Elisa Donati
Neuromorphic Spiking Ring Attractor for Proprioceptive Joint-State Estimation
Apr 15, 2026Abstract:Maintaining stable internal representations of continuous variables is fundamental for effective robotic control. Continuous attractor networks provide a biologically inspired mechanism for encoding such variables, yet neuromorphic realizations have rarely addressed proprioceptive estimation under resource constraints. This work introduces a spiking ring-attractor network representing a robot joint angle through self-sustaining population activity. Local excitation and broad inhibition support a stable activity bump, while velocity-modulated asymmetries drive its translation and boundary conditions confine motion within mechanical limits. The network reproduces smooth trajectory tracking and remains stable near joint limits, showing reduced drift and improved accuracy compared to unbounded models. Such compact hardware-compatible implementation preserves multi-second stability demonstrating a near-linear relationship between bump velocity and synaptic modulation.
Lightweight Test-Time Adaptation for EMG-Based Gesture Recognition
Jan 07, 2026Abstract:Reliable long-term decoding of surface electromyography (EMG) is hindered by signal drift caused by electrode shifts, muscle fatigue, and posture changes. While state-of-the-art models achieve high intra-session accuracy, their performance often degrades sharply. Existing solutions typically demand large datasets or high-compute pipelines that are impractical for energy-efficient wearables. We propose a lightweight framework for Test-Time Adaptation (TTA) using a Temporal Convolutional Network (TCN) backbone. We introduce three deployment-ready strategies: (i) causal adaptive batch normalization for real-time statistical alignment; (ii) a Gaussian Mixture Model (GMM) alignment with experience replay to prevent forgetting; and (iii) meta-learning for rapid, few-shot calibration. Evaluated on the NinaPro DB6 multi-session dataset, our framework significantly bridges the inter-session accuracy gap with minimal overhead. Our results show that experience-replay updates yield superior stability under limited data, while meta-learning achieves competitive performance in one- and two-shot regimes using only a fraction of the data required by current benchmarks. This work establishes a path toward robust, "plug-and-play" myoelectric control for long-term prosthetic use.
Queen Detection in Beehives via Environmental Sensor Fusion for Low-Power Edge Computing
Sep 17, 2025Abstract:Queen bee presence is essential for the health and stability of honeybee colonies, yet current monitoring methods rely on manual inspections that are labor-intensive, disruptive, and impractical for large-scale beekeeping. While recent audio-based approaches have shown promise, they often require high power consumption, complex preprocessing, and are susceptible to ambient noise. To overcome these limitations, we propose a lightweight, multimodal system for queen detection based on environmental sensor fusion-specifically, temperature, humidity, and pressure differentials between the inside and outside of the hive. Our approach employs quantized decision tree inference on a commercial STM32 microcontroller, enabling real-time, low-power edge computing without compromising accuracy. We show that our system achieves over 99% queen detection accuracy using only environmental inputs, with audio features offering no significant performance gain. This work presents a scalable and sustainable solution for non-invasive hive monitoring, paving the way for autonomous, precision beekeeping using off-the-shelf, energy-efficient hardware.
Spiking Neural Network Decoders of Finger Forces from High-Density Intramuscular Microelectrode Arrays
Sep 04, 2025



Abstract:Restoring naturalistic finger control in assistive technologies requires the continuous decoding of motor intent with high accuracy, efficiency, and robustness. Here, we present a spike-based decoding framework that integrates spiking neural networks (SNNs) with motor unit activity extracted from high-density intramuscular microelectrode arrays. We demonstrate simultaneous and proportional decoding of individual finger forces from motor unit spike trains during isometric contractions at 15% of maximum voluntary contraction using SNNs. We systematically evaluated alternative SNN decoder configurations and compared two possible input modalities: physiologically grounded motor unit spike trains and spike-encoded intramuscular EMG signals. Through this comparison, we quantified trade-offs between decoding accuracy, memory footprint, and robustness to input errors. The results showed that shallow SNNs can reliably decode finger-level motor intent with competitive accuracy and minimal latency, while operating with reduced memory requirements and without the need for external preprocessing buffers. This work provides a practical blueprint for integrating SNNs into finger-level force decoding systems, demonstrating how the choice of input representation can be strategically tailored to meet application-specific requirements for accuracy, robustness, and memory efficiency.
Heterogeneous Population Encoding for Multi-joint Regression using sEMG Signals
Jan 25, 2025Abstract:Regression-based decoding of continuous movements is essential for human-machine interfaces (HMIs), such as prosthetic control. This study explores a feature-based approach to encoding Surface Electromyography (sEMG) signals, focusing on the role of variability in neural-inspired population encoding. By employing heterogeneous populations of Leaky Integrate-and- Fire (LIF) neurons with varying sizes and diverse parameter distributions, we investigate how population size and variability in encoding parameters, such as membrane time constants and thresholds, influence decoding performance. Using a simple linear readout, we demonstrate that variability improves robustness and generalizability compared to single-neuron encoders. These findings emphasize the importance of optimizing variability and population size for efficient and scalable regression tasks in spiking neural networks (SNNs), paving the way for robust, low-power HMI implementations.
Intramuscular High-Density Micro-Electrode Arrays Enable High-Precision Decoding and Mapping of Spinal Motor Neurons to Reveal Hand Control
Oct 14, 2024



Abstract:Decoding nervous system activity is a key challenge in neuroscience and neural interfacing. In this study, we propose a novel neural decoding system that enables unprecedented large-scale sampling of muscle activity. Using micro-electrode arrays with more than 100 channels embedded within the forearm muscles, we recorded high-density signals that captured multi-unit motor neuron activity. This extensive sampling was complemented by advanced methods for neural decomposition, analysis, and classification, allowing us to accurately detect and interpret the spiking activity of spinal motor neurons that innervate hand muscles. We evaluated this system in two healthy participants, each implanted with three electromyogram (EMG) micro-electrode arrays (comprising 40 electrodes each) in the forearm. These arrays recorded muscle activity during both single- and multi-digit isometric contractions. For the first time under controlled conditions, we demonstrate that multi-digit tasks elicit unique patterns of motor neuron recruitment specific to each task, rather than employing combinations of recruitment patterns from single-digit tasks. This observation led us to hypothesize that hand tasks could be classified with high precision based on the decoded neural activity. We achieved perfect classification accuracy (100%) across 12 distinct single- and multi-digit tasks, and consistently high accuracy (>96\%) across all conditions and subjects, for up to 16 task classes. These results significantly outperformed conventional EMG classification methods. The exceptional performance of this system paves the way for developing advanced neural interfaces based on invasive high-density EMG technology. This innovation could greatly enhance human-computer interaction and lead to substantial improvements in assistive technologies, offering new possibilities for restoring motor function in clinical applications.
Evaluation of Encoding Schemes on Ubiquitous Sensor Signal for Spiking Neural Network
Jul 12, 2024Abstract:Spiking neural networks (SNNs), a brain-inspired computing paradigm, are emerging for their inference performance, particularly in terms of energy efficiency and latency attributed to the plasticity in signal processing. To deploy SNNs in ubiquitous computing systems, signal encoding of sensors is crucial for achieving high accuracy and robustness. Using inertial sensor readings for gym activity recognition as a case study, this work comprehensively evaluates four main encoding schemes and deploys the corresponding SNN on the neuromorphic processor Loihi2 for post-deployment encoding assessment. Rate encoding, time-to-first-spike encoding, binary encoding, and delta modulation are evaluated using metrics like average fire rate, signal-to-noise ratio, classification accuracy, robustness, and inference latency and energy. In this case study, the time-to-first-spike encoding required the lowest firing rate (2%) and achieved a comparative accuracy (89%), although it was the least robust scheme against error spikes (over 20% accuracy drop with 0.1 noisy spike rate). Rate encoding with optimal value-to-probability mapping achieved the highest accuracy (91.7%). Binary encoding provided a balance between information reconstruction and noise resistance. Multi-threshold delta modulation showed the best robustness, with only a 0.7% accuracy drop at a 0.1 noisy spike rate. This work serves researchers in selecting the best encoding scheme for SNN-based ubiquitous sensor signal processing, tailored to specific performance requirements.
Feed-forward and recurrent inhibition for compressing and classifying high dynamic range biosignals in spiking neural network architectures
Sep 28, 2023



Abstract:Neuromorphic processors that implement Spiking Neural Networks (SNNs) using mixed-signal analog/digital circuits represent a promising technology for closed-loop real-time processing of biosignals. As in biology, to minimize power consumption, the silicon neurons' circuits are configured to fire with a limited dynamic range and with maximum firing rates restricted to a few tens or hundreds of Herz. However, biosignals can have a very large dynamic range, so encoding them into spikes without saturating the neuron outputs represents an open challenge. In this work, we present a biologically-inspired strategy for compressing this high-dynamic range in SNN architectures, using three adaptation mechanisms ubiquitous in the brain: spike-frequency adaptation at the single neuron level, feed-forward inhibitory connections from neurons belonging to the input layer, and Excitatory-Inhibitory (E-I) balance via recurrent inhibition among neurons in the output layer. We apply this strategy to input biosignals encoded using both an asynchronous delta modulation method and an energy-based pulse-frequency modulation method. We validate this approach in silico, simulating a simple network applied to a gesture classification task from surface EMG recordings.
Long-term stable Electromyography classification using Canonical Correlation Analysis
Jan 23, 2023


Abstract:Discrimination of hand gestures based on the decoding of surface electromyography (sEMG) signals is a well-establish approach for controlling prosthetic devices and for Human-Machine Interfaces (HMI). However, despite the promising results achieved by this approach in well-controlled experimental conditions, its deployment in long-term real-world application scenarios is still hindered by several challenges. One of the most critical challenges is maintaining high EMG data classification performance across multiple days without retraining the decoding system. The drop in performance is mostly due to the high EMG variability caused by electrodes shift, muscle artifacts, fatigue, user adaptation, or skin-electrode interfacing issues. Here we propose a novel statistical method based on canonical correlation analysis (CCA) that stabilizes EMG classification performance across multiple days for long-term control of prosthetic devices. We show how CCA can dramatically decrease the performance drop of standard classifiers observed across days, by maximizing the correlation among multiple-day acquisition data sets. Our results show how the performance of a classifier trained on EMG data acquired only of the first day of the experiment maintains 90% relative accuracy across multiple days, compensating for the EMG data variability that occurs over long-term periods, using the CCA transformation on data obtained from a small number of gestures. This approach eliminates the need for large data sets and multiple or periodic training sessions, which currently hamper the usability of conventional pattern recognition based approaches
Neuromorphic implementation of ECG anomaly detection using delay chains
Sep 02, 2022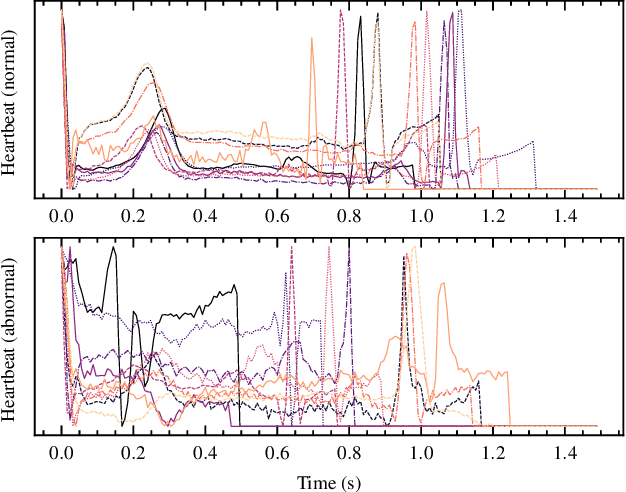
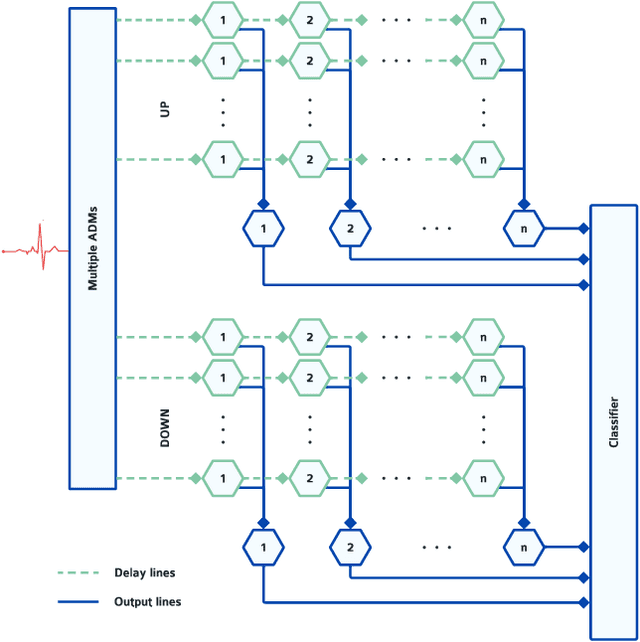
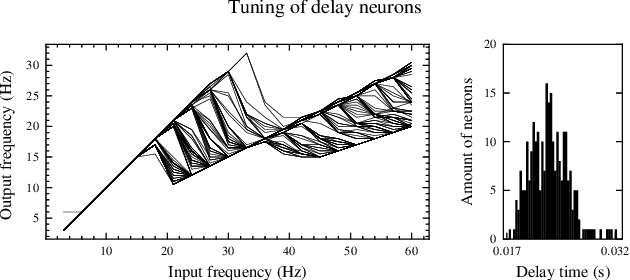
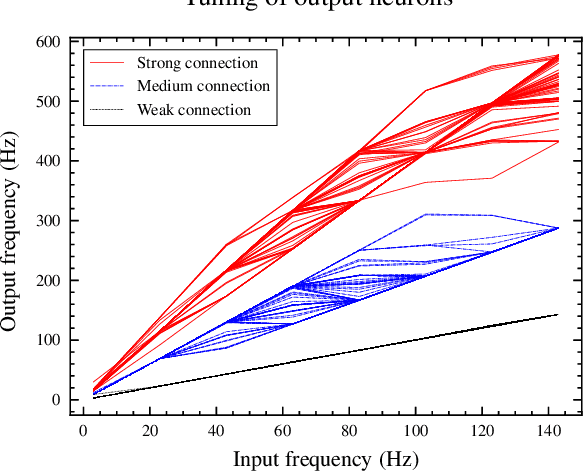
Abstract:Real-time analysis and classification of bio-signals measured using wearable devices is computationally costly and requires dedicated low-power hardware. One promising approach is to use spiking neural networks implemented using in-memory computing architectures and neuromorphic electronic circuits. However, as these circuits process data in streaming mode without the possibility of storing it in external buffers, a major challenge lies in the processing of spatio-temporal signals that last longer than the time constants present in the network synapses and neurons. Here we propose to extend the memory capacity of a spiking neural network by using parallel delay chains. We show that it is possible to map temporal signals of multiple seconds into spiking activity distributed across multiple neurons which have time constants of few milliseconds. We validate this approach on an ECG anomaly detection task and present experimental results that demonstrate how temporal information is properly preserved in the network activity.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge