Yong Fan
from the iSTAGING consortium, for the ADNI
DeepSEED: 3D Squeeze-and-Excitation Encoder-Decoder ConvNets for Pulmonary Nodule Detection
Apr 06, 2019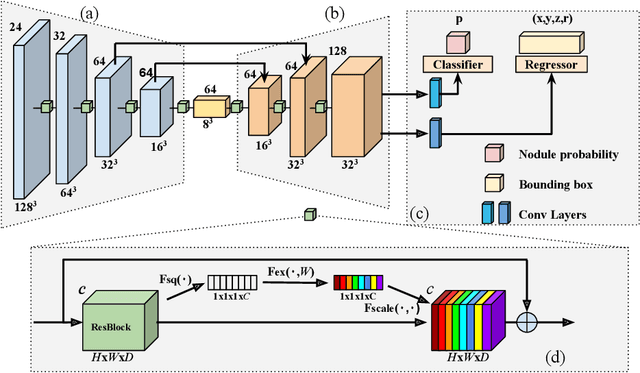
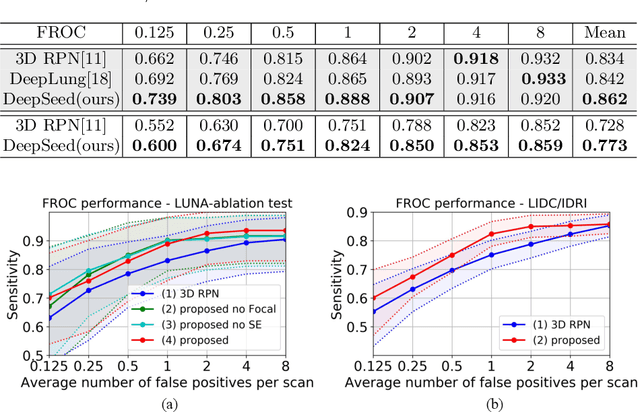

Abstract:Pulmonary nodule detection plays an important role in lung cancer screening with low-dose computed tomography (CT) scans. Although promising performance has been achieved by deep learning based nodule detection methods, it remains challenging to build nodule detection networks with good generalization performance due to unbalanced positive and negative samples. In order to overcome this problem and further improve state-of-the-art region proposal network methods, we develop a novel deep 3D convolutional neural network with an Encoder-Decoder structure for pulmonary nodule detection. Particularly, we utilize a dynamically scaled cross entropy loss to reduce the false positive rate and compensate the significant data imbalance problem. We adopt the squeeze-and-excitation structure to learn effective image features and fully utilize channel inter-dependency. We have validated our method based on publicly available CT scans from LIDC/IDRI dataset and its subset LUNA16 with thinner slices. Ablation studies and experimental results have demonstrated that our method could outperform state-of-the-art nodule detection methods by a large margin, with an average FROC score of 86.2% on LUNA16, and an average FROC score of 77.3% on LIDC/IDRI dataset when trained on LUNA16 only.
Fully-automatic segmentation of kidneys in clinical ultrasound images using a boundary distance regression network
Jan 05, 2019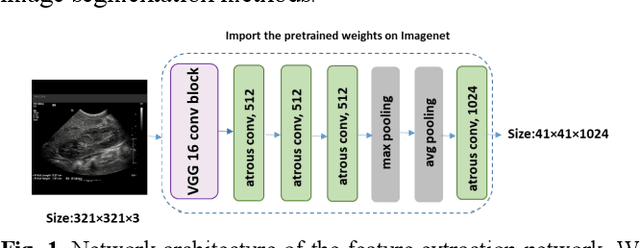
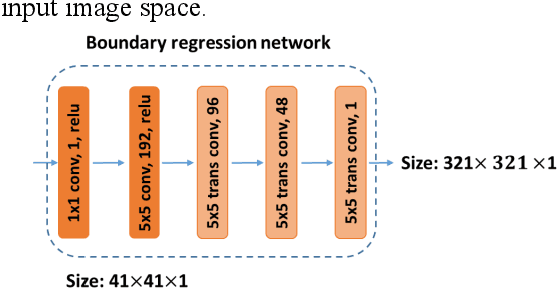
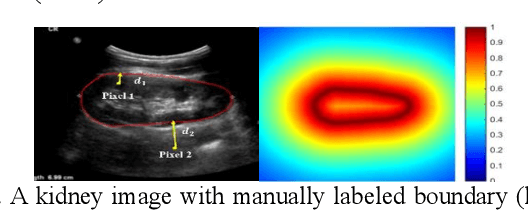
Abstract:It remains challenging to automatically segment kidneys in clinical ultrasound images due to the kidneys' varied shapes and image intensity distributions, although semi-automatic methods have achieved promising performance. In this study, we developed a novel boundary distance regression deep neural network to segment the kidneys, informed by the fact that the kidney boundaries are relatively consistent across images in terms of their appearance. Particularly, we first use deep neural networks pre-trained for classification of natural images to extract high-level image features from ultrasound images, then these feature maps are used as input to learn kidney boundary distance maps using a boundary distance regression network, and finally the predicted boundary distance maps are classified as kidney pixels or non-kidney pixels using a pixel classification network in an end-to-end learning fashion. Experimental results have demonstrated that our method could effectively improve the performance of automatic kidney segmentation, significantly better than deep learning based pixel classification networks.
Early Prediction of Alzheimer's Disease Dementia Based on Baseline Hippocampal MRI and 1-Year Follow-Up Cognitive Measures Using Deep Recurrent Neural Networks
Jan 05, 2019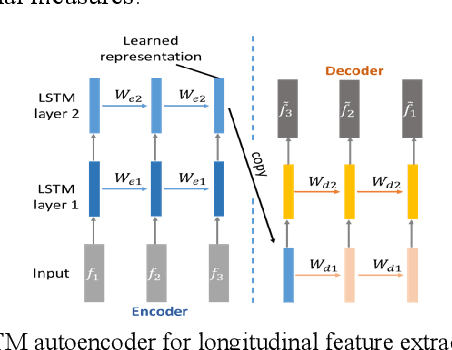
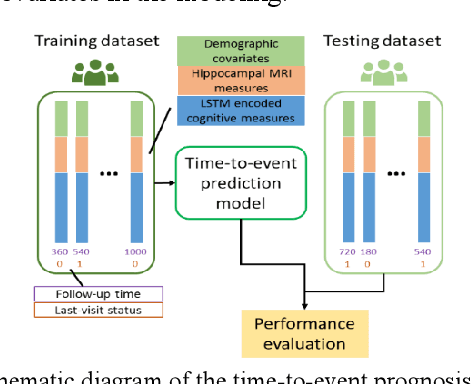
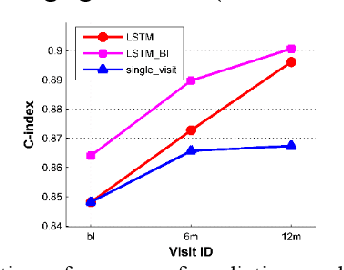
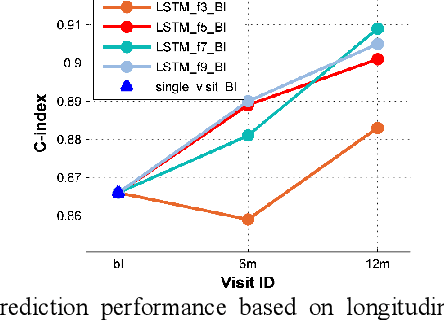
Abstract:Multi-modal biological, imaging, and neuropsychological markers have demonstrated promising performance for distinguishing Alzheimer's disease (AD) patients from cognitively normal elders. However, it remains difficult to early predict when and which mild cognitive impairment (MCI) individuals will convert to AD dementia. Informed by pattern classification studies which have demonstrated that pattern classifiers built on longitudinal data could achieve better classification performance than those built on cross-sectional data, we develop a deep learning model based on recurrent neural networks (RNNs) to learn informative representation and temporal dynamics of longitudinal cognitive measures of individual subjects and combine them with baseline hippocampal MRI for building a prognostic model of AD dementia progression. Experimental results on a large cohort of MCI subjects have demonstrated that the deep learning model could learn informative measures from longitudinal data for characterizing the progression of MCI subjects to AD dementia, and the prognostic model could early predict AD progression with high accuracy.
Deep Convolutional Neural Networks for Imaging Data Based Survival Analysis of Rectal Cancer
Jan 05, 2019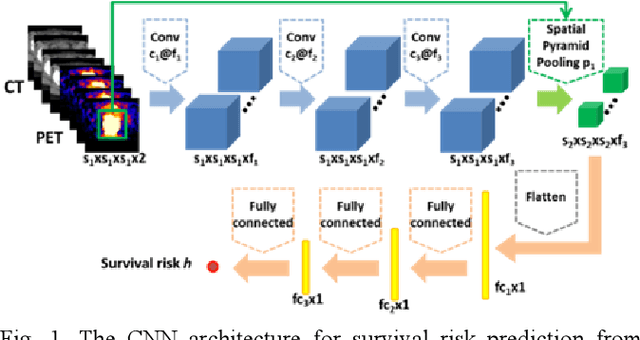
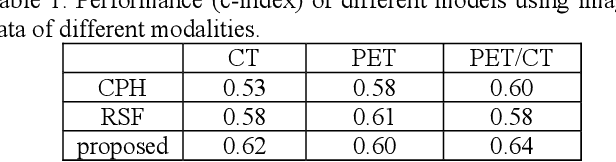
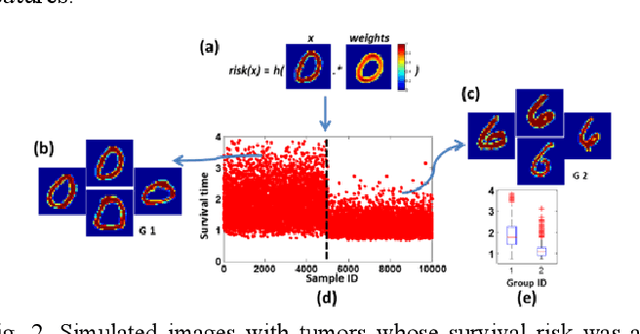
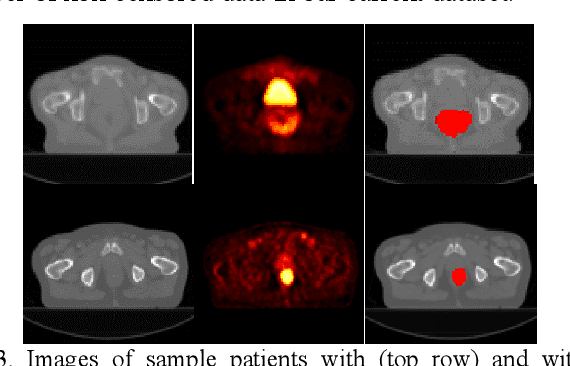
Abstract:Recent radiomic studies have witnessed promising performance of deep learning techniques in learning radiomic features and fusing multimodal imaging data. Most existing deep learning based radiomic studies build predictive models in a setting of pattern classification, not appropriate for survival analysis studies where some data samples have incomplete observations. To improve existing survival analysis techniques whose performance is hinged on imaging features, we propose a deep learning method to build survival regression models by optimizing imaging features with deep convolutional neural networks (CNNs) in a proportional hazards model. To make the CNNs applicable to tumors with varied sizes, a spatial pyramid pooling strategy is adopted. Our method has been validated based on a simulated imaging dataset and a FDG-PET/CT dataset of rectal cancer patients treated for locally advanced rectal cancer. Compared with survival prediction models built upon hand-crafted radiomic features using Cox proportional hazards model and random survival forests, our method achieved competitive prediction performance.
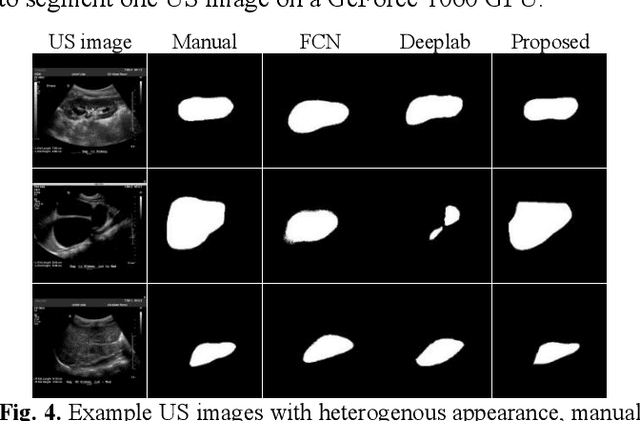
Subsequent Boundary Distance Regression and Pixelwise Classification Networks for Automatic Kidney Segmentation in Ultrasound Images
Nov 12, 2018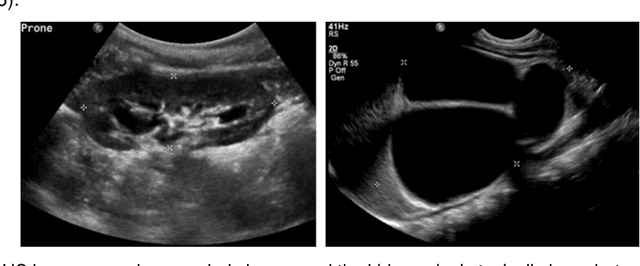
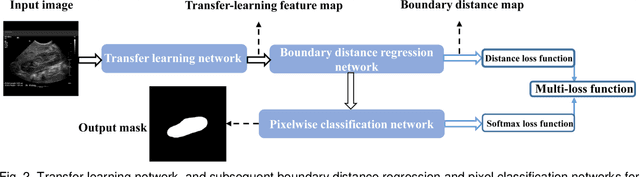
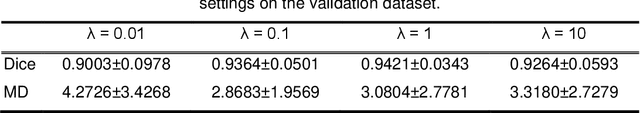
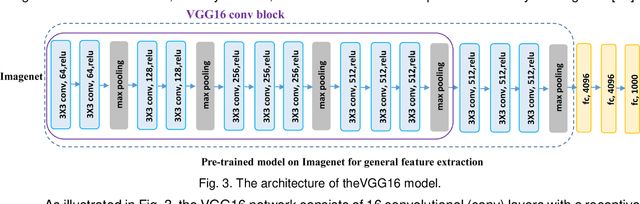
Abstract:It remains challenging to automatically segment kidneys in clinical ultrasound (US) images due to the kidneys' varied shapes and image intensity distributions, although semi-automatic methods have achieved promising performance. In this study, we propose subsequent boundary distance regression and pixel classification networks to segment the kidneys, informed by the fact that the kidney boundaries have relatively homogenous texture patterns across images. Particularly, we first use deep neural networks pre-trained for classification of natural images to extract high-level image features from US images, then these features are used as input to learn kidney boundary distance maps using a boundary distance regression network, and finally the predicted boundary distance maps are classified as kidney pixels or non-kidney pixels using a pixel classification network in an end-to-end learning fashion. We also proposed a novel data-augmentation method based on kidney shape registration to generate enriched training data from a small number of US images with manually segmented kidney labels. Experimental results have demonstrated that our method could effectively improve the performance of automatic kidney segmentation, significantly better than deep learning based pixel classification networks.
Brain decoding from functional MRI using long short-term memory recurrent neural networks
Sep 14, 2018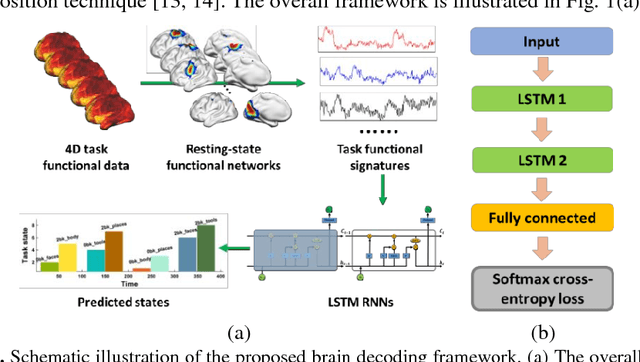
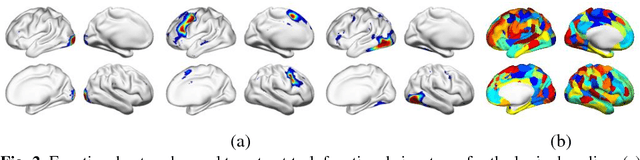
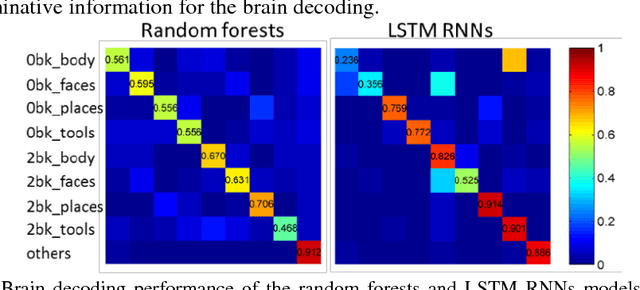
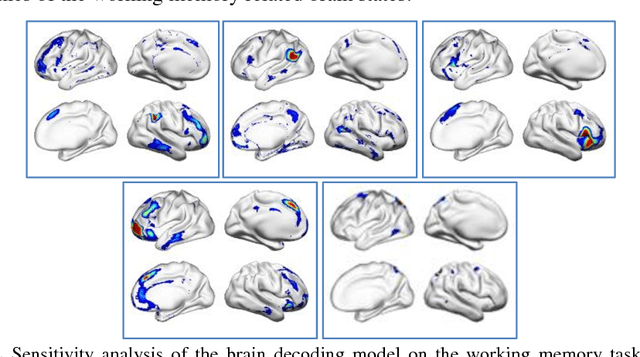
Abstract:Decoding brain functional states underlying different cognitive processes using multivariate pattern recognition techniques has attracted increasing interests in brain imaging studies. Promising performance has been achieved using brain functional connectivity or brain activation signatures for a variety of brain decoding tasks. However, most of existing studies have built decoding models upon features extracted from imaging data at individual time points or temporal windows with a fixed interval, which might not be optimal across different cognitive processes due to varying temporal durations and dependency of different cognitive processes. In this study, we develop a deep learning based framework for brain decoding by leveraging recent advances in sequence modeling using long short-term memory (LSTM) recurrent neural networks (RNNs). Particularly, functional profiles extracted from task functional imaging data based on their corresponding subject-specific intrinsic functional networks are used as features to build brain decoding models, and LSTM RNNs are adopted to learn decoding mappings between functional profiles and brain states. We evaluate the proposed method using task fMRI data from the HCP dataset, and experimental results have demonstrated that the proposed method could effectively distinguish brain states under different task events and obtain higher accuracy than conventional decoding models.
Identification of temporal transition of functional states using recurrent neural networks from functional MRI
Sep 14, 2018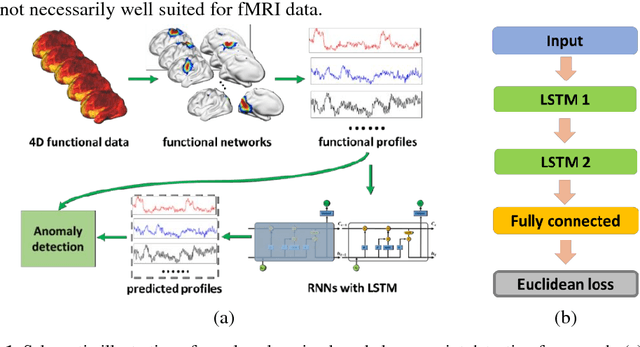
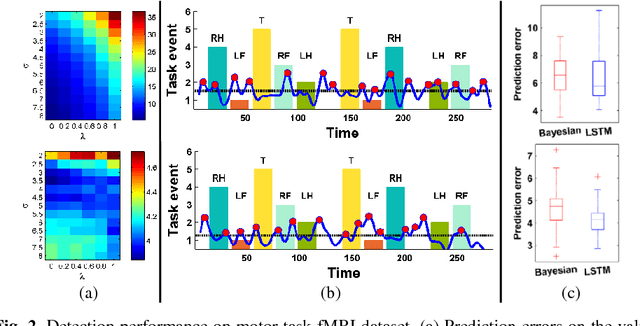
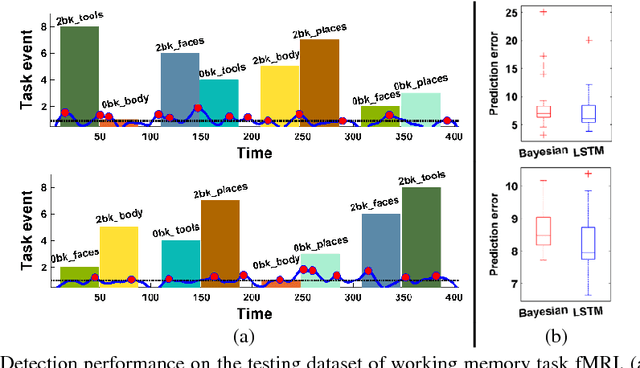
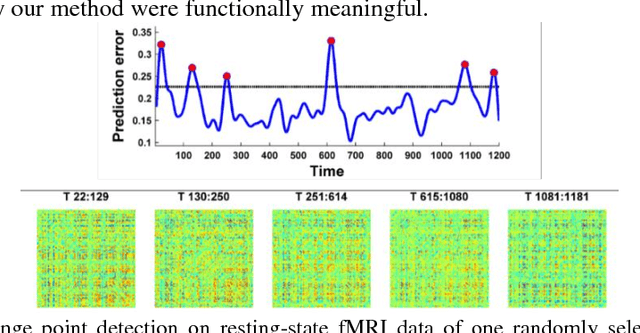
Abstract:Dynamic functional connectivity analysis provides valuable information for understanding brain functional activity underlying different cognitive processes. Besides sliding window based approaches, a variety of methods have been developed to automatically split the entire functional MRI scan into segments by detecting change points of functional signals to facilitate better characterization of temporally dynamic functional connectivity patterns. However, these methods are based on certain assumptions for the functional signals, such as Gaussian distribution, which are not necessarily suitable for the fMRI data. In this study, we develop a deep learning based framework for adaptively detecting temporally dynamic functional state transitions in a data-driven way without any explicit modeling assumptions, by leveraging recent advances in recurrent neural networks (RNNs) for sequence modeling. Particularly, we solve this problem in an anomaly detection framework with an assumption that the functional profile of one single time point could be reliably predicted based on its preceding profiles within stable functional state, while large prediction errors would occur around change points of functional states. We evaluate the proposed method using both task and resting-state fMRI data obtained from the human connectome project and experimental results have demonstrated that the proposed change point detection method could effectively identify change points between different task events and split the resting-state fMRI into segments with distinct functional connectivity patterns.
Identification of multi-scale hierarchical brain functional networks using deep matrix factorization
Sep 14, 2018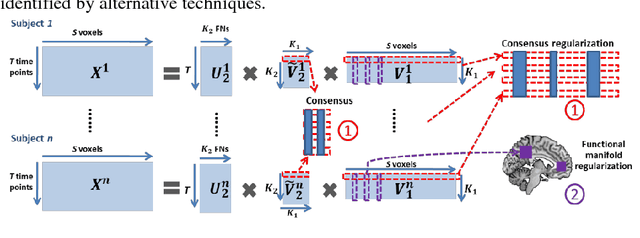
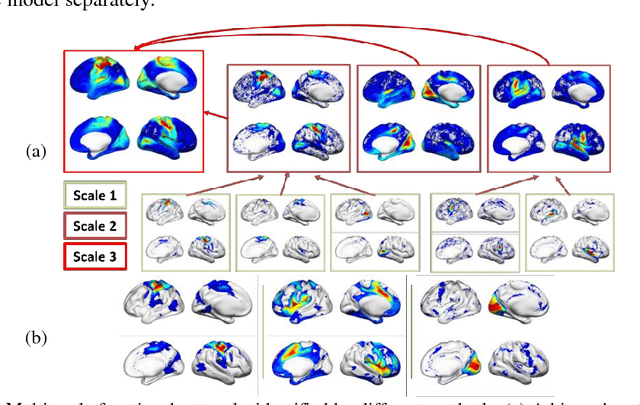
Abstract:We present a deep semi-nonnegative matrix factorization method for identifying subject-specific functional networks (FNs) at multiple spatial scales with a hierarchical organization from resting state fMRI data. Our method is built upon a deep semi-nonnegative matrix factorization framework to jointly detect the FNs at multiple scales with a hierarchical organization, enhanced by group sparsity regularization that helps identify subject-specific FNs without loss of inter-subject comparability. The proposed method has been validated for predicting subject-specific functional activations based on functional connectivity measures of the hierarchical multi-scale FNs of the same subjects. Experimental results have demonstrated that our method could obtain subject-specific multi-scale hierarchical FNs and their functional connectivity measures across different scales could better predict subject-specific functional activations than those obtained by alternative techniques.
Brain Age Prediction Based on Resting-State Functional Connectivity Patterns Using Convolutional Neural Networks
Jan 11, 2018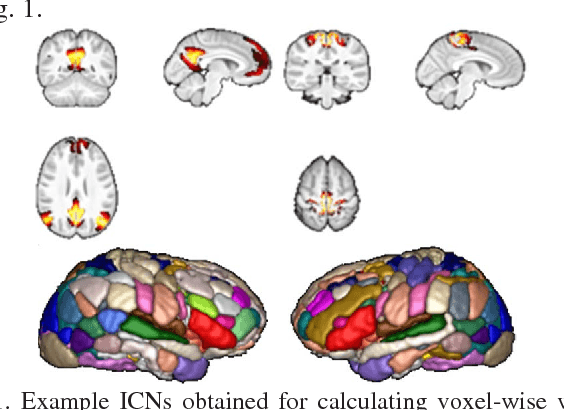
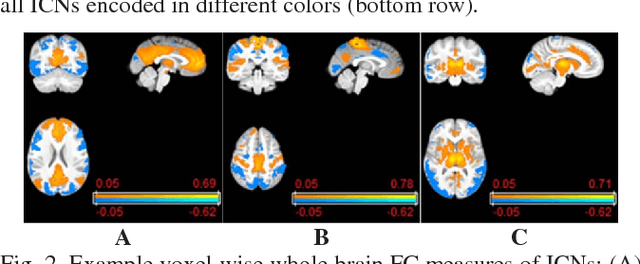
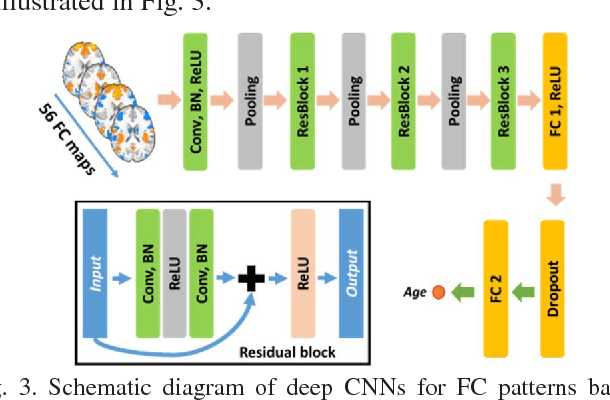
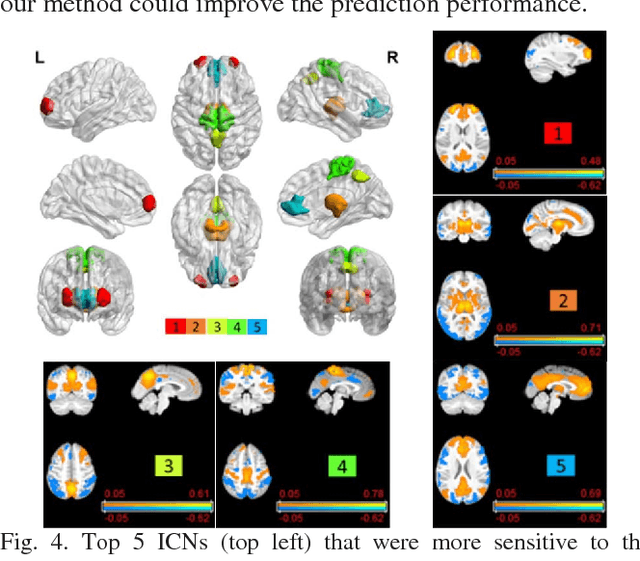
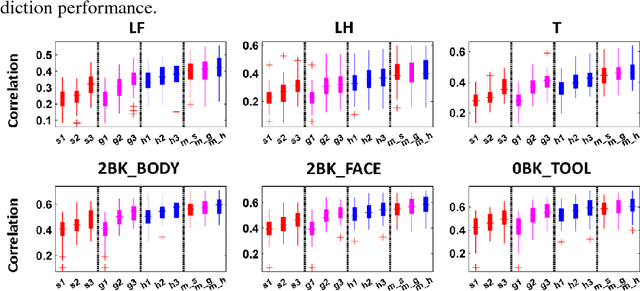
Abstract:Brain age prediction based on neuroimaging data could help characterize both the typical brain development and neuropsychiatric disorders. Pattern recognition models built upon functional connectivity (FC) measures derived from resting state fMRI (rsfMRI) data have been successfully used to predict the brain age. However, most existing studies focus on coarse-grained FC measures between brain regions or intrinsic connectivity networks (ICNs), which may sacrifice fine-grained FC information of the rsfMRI data. Whole brain voxel-wise FC measures could provide fine-grained FC information of the brain and may improve the prediction performance. In this study, we develop a deep learning method to use convolutional neural networks (CNNs) to learn informative features from the fine-grained whole brain FC measures for the brain age prediction. Experimental results on a large dataset of resting-state fMRI demonstrate that the deep learning model with fine-grained FC measures could better predict the brain age.
Non-Rigid Image Registration Using Self-Supervised Fully Convolutional Networks without Training Data
Jan 11, 2018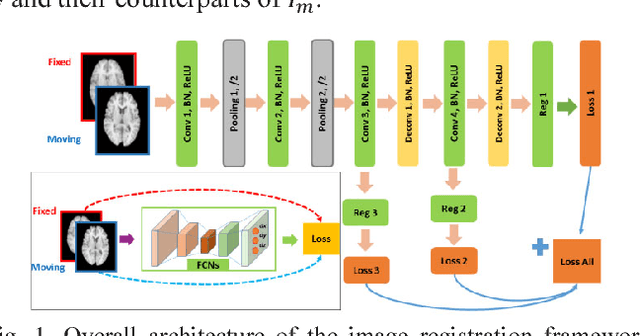
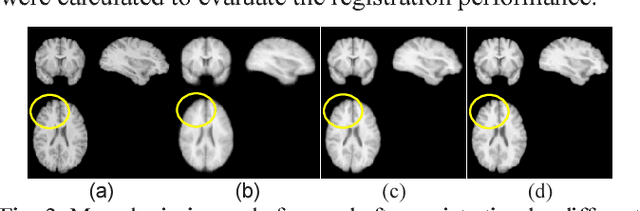
Abstract:A novel non-rigid image registration algorithm is built upon fully convolutional networks (FCNs) to optimize and learn spatial transformations between pairs of images to be registered in a self-supervised learning framework. Different from most existing deep learning based image registration methods that learn spatial transformations from training data with known corresponding spatial transformations, our method directly estimates spatial transformations between pairs of images by maximizing an image-wise similarity metric between fixed and deformed moving images, similar to conventional image registration algorithms. The image registration is implemented in a multi-resolution image registration framework to jointly optimize and learn spatial transformations and FCNs at different spatial resolutions with deep self-supervision through typical feedforward and backpropagation computation. The proposed method has been evaluated for registering 3D structural brain magnetic resonance (MR) images and obtained better performance than state-of-the-art image registration algorithms.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge