Yidong Zhao
Set-Based Groupwise Registration for Variable-Length, Variable-Contrast Cardiac MRI
May 11, 2026Abstract:Quantitative cardiac magnetic resonance imaging (MRI) enables non-invasive myocardial tissue characterization but relies on robust motion correction within these variable-length, variable-contrast image sequences. Groupwise registration, which simultaneously aligns all images, has shown greater robustness than pairwise registration for motion correction. However, current deep-learning-based groupwise registration methods cannot generalize across MRI sequences: the architecture typically encodes input data as a fixed-length channel stack, which rigidly couples network design to protocol-specific sequence length, input ordering, and contrast dynamics. At inference time, any change in imaging protocols will render the network unusable. In this work, we introduce \emph{\AnyTwoReg}, a new set-based groupwise registration framework that takes a quantitative MRI sequence as an unordered set. This set formulation fundamentally decouples network design from sequence length and input ordering. By utilizing a shared encoder and correlation-guided feature aggregation, \emph{\AnyTwoReg} constructs a permutation-invariant canonical reference for registration, and learns a permutation-equivariant mapping from images to deformation fields. Additionally, we extract contrast-insensitive image features from an existing foundation model to handle extreme contrast variations. Trained exclusively on a single public $T_1$ mapping dataset (STONE, sequence length $L=11$), \AnyTwoReg generalizes to two unseen quantitative MRI datasets (MOLLI, ASL) with variable lengths ($L \in [11, 60]$) and different contrast dynamics. It achieves strong cross-protocol generalization in a zero-shot manner, and consistently improves downstream quantitative mapping quality. Notably, while designed for quantitative MRI sequences, our framework is directly applicable to Cine MRI sequences for inter-cardiac-phase registration.
Adapting Frozen Mono-modal Backbones for Multi-modal Registration via Contrast-Agnostic Instance Optimization
Mar 27, 2026Abstract:Deformable image registration remains a central challenge in medical image analysis, particularly under multi-modal scenarios where intensity distributions vary significantly across scans. While deep learning methods provide efficient feed-forward predictions, they often fail to generalize robustly under distribution shifts at test time. A straightforward remedy is full network fine-tuning, yet for modern architectures such as Transformers or deep U-Nets, this adaptation is prohibitively expensive in both memory and runtime when operating in 3D. Meanwhile, the naive fine-tuning struggles more with potential degradation in performance in the existence of drastic domain shifts. In this work, we propose a registration framework that integrates a frozen pretrained \textbf{mono-modal} registration model with a lightweight adaptation pipeline for \textbf{multi-modal} image registration. Specifically, we employ style transfer based on contrast-agnostic representation generation and refinement modules to bridge modality and domain gaps with instance optimization at test time. This design is orthogonal to the choice of backbone mono-modal model, thus avoids the computational burden of full fine-tuning while retaining the flexibility to adapt to unseen domains. We evaluate our approach on the Learn2Reg 2025 LUMIR validation set and observe consistent improvements over the pretrained state-of-the-art mono-modal backbone. In particular, the method ranks second on the multi-modal subset, third on the out-of-domain subset, and achieves fourth place overall in Dice score. These results demonstrate that combining frozen mono-modal models with modality adaptation and lightweight instance optimization offers an effective and practical pathway toward robust multi-modal registration.
Improving Instance Optimization in Deformable Image Registration with Gradient Projection
Oct 21, 2024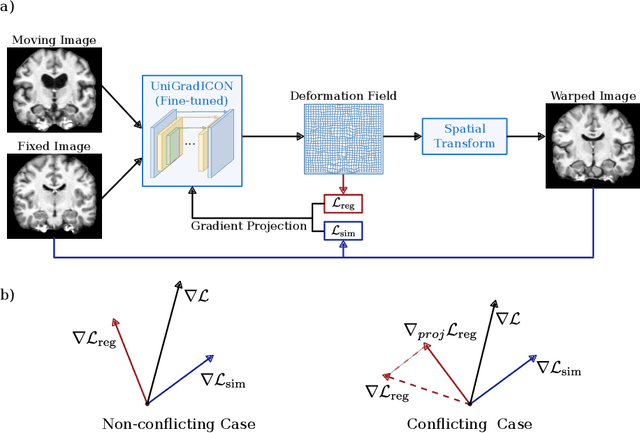
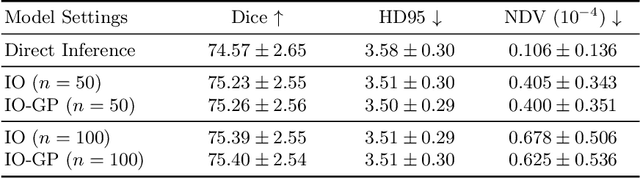
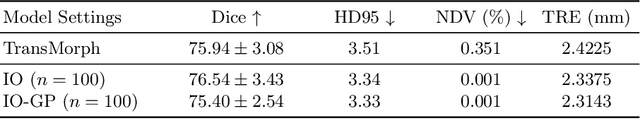
Abstract:Deformable image registration is inherently a multi-objective optimization (MOO) problem, requiring a delicate balance between image similarity and deformation regularity. These conflicting objectives often lead to poor optimization outcomes, such as being trapped in unsatisfactory local minima or experiencing slow convergence. Deep learning methods have recently gained popularity in this domain due to their efficiency in processing large datasets and achieving high accuracy. However, they often underperform during test time compared to traditional optimization techniques, which further explore iterative, instance-specific gradient-based optimization. This performance gap is more pronounced when a distribution shift between training and test data exists. To address this issue, we focus on the instance optimization (IO) paradigm, which involves additional optimization for test-time instances based on a pre-trained model. IO effectively combines the generalization capabilities of deep learning with the fine-tuning advantages of instance-specific optimization. Within this framework, we emphasize the use of gradient projection to mitigate conflicting updates in MOO. This technique projects conflicting gradients into a common space, better aligning the dual objectives and enhancing optimization stability. We validate our method using a state-of-the-art foundation model on the 3D Brain inter-subject registration task (LUMIR) from the Learn2Reg 2024 Challenge. Our results show significant improvements over standard gradient descent, leading to more accurate and reliable registration results.
Lost in Tracking: Uncertainty-guided Cardiac Cine MRI Segmentation at Right Ventricle Base
Oct 04, 2024Abstract:Accurate biventricular segmentation of cardiac magnetic resonance (CMR) cine images is essential for the clinical evaluation of heart function. However, compared to left ventricle (LV), right ventricle (RV) segmentation is still more challenging and less reproducible. Degenerate performance frequently occurs at the RV base, where the in-plane anatomical structures are complex (with atria, valve, and aorta) and vary due to the strong interplanar motion. In this work, we propose to address the currently unsolved issues in CMR segmentation, specifically at the RV base, with two strategies: first, we complemented the public resource by reannotating the RV base in the ACDC dataset, with refined delineation of the right ventricle outflow tract (RVOT), under the guidance of an expert cardiologist. Second, we proposed a novel dual encoder U-Net architecture that leverages temporal incoherence to inform the segmentation when interplanar motions occur. The inter-planar motion is characterized by loss-of-tracking, via Bayesian uncertainty of a motion-tracking model. Our experiments showed that our method significantly improved RV base segmentation taking into account temporal incoherence. Furthermore, we investigated the reproducibility of deep learning-based segmentation and showed that the combination of consistent annotation and loss of tracking could enhance the reproducibility of RV segmentation, potentially facilitating a large number of clinical studies focusing on RV.
Deep-learning-based groupwise registration for motion correction of cardiac $T_1$ mapping
Jun 21, 2024Abstract:Quantitative $T_1$ mapping by MRI is an increasingly important tool for clinical assessment of cardiovascular diseases. The cardiac $T_1$ map is derived by fitting a known signal model to a series of baseline images, while the quality of this map can be deteriorated by involuntary respiratory and cardiac motion. To correct motion, a template image is often needed to register all baseline images, but the choice of template is nontrivial, leading to inconsistent performance sensitive to image contrast. In this work, we propose a novel deep-learning-based groupwise registration framework, which omits the need for a template, and registers all baseline images simultaneously. We design two groupwise losses for this registration framework: the first is a linear principal component analysis (PCA) loss that enforces alignment of baseline images irrespective of the intensity variation, and the second is an auxiliary relaxometry loss that enforces adherence of intensity profile to the signal model. We extensively evaluated our method, termed ``PCA-Relax'', and other baseline methods on an in-house cardiac MRI dataset including both pre- and post-contrast $T_1$ sequences. All methods were evaluated under three distinct training-and-evaluation strategies, namely, standard, one-shot, and test-time-adaptation. The proposed PCA-Relax showed further improved performance of registration and mapping over well-established baselines. The proposed groupwise framework is generic and can be adapted to applications involving multiple images.
Recurrent Inference Machine for Medical Image Registration
Jun 19, 2024Abstract:Image registration is essential for medical image applications where alignment of voxels across multiple images is needed for qualitative or quantitative analysis. With recent advancements in deep neural networks and parallel computing, deep learning-based medical image registration methods become competitive with their flexible modelling and fast inference capabilities. However, compared to traditional optimization-based registration methods, the speed advantage may come at the cost of registration performance at inference time. Besides, deep neural networks ideally demand large training datasets while optimization-based methods are training-free. To improve registration accuracy and data efficiency, we propose a novel image registration method, termed Recurrent Inference Image Registration (RIIR) network. RIIR is formulated as a meta-learning solver to the registration problem in an iterative manner. RIIR addresses the accuracy and data efficiency issues, by learning the update rule of optimization, with implicit regularization combined with explicit gradient input. We evaluated RIIR extensively on brain MRI and quantitative cardiac MRI datasets, in terms of both registration accuracy and training data efficiency. Our experiments showed that RIIR outperformed a range of deep learning-based methods, even with only $5\%$ of the training data, demonstrating high data efficiency. Key findings from our ablation studies highlighted the important added value of the hidden states introduced in the recurrent inference framework for meta-learning. Our proposed RIIR offers a highly data-efficient framework for deep learning-based medical image registration.
PCA-Relax: Deep-learning-based groupwise registration for motion correction of cardiac $T_1$ mapping
Jun 18, 2024Abstract:Quantitative MRI (qMRI) is an increasingly important tool for clinical assessment of cardiovascular diseases. Quantitative maps are derived by fitting a known signal model to a series of baseline images, while the quality of the map can be deteriorated by involuntary respiratory and cardiac motion. To correct motion, a template image is often needed to register all baseline images, but the choice of template is nontrivial, leading to inconsistent performance sensitive to image contrast. In this work, we propose a novel deep-learning-based groupwise registration framework, which omits the need for a template, and registers all baseline images simultaneously. We design two groupwise losses for this registration framework: the first is a linear principal component analysis (PCA) loss that enforces alignment of baseline images irrespective of the intensity variation, and the second is an auxiliary relaxometry loss that enforces adherence of intensity profile to the signal model. We extensively evaluated our method, termed ``PCA-Relax'', and other baseline methods on an in-house cardiac MRI dataset including both pre- and post-contrast $T_1$ sequences. All methods were evaluated under three distinct training-and-evaluation strategies, namely, standard, one-shot, and test-time-adaptation. The proposed PCA-Relax showed further improved performance of registration and mapping over well-established baselines. The proposed groupwise framework is generic and can be adapted to applications involving multiple images.
Bayesian Uncertainty Estimation by Hamiltonian Monte Carlo: Applications to Cardiac MRI Segmentation
Mar 04, 2024Abstract:Deep learning (DL)-based methods have achieved state-of-the-art performance for a wide range of medical image segmentation tasks. Nevertheless, recent studies show that deep neural networks (DNNs) can be miscalibrated and overconfident, leading to "silent failures" that are risky} for clinical applications. Bayesian statistics provide an intuitive approach to DL failure detection, based on posterior probability estimation. However, Bayesian DL, and in particular the posterior estimation, is intractable for large medical image segmentation DNNs. To tackle this challenge, we propose a Bayesian learning framework by Hamiltonian Monte Carlo (HMC), tempered by cold posterior (CP) to accommodate medical data augmentation, named HMC-CP. For HMC computation, we further propose a cyclical annealing strategy, which captures both local and global geometries of the posterior distribution, enabling highly efficient Bayesian DNN training with the same computational budget requirements as training a single DNN. The resulting Bayesian DNN outputs an ensemble segmentation along with the segmentation uncertainty. We evaluate the proposed HMC-CP extensively on cardiac magnetic resonance image (MRI) segmentation, using in-domain steady-state free precession (SSFP) cine images as well as out-of-domain datasets of quantitative $T_1$ and $T_2$ mapping.
Relaxometry Guided Quantitative Cardiac Magnetic Resonance Image Reconstruction
Mar 01, 2024Abstract:Deep learning-based methods have achieved prestigious performance for magnetic resonance imaging (MRI) reconstruction, enabling fast imaging for many clinical applications. Previous methods employ convolutional networks to learn the image prior as the regularization term. In quantitative MRI, the physical model of nuclear magnetic resonance relaxometry is known, providing additional prior knowledge for image reconstruction. However, traditional reconstruction networks are limited to learning the spatial domain prior knowledge, ignoring the relaxometry prior. Therefore, we propose a relaxometry-guided quantitative MRI reconstruction framework to learn the spatial prior from data and the relaxometry prior from MRI physics. Additionally, we also evaluated the performance of two popular reconstruction backbones, namely, recurrent variational networks (RVN) and variational networks (VN) with U- Net. Experiments demonstrate that the proposed method achieves highly promising results in quantitative MRI reconstruction.
Contrast-Agnostic Groupwise Registration by Robust PCA for Quantitative Cardiac MRI
Nov 03, 2023



Abstract:Quantitative cardiac magnetic resonance imaging (MRI) is an increasingly important diagnostic tool for cardiovascular diseases. Yet, co-registration of all baseline images within the quantitative MRI sequence is essential for the accuracy and precision of quantitative maps. However, co-registering all baseline images from a quantitative cardiac MRI sequence remains a nontrivial task because of the simultaneous changes in intensity and contrast, in combination with cardiac and respiratory motion. To address the challenge, we propose a novel motion correction framework based on robust principle component analysis (rPCA) that decomposes quantitative cardiac MRI into low-rank and sparse components, and we integrate the groupwise CNN-based registration backbone within the rPCA framework. The low-rank component of rPCA corresponds to the quantitative mapping (i.e. limited degree of freedom in variation), while the sparse component corresponds to the residual motion, making it easier to formulate and solve the groupwise registration problem. We evaluated our proposed method on cardiac T1 mapping by the modified Look-Locker inversion recovery (MOLLI) sequence, both before and after the Gadolinium contrast agent administration. Our experiments showed that our method effectively improved registration performance over baseline methods without introducing rPCA, and reduced quantitative mapping error in both in-domain (pre-contrast MOLLI) and out-of-domain (post-contrast MOLLI) inference. The proposed rPCA framework is generic and can be integrated with other registration backbones.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge