Junsu Ko
IgPose: A Generative Data-Augmented Pipeline for Robust Immunoglobulin-Antigen Binding Prediction
Mar 16, 2026Abstract:Predicting immunoglobulin-antigen (Ig-Ag) binding remains a significant challenge due to the paucity of experimentally-resolved complexes and the limited accuracy of de novo Ig structure prediction. We introduce IgPose, a generalizable framework for Ig-Ag pose identification and scoring, built on a generative data-augmentation pipeline. To mitigate data scarcity, we constructed the Structural Immunoglobulin Decoy Database (SIDD), a comprehensive repository of high-fidelity synthetic decoys. IgPose integrates equivariant graph neural networks, ESM-2 embeddings, and gated recurrent units to synergistically capture both geometric and evolutionary features. We implemented interface-focused k-hop sampling with biologically guided pooling to enhance generalization across diverse interfaces. The framework comprises two sub-networks--IgPoseClassifier for binding pose discrimination and IgPoseScore for DockQ score estimation--and achieves robust performance on curated internal test sets and the CASP-16 benchmark compared to physics and deep learning baselines. IgPose serves as a versatile computational tool for high-throughput antibody discovery pipelines by providing accurate pose filtering and ranking. IgPose is available on GitHub (https://github.com/arontier/igpose).
* 11 pages, 4 figures, Bioinformatics
Deep Learning Methods for Lung Cancer Segmentation in Whole-slide Histopathology Images -- the ACDC@LungHP Challenge 2019
Aug 21, 2020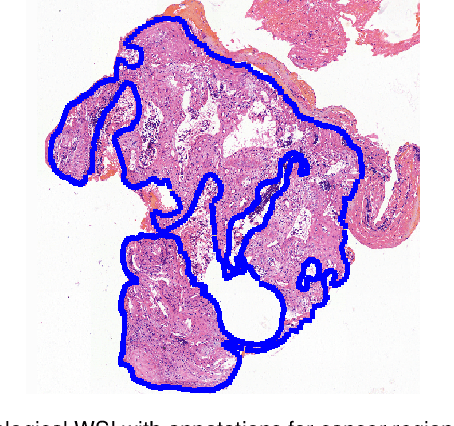
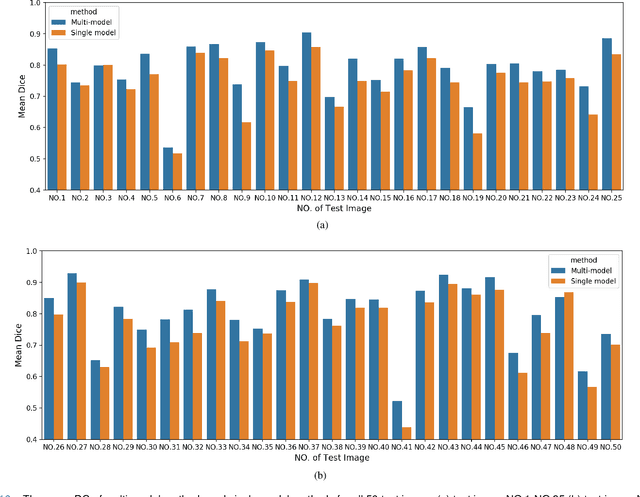
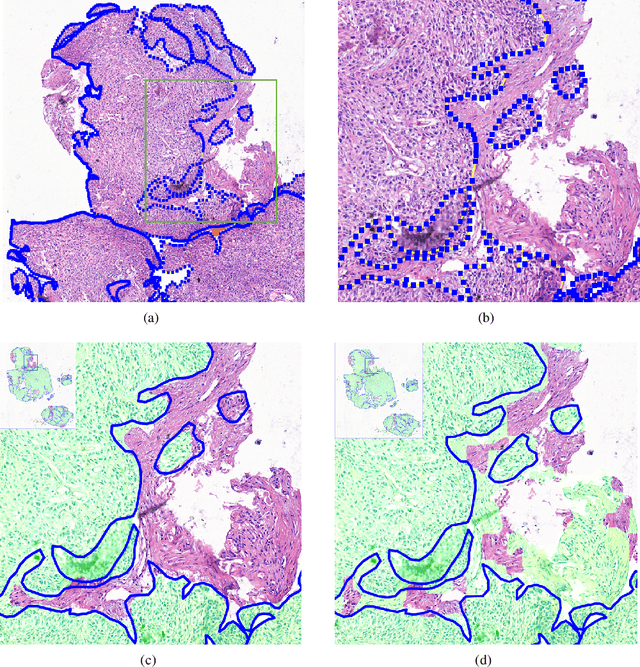

Abstract:Accurate segmentation of lung cancer in pathology slides is a critical step in improving patient care. We proposed the ACDC@LungHP (Automatic Cancer Detection and Classification in Whole-slide Lung Histopathology) challenge for evaluating different computer-aided diagnosis (CADs) methods on the automatic diagnosis of lung cancer. The ACDC@LungHP 2019 focused on segmentation (pixel-wise detection) of cancer tissue in whole slide imaging (WSI), using an annotated dataset of 150 training images and 50 test images from 200 patients. This paper reviews this challenge and summarizes the top 10 submitted methods for lung cancer segmentation. All methods were evaluated using the false positive rate, false negative rate, and DICE coefficient (DC). The DC ranged from 0.7354$\pm$0.1149 to 0.8372$\pm$0.0858. The DC of the best method was close to the inter-observer agreement (0.8398$\pm$0.0890). All methods were based on deep learning and categorized into two groups: multi-model method and single model method. In general, multi-model methods were significantly better ($\textit{p}$<$0.01$) than single model methods, with mean DC of 0.7966 and 0.7544, respectively. Deep learning based methods could potentially help pathologists find suspicious regions for further analysis of lung cancer in WSI.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge