Jiancong Hu
A Comprehensive Evaluation of Multi-Modal Large Language Models for Endoscopy Analysis
May 29, 2025
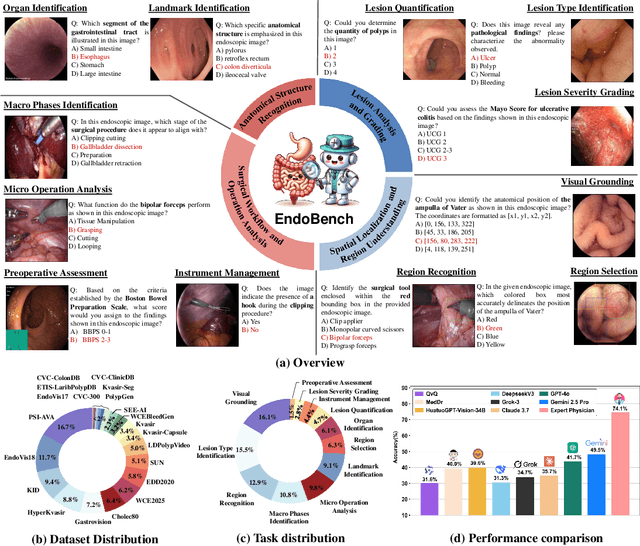


Abstract:Endoscopic procedures are essential for diagnosing and treating internal diseases, and multi-modal large language models (MLLMs) are increasingly applied to assist in endoscopy analysis. However, current benchmarks are limited, as they typically cover specific endoscopic scenarios and a small set of clinical tasks, failing to capture the real-world diversity of endoscopic scenarios and the full range of skills needed in clinical workflows. To address these issues, we introduce EndoBench, the first comprehensive benchmark specifically designed to assess MLLMs across the full spectrum of endoscopic practice with multi-dimensional capacities. EndoBench encompasses 4 distinct endoscopic scenarios, 12 specialized clinical tasks with 12 secondary subtasks, and 5 levels of visual prompting granularities, resulting in 6,832 rigorously validated VQA pairs from 21 diverse datasets. Our multi-dimensional evaluation framework mirrors the clinical workflow--spanning anatomical recognition, lesion analysis, spatial localization, and surgical operations--to holistically gauge the perceptual and diagnostic abilities of MLLMs in realistic scenarios. We benchmark 23 state-of-the-art models, including general-purpose, medical-specialized, and proprietary MLLMs, and establish human clinician performance as a reference standard. Our extensive experiments reveal: (1) proprietary MLLMs outperform open-source and medical-specialized models overall, but still trail human experts; (2) medical-domain supervised fine-tuning substantially boosts task-specific accuracy; and (3) model performance remains sensitive to prompt format and clinical task complexity. EndoBench establishes a new standard for evaluating and advancing MLLMs in endoscopy, highlighting both progress and persistent gaps between current models and expert clinical reasoning. We publicly release our benchmark and code.
Polyp-Gen: Realistic and Diverse Polyp Image Generation for Endoscopic Dataset Expansion
Jan 29, 2025



Abstract:Automated diagnostic systems (ADS) have shown significant potential in the early detection of polyps during endoscopic examinations, thereby reducing the incidence of colorectal cancer. However, due to high annotation costs and strict privacy concerns, acquiring high-quality endoscopic images poses a considerable challenge in the development of ADS. Despite recent advancements in generating synthetic images for dataset expansion, existing endoscopic image generation algorithms failed to accurately generate the details of polyp boundary regions and typically required medical priors to specify plausible locations and shapes of polyps, which limited the realism and diversity of the generated images. To address these limitations, we present Polyp-Gen, the first full-automatic diffusion-based endoscopic image generation framework. Specifically, we devise a spatial-aware diffusion training scheme with a lesion-guided loss to enhance the structural context of polyp boundary regions. Moreover, to capture medical priors for the localization of potential polyp areas, we introduce a hierarchical retrieval-based sampling strategy to match similar fine-grained spatial features. In this way, our Polyp-Gen can generate realistic and diverse endoscopic images for building reliable ADS. Extensive experiments demonstrate the state-of-the-art generation quality, and the synthetic images can improve the downstream polyp detection task. Additionally, our Polyp-Gen has shown remarkable zero-shot generalizability on other datasets. The source code is available at https://github.com/CUHK-AIM-Group/Polyp-Gen.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge