Serge Assaad
VN-Transformer: Rotation-Equivariant Attention for Vector Neurons
Jun 08, 2022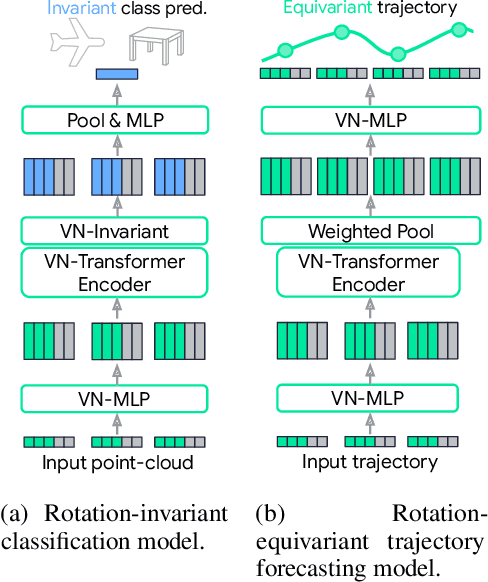
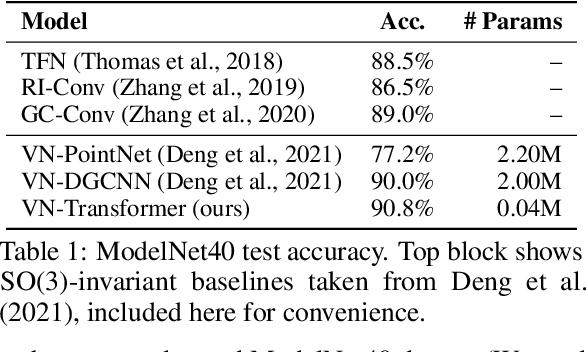

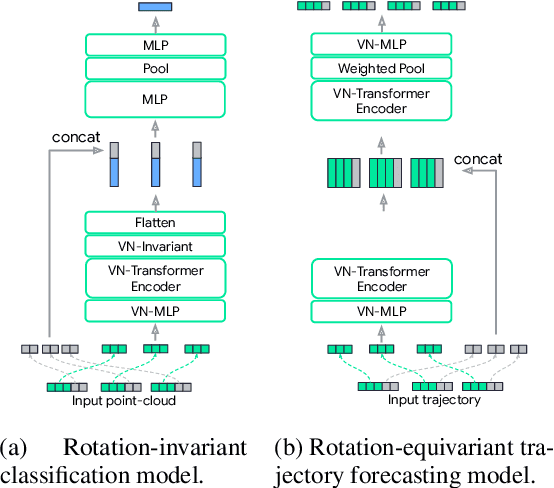
Abstract:Rotation equivariance is a desirable property in many practical applications such as motion forecasting and 3D perception, where it can offer benefits like sample efficiency, better generalization, and robustness to input perturbations. Vector Neurons (VN) is a recently developed framework offering a simple yet effective approach for deriving rotation-equivariant analogs of standard machine learning operations by extending one-dimensional scalar neurons to three-dimensional "vector neurons." We introduce a novel "VN-Transformer" architecture to address several shortcomings of the current VN models. Our contributions are: $(i)$ we derive a rotation-equivariant attention mechanism which eliminates the need for the heavy feature preprocessing required by the original Vector Neurons models; $(ii)$ we extend the VN framework to support non-spatial attributes, expanding the applicability of these models to real-world datasets; $(iii)$ we derive a rotation-equivariant mechanism for multi-scale reduction of point-cloud resolution, greatly speeding up inference and training; $(iv)$ we show that small tradeoffs in equivariance ($\epsilon$-approximate equivariance) can be used to obtain large improvements in numerical stability and training robustness on accelerated hardware, and we bound the propagation of equivariance violations in our models. Finally, we apply our VN-Transformer to 3D shape classification and motion forecasting with compelling results.
Hölder Bounds for Sensitivity Analysis in Causal Reasoning
Jul 09, 2021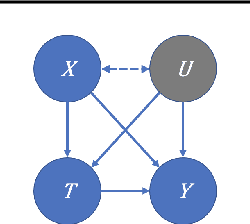

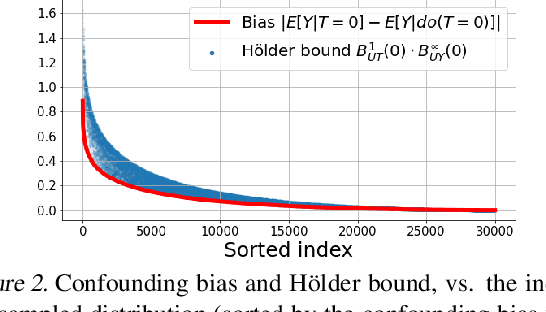
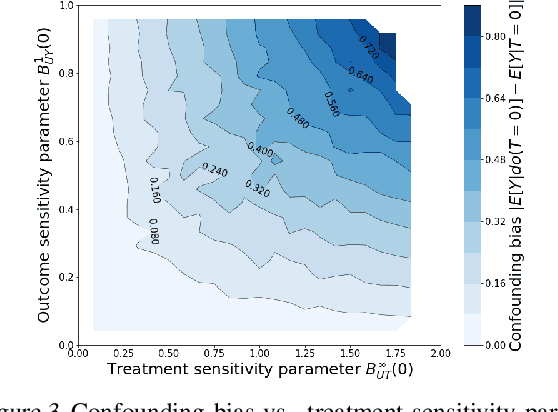
Abstract:We examine interval estimation of the effect of a treatment T on an outcome Y given the existence of an unobserved confounder U. Using H\"older's inequality, we derive a set of bounds on the confounding bias |E[Y|T=t]-E[Y|do(T=t)]| based on the degree of unmeasured confounding (i.e., the strength of the connection U->T, and the strength of U->Y). These bounds are tight either when U is independent of T or when U is independent of Y given T (when there is no unobserved confounding). We focus on a special case of this bound depending on the total variation distance between the distributions p(U) and p(U|T=t), as well as the maximum (over all possible values of U) deviation of the conditional expected outcome E[Y|U=u,T=t] from the average expected outcome E[Y|T=t]. We discuss possible calibration strategies for this bound to get interval estimates for treatment effects, and experimentally validate the bound using synthetic and semi-synthetic datasets.
Counterfactual Representation Learning with Balancing Weights
Oct 23, 2020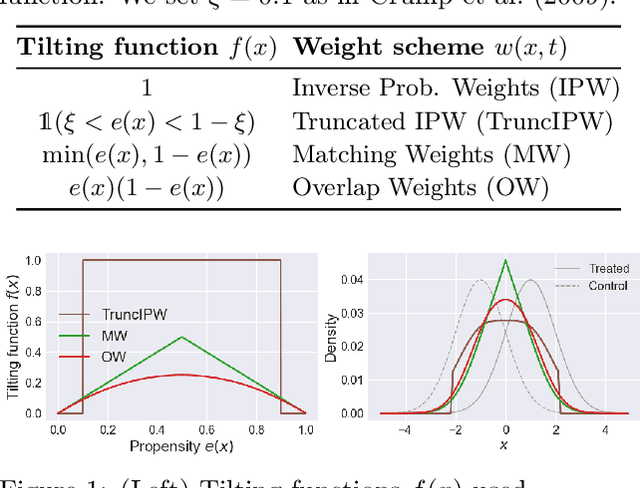
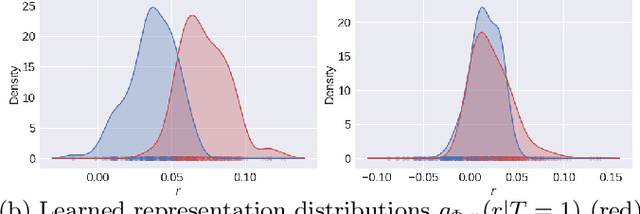
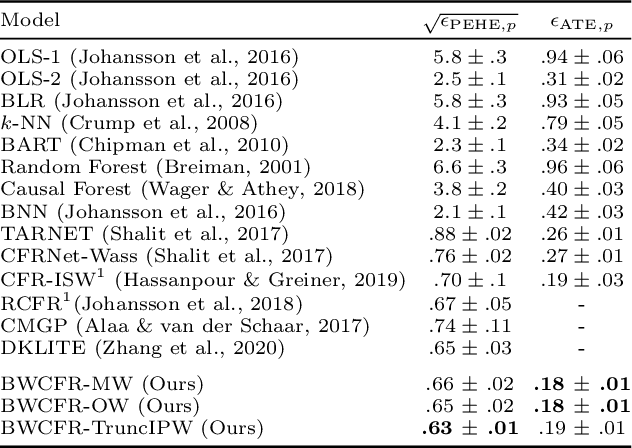
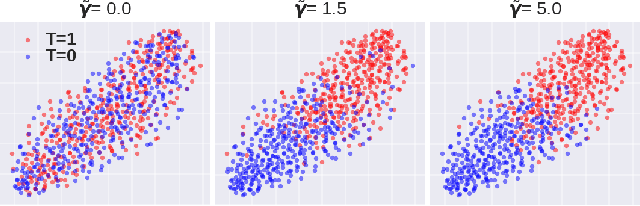
Abstract:A key to causal inference with observational data is achieving balance in predictive features associated with each treatment type. Recent literature has explored representation learning to achieve this goal. In this work, we discuss the pitfalls of these strategies - such as a steep trade-off between achieving balance and predictive power - and present a remedy via the integration of balancing weights in causal learning. Specifically, we theoretically link balance to the quality of propensity estimation, emphasize the importance of identifying a proper target population, and elaborate on the complementary roles of feature balancing and weight adjustments. Using these concepts, we then develop an algorithm for flexible, scalable and accurate estimation of causal effects. Finally, we show how the learned weighted representations may serve to facilitate alternative causal learning procedures with appealing statistical features. We conduct an extensive set of experiments on both synthetic examples and standard benchmarks, and report encouraging results relative to state-of-the-art baselines.
Double Robust Representation Learning for Counterfactual Prediction
Oct 16, 2020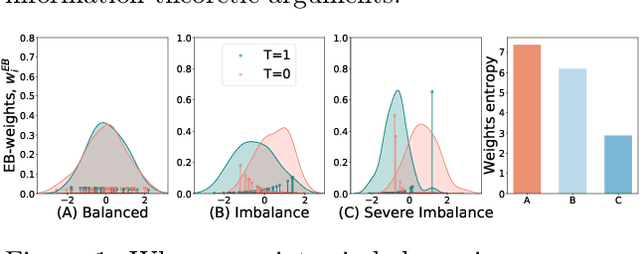
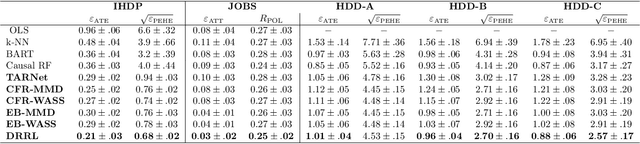
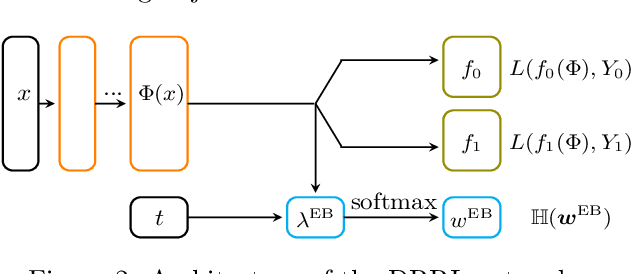

Abstract:Causal inference, or counterfactual prediction, is central to decision making in healthcare, policy and social sciences. To de-bias causal estimators with high-dimensional data in observational studies, recent advances suggest the importance of combining machine learning models for both the propensity score and the outcome function. We propose a novel scalable method to learn double-robust representations for counterfactual predictions, leading to consistent causal estimation if the model for either the propensity score or the outcome, but not necessarily both, is correctly specified. Specifically, we use the entropy balancing method to learn the weights that minimize the Jensen-Shannon divergence of the representation between the treated and control groups, based on which we make robust and efficient counterfactual predictions for both individual and average treatment effects. We provide theoretical justifications for the proposed method. The algorithm shows competitive performance with the state-of-the-art on real world and synthetic data.
Survival Analysis meets Counterfactual Inference
Jun 14, 2020
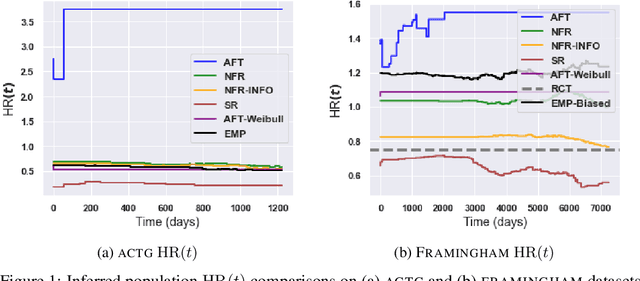
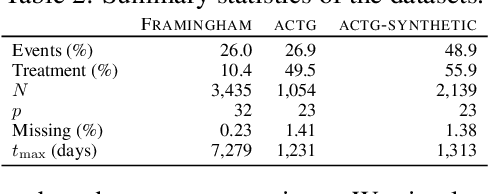
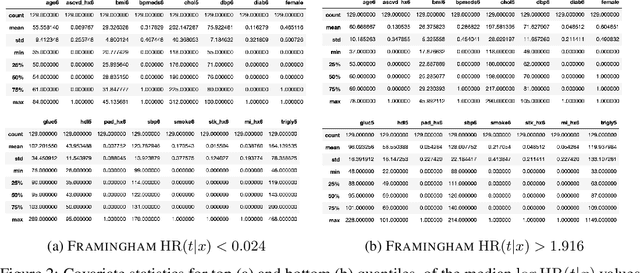
Abstract:There is growing interest in applying machine learning methods for counterfactual inference from observational data. However, approaches that account for survival outcomes are relatively limited. Survival data are frequently encountered across diverse medical applications, \textit{i.e.}, drug development, risk profiling, and clinical trials, and such data are also relevant in fields like manufacturing (for equipment monitoring). When the outcome of interest is time-to-event, special precautions for handling censored events need to be taken, as ignoring censored outcomes may lead to biased estimates. We propose a theoretically grounded unified framework for counterfactual inference applicable to survival outcomes. Further, we formulate a nonparametric hazard ratio metric for evaluating average and individualized treatment effects. Experimental results on real-world and semi-synthetic datasets, the latter which we introduce, demonstrate that the proposed approach significantly outperforms competitive alternatives in both survival-outcome predictions and treatment-effect estimation.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge