Caroline Moore
Collaborative Quantization Embeddings for Intra-Subject Prostate MR Image Registration
Jul 14, 2022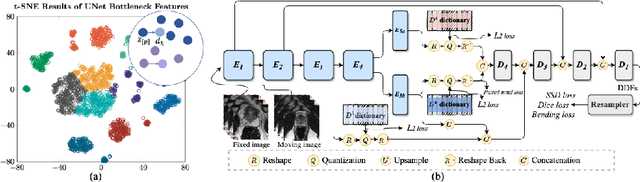
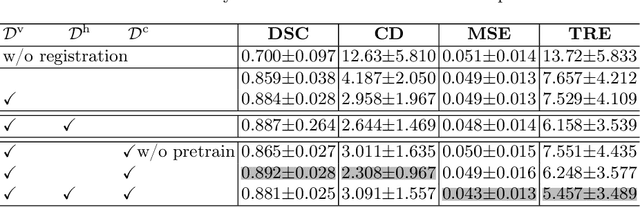
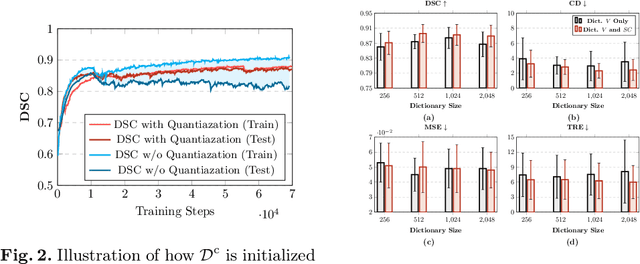
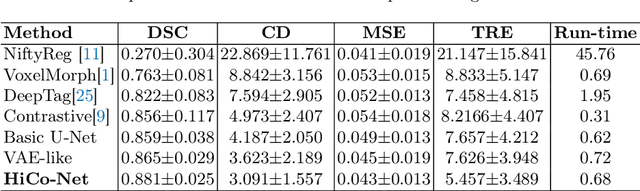
Abstract:Image registration is useful for quantifying morphological changes in longitudinal MR images from prostate cancer patients. This paper describes a development in improving the learning-based registration algorithms, for this challenging clinical application often with highly variable yet limited training data. First, we report that the latent space can be clustered into a much lower dimensional space than that commonly found as bottleneck features at the deep layer of a trained registration network. Based on this observation, we propose a hierarchical quantization method, discretizing the learned feature vectors using a jointly-trained dictionary with a constrained size, in order to improve the generalisation of the registration networks. Furthermore, a novel collaborative dictionary is independently optimised to incorporate additional prior information, such as the segmentation of the gland or other regions of interest, in the latent quantized space. Based on 216 real clinical images from 86 prostate cancer patients, we show the efficacy of both the designed components. Improved registration accuracy was obtained with statistical significance, in terms of both Dice on gland and target registration error on corresponding landmarks, the latter of which achieved 5.46 mm, an improvement of 28.7\% from the baseline without quantization. Experimental results also show that the difference in performance was indeed minimised between training and testing data.
Morphological Change Forecasting for Prostate Glands using Feature-based Registration and Kernel Density Extrapolation
Jan 16, 2021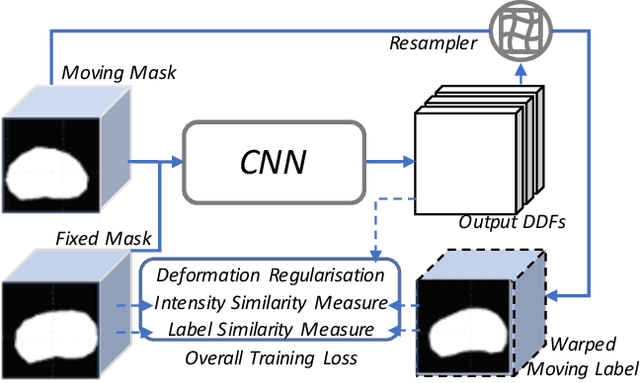
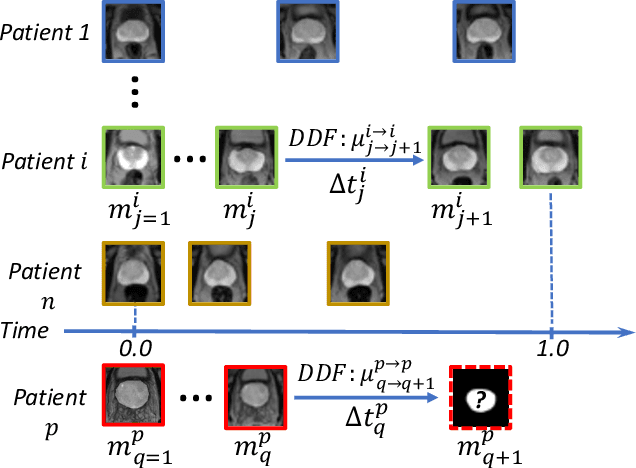
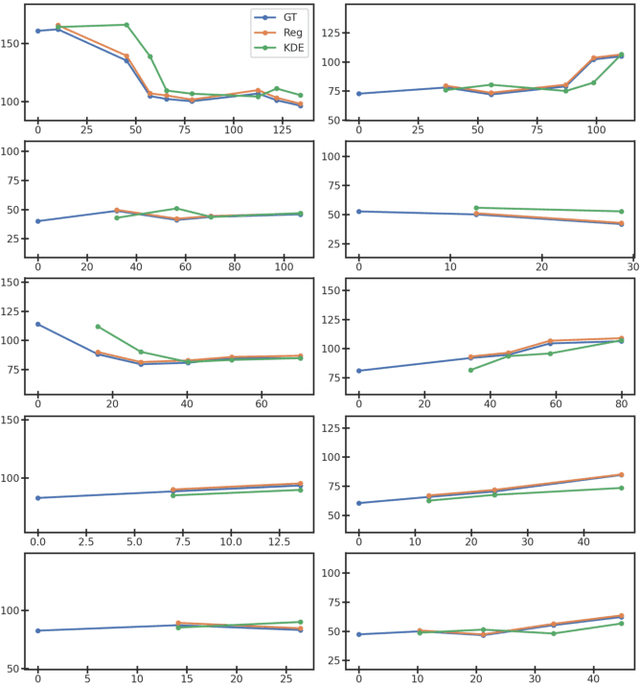
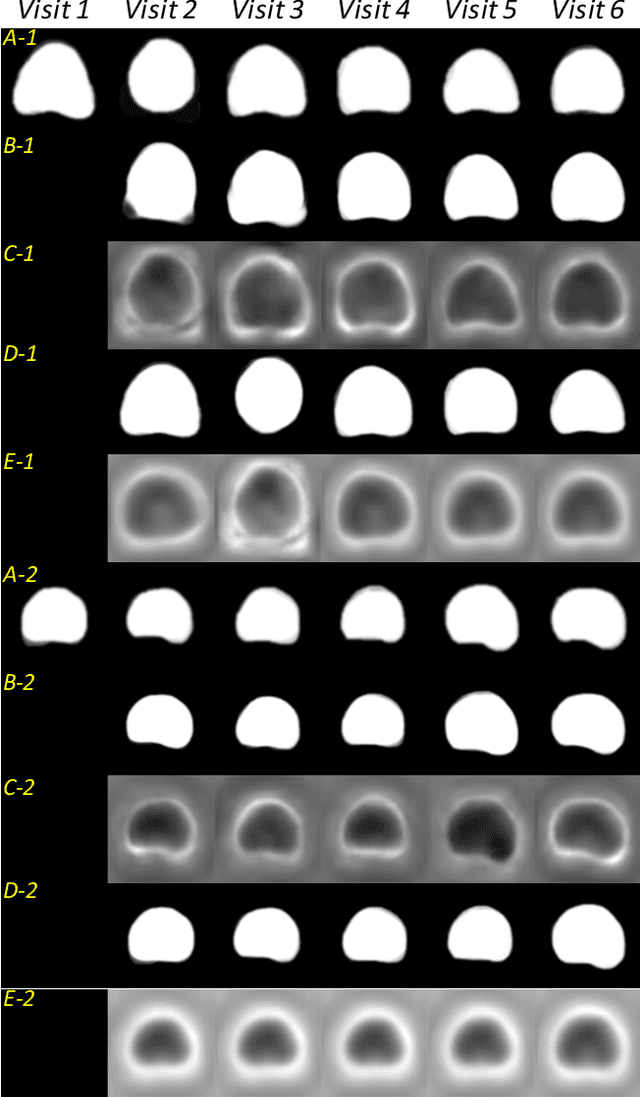
Abstract:Organ morphology is a key indicator for prostate disease diagnosis and prognosis. For instance, In longitudinal study of prostate cancer patients under active surveillance, the volume, boundary smoothness and their changes are closely monitored on time-series MR image data. In this paper, we describe a new framework for forecasting prostate morphological changes, as the ability to detect such changes earlier than what is currently possible may enable timely treatment or avoiding unnecessary confirmatory biopsies. In this work, an efficient feature-based MR image registration is first developed to align delineated prostate gland capsules to quantify the morphological changes using the inferred dense displacement fields (DDFs). We then propose to use kernel density estimation (KDE) of the probability density of the DDF-represented \textit{future morphology changes}, between current and future time points, before the future data become available. The KDE utilises a novel distance function that takes into account morphology, stage-of-progression and duration-of-change, which are considered factors in such subject-specific forecasting. We validate the proposed approach on image masks unseen to registration network training, without using any data acquired at the future target time points. The experiment results are presented on a longitudinal data set with 331 images from 73 patients, yielding an average Dice score of 0.865 on a holdout set, between the ground-truth and the image masks warped by the KDE-predicted-DDFs.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge