Eliot Siegel
One Copy Is All You Need: Resource-Efficient Streaming of Medical Imaging Data at Scale
Jul 01, 2023Abstract:Large-scale medical imaging datasets have accelerated development of artificial intelligence tools for clinical decision support. However, the large size of these datasets is a bottleneck for users with limited storage and bandwidth. Many users may not even require such large datasets as AI models are often trained on lower resolution images. If users could directly download at their desired resolution, storage and bandwidth requirements would significantly decrease. However, it is impossible to anticipate every users' requirements and impractical to store the data at multiple resolutions. What if we could store images at a single resolution but send them at different ones? We propose MIST, an open-source framework to operationalize progressive resolution for streaming medical images at multiple resolutions from a single high-resolution copy. We demonstrate that MIST can dramatically reduce imaging infrastructure inefficiencies for hosting and streaming medical images by >90%, while maintaining diagnostic quality for deep learning applications.
High-Throughput AI Inference for Medical Image Classification and Segmentation using Intelligent Streaming
May 24, 2023Abstract:As the adoption of AI systems within the clinical setup grows, limitations in bandwidth could create communication bottlenecks when streaming imaging data, leading to delays in patient diagnosis and treatment. As such, healthcare providers and AI vendors will require greater computational infrastructure, therefore dramatically increasing costs. To that end, we developed intelligent streaming, a state-of-the-art framework to enable accelerated, cost-effective, bandwidth-optimized, and computationally efficient AI inference for clinical decision making at scale. For classification, intelligent streaming reduced the data transmission by 99.01% and decoding time by 98.58%, while increasing throughput by 27.43x. For segmentation, our framework reduced data transmission by 90.32%, decoding time by 90.26%, while increasing throughput by 4.20x. Our work demonstrates that intelligent streaming results in faster turnaround times, and reduced overall cost of data and transmission, without negatively impacting clinical decision making using AI systems.
Issues and Challenges in Applications of Artificial Intelligence to Nuclear Medicine -- The Bethesda Report (AI Summit 2022)
Nov 07, 2022

Abstract:The SNMMI Artificial Intelligence (SNMMI-AI) Summit, organized by the SNMMI AI Task Force, took place in Bethesda, MD on March 21-22, 2022. It brought together various community members and stakeholders from academia, healthcare, industry, patient representatives, and government (NIH, FDA), and considered various key themes to envision and facilitate a bright future for routine, trustworthy use of AI in nuclear medicine. In what follows, essential issues, challenges, controversies and findings emphasized in the meeting are summarized.
A brief history of AI: how to prevent another winter
Sep 08, 2021
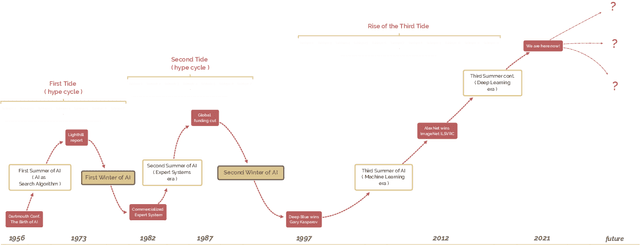

Abstract:The field of artificial intelligence (AI), regarded as one of the most enigmatic areas of science, has witnessed exponential growth in the past decade including a remarkably wide array of applications, having already impacted our everyday lives. Advances in computing power and the design of sophisticated AI algorithms have enabled computers to outperform humans in a variety of tasks, especially in the areas of computer vision and speech recognition. Yet, AI's path has never been smooth, having essentially fallen apart twice in its lifetime ('winters' of AI), both after periods of popular success ('summers' of AI). We provide a brief rundown of AI's evolution over the course of decades, highlighting its crucial moments and major turning points from inception to the present. In doing so, we attempt to learn, anticipate the future, and discuss what steps may be taken to prevent another 'winter'.
Rapid AI Development Cycle for the Coronavirus (COVID-19) Pandemic: Initial Results for Automated Detection & Patient Monitoring using Deep Learning CT Image Analysis
Mar 24, 2020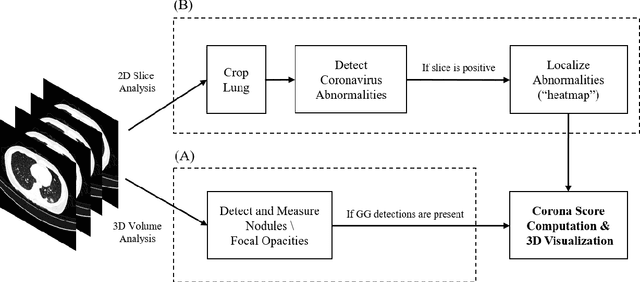
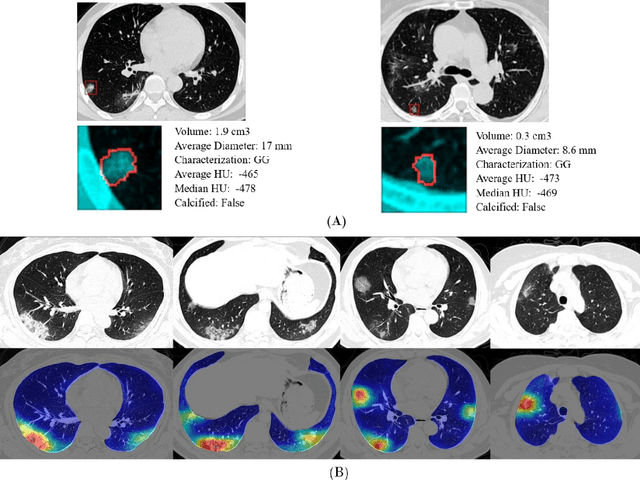
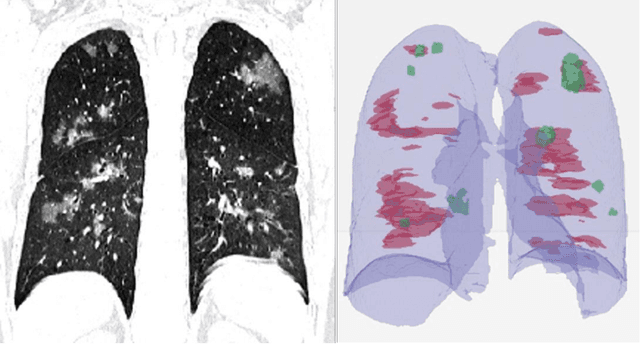
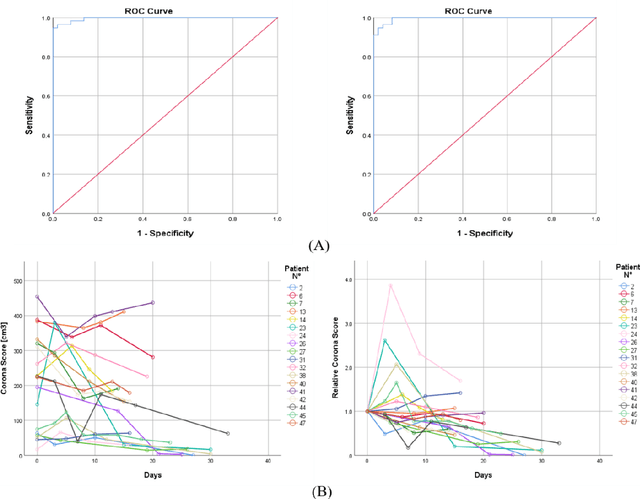
Abstract:Purpose: Develop AI-based automated CT image analysis tools for detection, quantification, and tracking of Coronavirus; demonstrate they can differentiate coronavirus patients from non-patients. Materials and Methods: Multiple international datasets, including from Chinese disease-infected areas were included. We present a system that utilizes robust 2D and 3D deep learning models, modifying and adapting existing AI models and combining them with clinical understanding. We conducted multiple retrospective experiments to analyze the performance of the system in the detection of suspected COVID-19 thoracic CT features and to evaluate evolution of the disease in each patient over time using a 3D volume review, generating a Corona score. The study includes a testing set of 157 international patients (China and U.S). Results: Classification results for Coronavirus vs Non-coronavirus cases per thoracic CT studies were 0.996 AUC (95%CI: 0.989-1.00) ; on datasets of Chinese control and infected patients. Possible working point: 98.2% sensitivity, 92.2% specificity. For time analysis of Coronavirus patients, the system output enables quantitative measurements for smaller opacities (volume, diameter) and visualization of the larger opacities in a slice-based heat map or a 3D volume display. Our suggested Corona score measures the progression of disease over time. Conclusion: This initial study, which is currently being expanded to a larger population, demonstrated that rapidly developed AI-based image analysis can achieve high accuracy in detection of Coronavirus as well as quantification and tracking of disease burden.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge