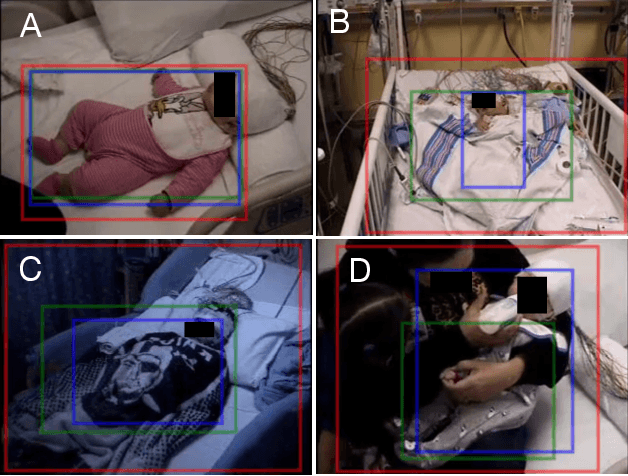
Abstract:Purpose: To evaluate a fully-automated deep-learning-based method for assessment of intracranial carotid artery calcification (ICAC). Methods: Two observers manually delineated ICAC in non-contrast CT scans of 2,319 participants (mean age 69 (SD 7) years; 1154 women) of the Rotterdam Study, prospectively collected between 2003 and 2006. These data were used to retrospectively develop and validate a deep-learning-based method for automated ICAC delineation and volume measurement. To evaluate the method, we compared manual and automatic assessment (computed using ten-fold cross-validation) with respect to 1) the agreement with an independent observer's assessment (available in a random subset of 47 scans); 2) the accuracy in delineating ICAC as judged via blinded visual comparison by an expert; 3) the association with first stroke incidence from the scan date until 2012. All method performance metrics were computed using 10-fold cross-validation. Results: The automated delineation of ICAC reached sensitivity of 83.8% and positive predictive value (PPV) of 88%. The intraclass correlation between automatic and manual ICAC volume measures was 0.98 (95% CI: 0.97, 0.98; computed in the entire dataset). Measured between the assessments of independent observers, sensitivity was 73.9%, PPV was 89.5%, and intraclass correlation was 0.91 (95% CI: 0.84, 0.95; computed in the 47-scan subset). In the blinded visual comparisons, automatic delineations were more accurate than manual ones (p-value = 0.01). The association of ICAC volume with incident stroke was similarly strong for both automated (hazard ratio, 1.38 (95% CI: 1.12, 1.75) and manually measured volumes (hazard ratio, 1.48 (95% CI: 1.20, 1.87)). Conclusions: The developed model was capable of automated segmentation and volume quantification of ICAC with accuracy comparable to human experts.
Double Descent Optimization Pattern and Aliasing: Caveats of Noisy Labels
Jun 03, 2021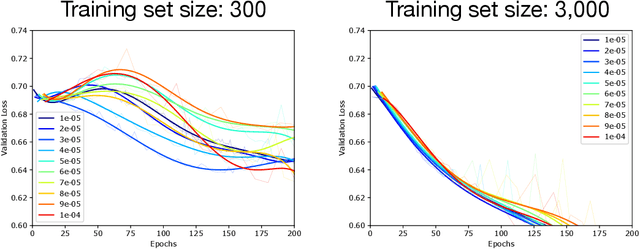
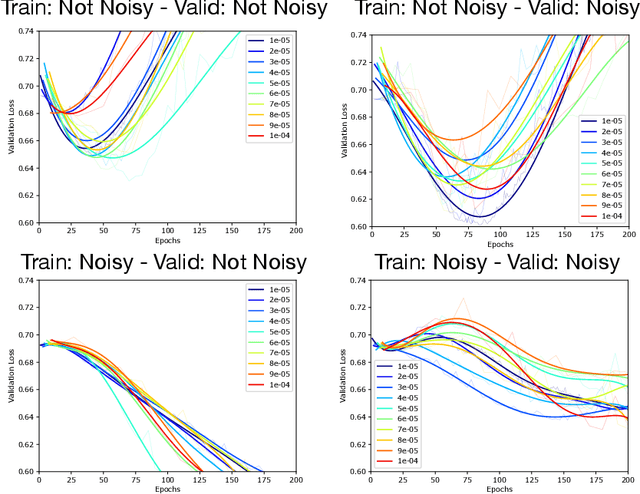
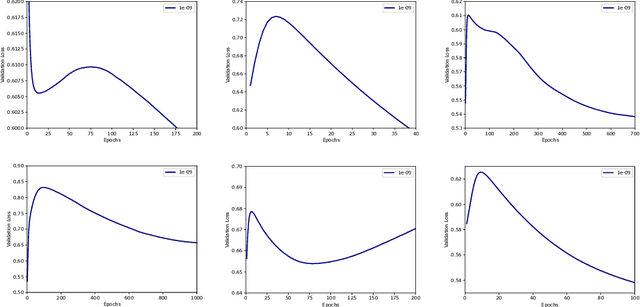
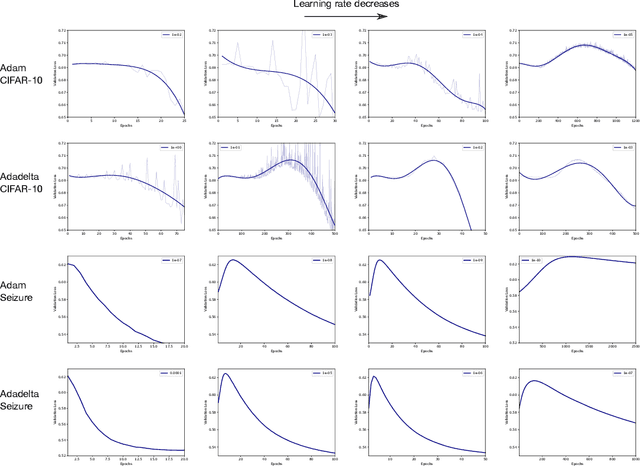
Abstract:Optimization plays a key role in the training of deep neural networks. Deciding when to stop training can have a substantial impact on the performance of the network during inference. Under certain conditions, the generalization error can display a double descent pattern during training: the learning curve is non-monotonic and seemingly diverges before converging again after additional epochs. This optimization pattern can lead to early stopping procedures to stop training before the second convergence and consequently select a suboptimal set of parameters for the network, with worse performance during inference. In this work, in addition to confirming that double descent occurs with small datasets and noisy labels as evidenced by others, we show that noisy labels must be present both in the training and generalization sets to observe a double descent pattern. We also show that the learning rate has an influence on double descent, and study how different optimizers and optimizer parameters influence the apparition of double descent. Finally, we show that increasing the learning rate can create an aliasing effect that masks the double descent pattern without suppressing it. We study this phenomenon through extensive experiments on variants of CIFAR-10 and show that they translate to a real world application: the forecast of seizure events in epileptic patients from continuous electroencephalographic recordings.
Automated Seizure Detection and Seizure Type Classification From Electroencephalography With a Graph Neural Network and Self-Supervised Pre-Training
Apr 16, 2021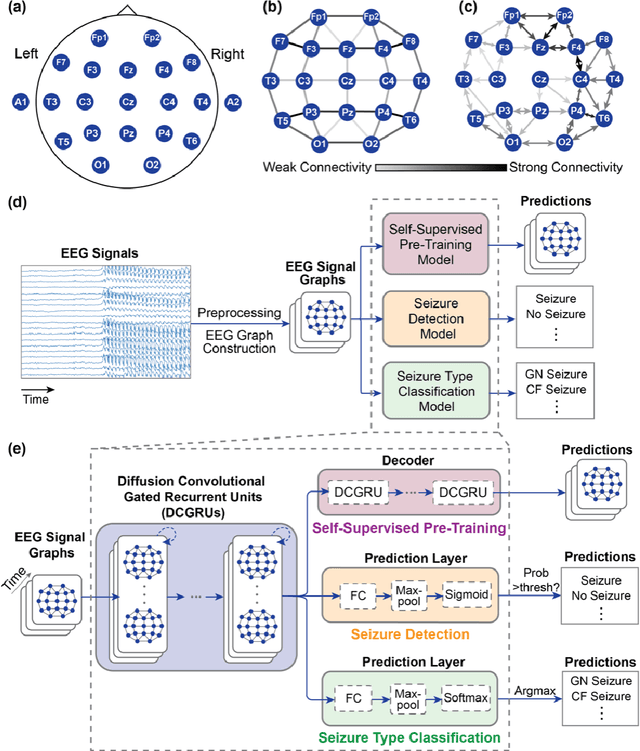
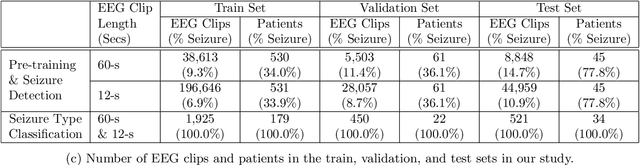
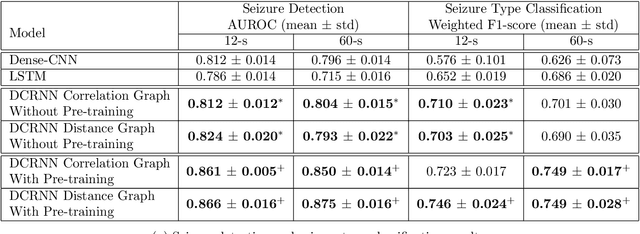
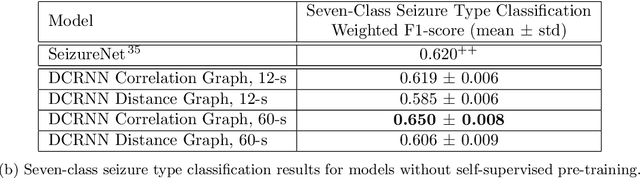
Abstract:Automated seizure detection and classification from electroencephalography (EEG) can greatly improve the diagnosis and treatment of seizures. While prior studies mainly used convolutional neural networks (CNNs) that assume image-like structure in EEG signals or spectrograms, this modeling choice does not reflect the natural geometry of or connectivity between EEG electrodes. In this study, we propose modeling EEGs as graphs and present a graph neural network for automated seizure detection and classification. In addition, we leverage unlabeled EEG data using a self-supervised pre-training strategy. Our graph model with self-supervised pre-training significantly outperforms previous state-of-the-art CNN and Long Short-Term Memory (LSTM) models by 6.3 points (7.8%) in Area Under the Receiver Operating Characteristic curve (AUROC) for seizure detection and 6.3 points (9.2%) in weighted F1-score for seizure type classification. Ablation studies show that our graph-based modeling approach significantly outperforms existing CNN or LSTM models, and that self-supervision helps further improve the model performance. Moreover, we find that self-supervised pre-training substantially improves model performance on combined tonic seizures, a low-prevalence seizure type. Furthermore, our model interpretability analysis suggests that our model is better at identifying seizure regions compared to an existing CNN. In summary, our graph-based modeling approach integrates domain knowledge about EEG, sets a new state-of-the-art for seizure detection and classification on a large public dataset (5,499 EEG files), and provides better ability to identify seizure regions.
Adversarial Heart Attack: Neural Networks Fooled to Segment Heart Symbols in Chest X-Ray Images
Apr 07, 2021
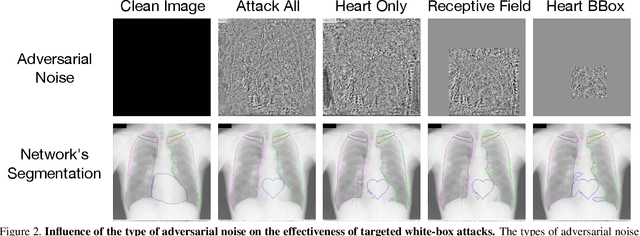
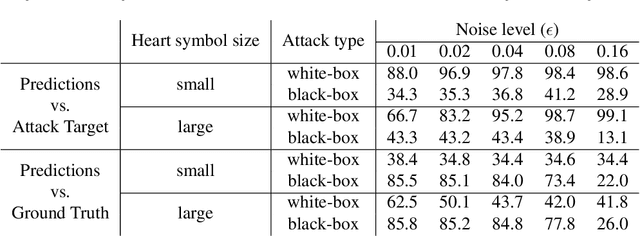
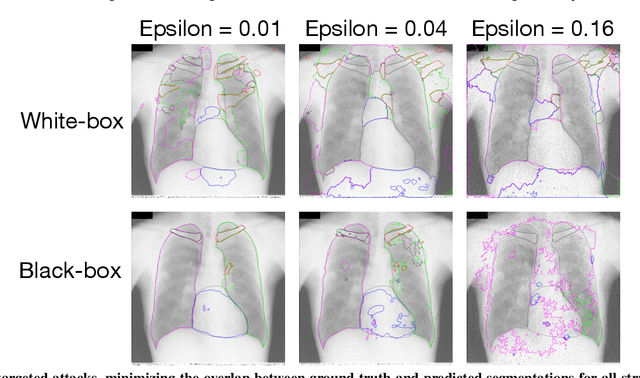
Abstract:Adversarial attacks consist in maliciously changing the input data to mislead the predictions of automated decision systems and are potentially a serious threat for automated medical image analysis. Previous studies have shown that it is possible to adversarially manipulate automated segmentations produced by neural networks in a targeted manner in the white-box attack setting. In this article, we studied the effectiveness of adversarial attacks in targeted modification of segmentations of anatomical structures in chest X-rays. Firstly, we experimented with using anatomically implausible shapes as targets for adversarial manipulation. We showed that, by adding almost imperceptible noise to the image, we can reliably force state-of-the-art neural networks to segment the heart as a heart symbol instead of its real anatomical shape. Moreover, such heart-shaping attack did not appear to require higher adversarial noise level than an untargeted attack based the same attack method. Secondly, we attempted to explore the limits of adversarial manipulation of segmentations. For that, we assessed the effectiveness of shrinking and enlarging segmentation contours for the three anatomical structures. We observed that adversarially extending segmentations of structures into regions with intensity and texture uncharacteristic for them presented a challenge to our attacks, as well as, in some cases, changing segmentations in ways that conflict with class adjacency priors learned by the target network. Additionally, we evaluated performances of the untargeted attacks and targeted heart attacks in the black-box attack scenario, using a surrogate network trained on a different subset of images. In both cases, the attacks were substantially less effective. We believe these findings bring novel insights into the current capabilities and limits of adversarial attacks for semantic segmentation.
Inaccurate Supervision of Neural Networks with Incorrect Labels: Application to Epilepsy
Dec 01, 2020
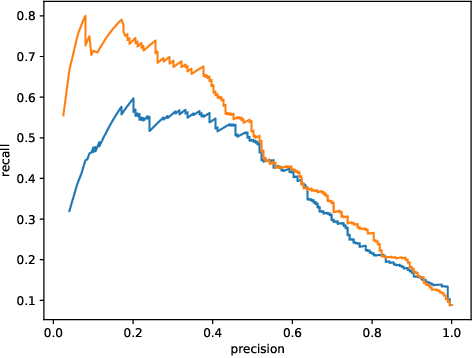
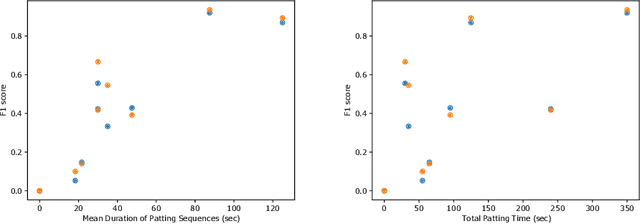
Abstract:This work describes multiple weak supervision strategies for video processing with neural networks in the context of seizure detection. To study seizure onset, we have designed automated methods to detect seizures from electroencephalography (EEG), a modality used for recording electrical brain activity. However, the EEG signal alone is sometimes not enough for existing detection methods to discriminate seizure from artifacts having a similar signal on EEG. For example, such artifacts could be triggered by patting, rocking or suctioning in the case of neonates. In this article, we addressed this problem by automatically detecting an example artifact -- patting of neonates -- from continuous video recordings of neonates acquired during clinical routine. We computed frame-to-frame cross-correlation matrices to isolate patterns showing repetitive movements indicative of patting of the patient. Next, a convolutional neural network was trained to classify whether these matrices contained patting events using weak training labels -- noisy labels generated during daily clinical procedure. The labels were considered weak as they were sometimes incorrect. We investigated whether networks trained with more samples, containing more uncertain and weak labels, could achieve a higher performance. Our results showed that, in the case of patting detection, such networks could achieve a higher recall, without sacrificing precision. These networks focused on areas of the cross-correlation matrices that were more meaningful to the task. More generally, our work gives insights into building more accurate models from weakly labelled time sequences.
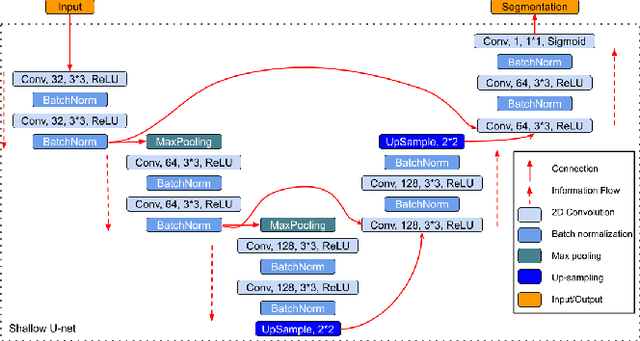
DS6: Deformation-aware learning for small vessel segmentation with small, imperfectly labeled dataset
Jun 18, 2020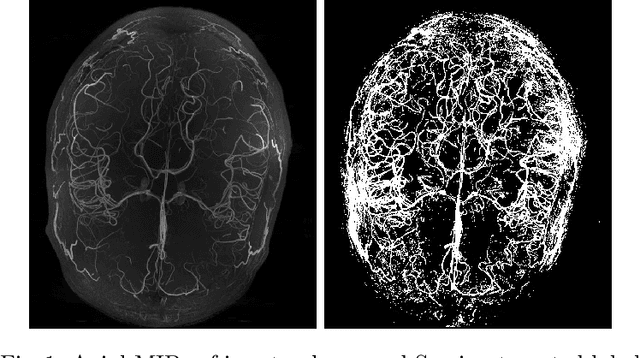
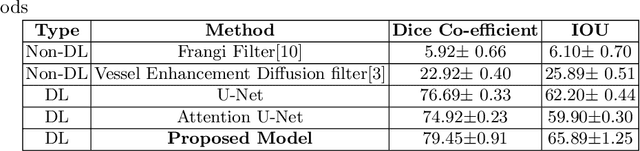


Abstract:Originating from the initial segment of the middle cerebral artery of the human brain, Lenticulostriate Arteries (LSA) are a collection of perforating vessels that supply blood to the basal ganglia region. With the advancement of 7 Tesla scanner, we are able to detect these LSA which are linked to Small Vessel Diseases(SVD) and potentially a cause for neurodegenerative diseases. Segmentation of LSA with traditional approaches like Frangi or semi-automated/manual techniques can depict medium to large vessels but fail to depict the small vessels. Also, semi-automated/manual approaches are time-consuming. In this paper, we put forth a study that incorporates deep learning techniques to automatically segment these LSA using 3D 7 Tesla Time-of-fight Magnetic Resonance Angiogram images. The algorithm is trained and evaluated on a small dataset of 11 volumes. Deep learning models based on Multi-Scale Supervision U-Net accompanied by elastic deformations to give equivariance to the model, were utilized for the vessel segmentation using semi-automated labeled images. We make a qualitative analysis of the output with the original image and also on imperfect semi-manual labels to confirm the presence and continuity of small vessels.
Adversarial Attack Vulnerability of Medical Image Analysis Systems: Unexplored Factors
Jun 12, 2020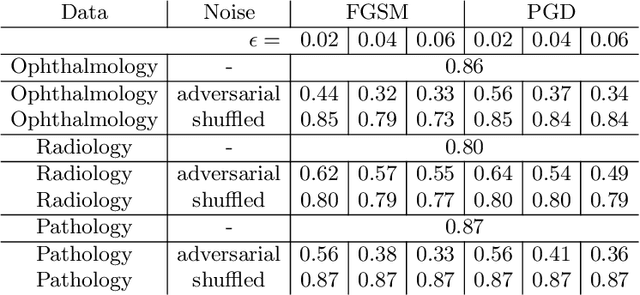
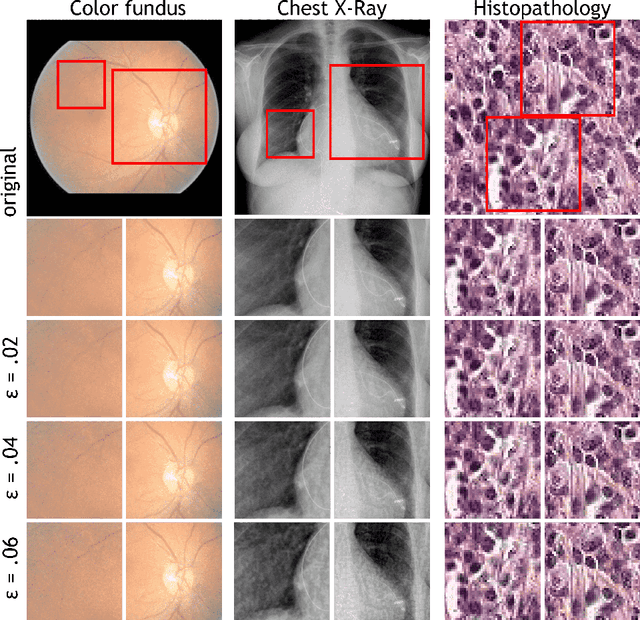
Abstract:Adversarial attacks are considered a potentially serious security threat for machine learning systems. Medical image analysis (MedIA) systems have recently been argued to be particularly vulnerable to adversarial attacks due to strong financial incentives. In this paper, we study several previously unexplored factors affecting adversarial attack vulnerability of deep learning MedIA systems in three medical domains: ophthalmology, radiology and pathology. Firstly, we study the effect of varying the degree of adversarial perturbation on the attack performance and its visual perceptibility. Secondly, we study how pre-training on a public dataset (ImageNet) affects the models' vulnerability to attacks. Thirdly, we study the influence of data and model architecture disparity between target and attacker models. Our experiments show that the degree of perturbation significantly affects both performance and human perceptibility of attacks. Pre-training may dramatically increase the transfer of adversarial examples; the larger the performance gain achieved by pre-training, the larger the transfer. Finally, disparity in data and/or model architecture between target and attacker models substantially decreases the success of attacks. We believe that these factors should be considered when designing cybersecurity-critical MedIA systems, as well as kept in mind when evaluating their vulnerability to adversarial attacks.
Spectral Data Augmentation Techniques to quantify Lung Pathology from CT-images
Apr 24, 2020
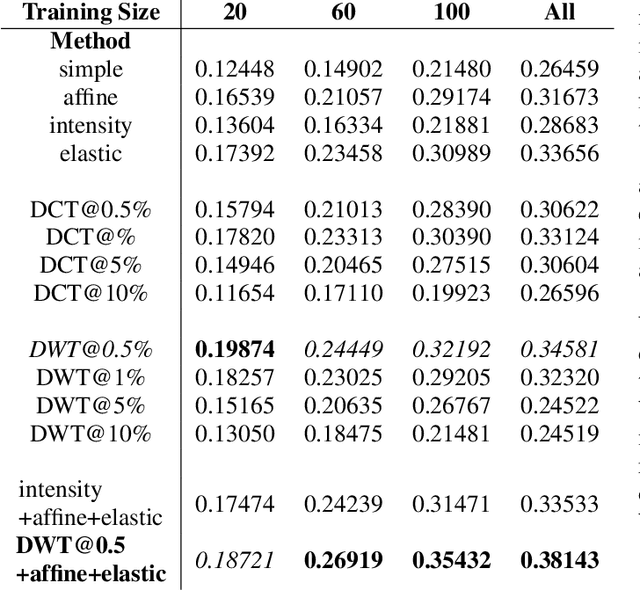
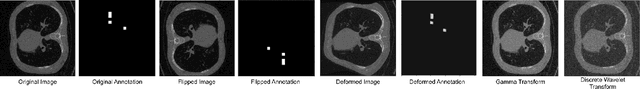
Abstract:Data augmentation is of paramount importance in biomedical image processing tasks, characterized by inadequate amounts of labelled data, to best use all of the data that is present. In-use techniques range from intensity transformations and elastic deformations, to linearly combining existing data points to make new ones. In this work, we propose the use of spectral techniques for data augmentation, using the discrete cosine and wavelet transforms. We empirically evaluate our approaches on a CT texture analysis task to detect abnormal lung-tissue in patients with cystic fibrosis. Empirical experiments show that the proposed spectral methods perform favourably as compared to the existing methods. When used in combination with existing methods, our proposed approach can increase the relative minor class segmentation performance by 44.1% over a simple replication baseline.
When Weak Becomes Strong: Robust Quantification of White Matter Hyperintensities in Brain MRI scans
Apr 12, 2020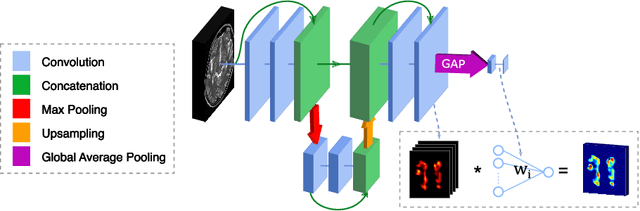
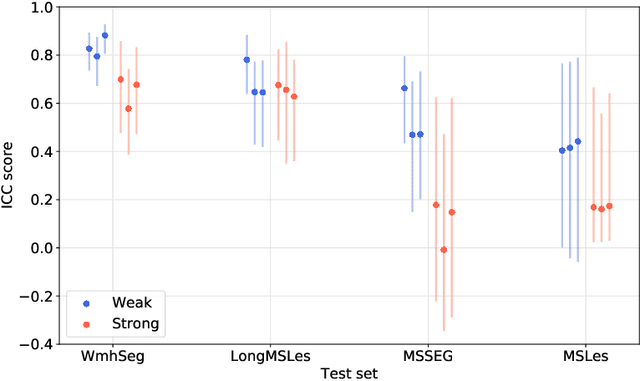
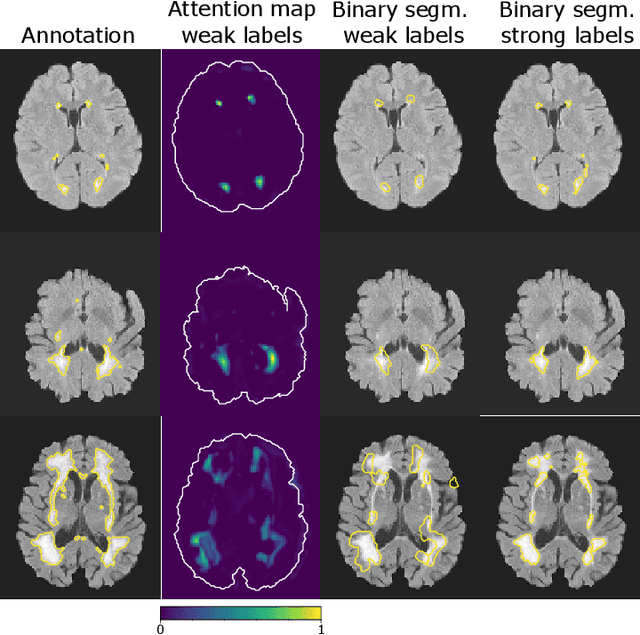
Abstract:To measure the volume of specific image structures, a typical approach is to first segment those structures using a neural network trained on voxel-wise (strong) labels and subsequently compute the volume from the segmentation. A more straightforward approach would be to predict the volume directly using a neural network based regression approach, trained on image-level (weak) labels indicating volume. In this article, we compared networks optimized with weak and strong labels, and study their ability to generalize to other datasets. We experimented with white matter hyperintensity (WMH) volume prediction in brain MRI scans. Neural networks were trained on a large local dataset and their performance was evaluated on four independent public datasets. We showed that networks optimized using only weak labels reflecting WMH volume generalized better for WMH volume prediction than networks optimized with voxel-wise segmentations of WMH. The attention maps of networks trained with weak labels did not seem to delineate WMHs, but highlighted instead areas with smooth contours around or near WMHs. By correcting for possible confounders we showed that networks trained on weak labels may have learnt other meaningful features that are more suited to generalization to unseen data. Our results suggest that for imaging biomarkers that can be derived from segmentations, training networks to predict the biomarker directly may provide more robust results than solving an intermediate segmentation step.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge