Stefan Klein
Department of Radiology and Nuclear Medicine, Erasmus MC Cancer Institute, University Medical Center Rotterdam, Rotterdam, the Netherlands
An Interpretable Machine Learning Model with Deep Learning-based Imaging Biomarkers for Diagnosis of Alzheimer's Disease
Aug 15, 2023



Abstract:Machine learning methods have shown large potential for the automatic early diagnosis of Alzheimer's Disease (AD). However, some machine learning methods based on imaging data have poor interpretability because it is usually unclear how they make their decisions. Explainable Boosting Machines (EBMs) are interpretable machine learning models based on the statistical framework of generalized additive modeling, but have so far only been used for tabular data. Therefore, we propose a framework that combines the strength of EBM with high-dimensional imaging data using deep learning-based feature extraction. The proposed framework is interpretable because it provides the importance of each feature. We validated the proposed framework on the Alzheimer's Disease Neuroimaging Initiative (ADNI) dataset, achieving accuracy of 0.883 and area-under-the-curve (AUC) of 0.970 on AD and control classification. Furthermore, we validated the proposed framework on an external testing set, achieving accuracy of 0.778 and AUC of 0.887 on AD and subjective cognitive decline (SCD) classification. The proposed framework significantly outperformed an EBM model using volume biomarkers instead of deep learning-based features, as well as an end-to-end convolutional neural network (CNN) with optimized architecture.
Comparison of different retinal regions-of-interest imaged by OCT for the classification of intermediate AMD
May 04, 2023



Abstract:To study whether it is possible to differentiate intermediate age-related macular degeneration (AMD) from healthy controls using partial optical coherence tomography (OCT) data, that is, restricting the input B-scans to certain pre-defined regions of interest (ROIs). A total of 15744 B-scans from 269 intermediate AMD patients and 115 normal subjects were used in this study (split on subject level in 80% train, 10% validation and 10% test). From each OCT B-scan, three ROIs were extracted: retina, complex between retinal pigment epithelium (RPE) and Bruch membrane (BM), and choroid (CHO). These ROIs were obtained using two different methods: masking and cropping. In addition to the six ROIs, the whole OCT B-scan and the binary mask corresponding to the segmentation of the RPE-BM complex were used. For each subset, a convolutional neural network (based on VGG16 architecture and pre-trained on ImageNet) was trained and tested. The performance of the models was evaluated using the area under the receiver operating characteristic (AUROC), accuracy, sensitivity, and specificity. All trained models presented an AUROC, accuracy, sensitivity, and specificity equal to or higher than 0.884, 0.816, 0.685, and 0.644, respectively. The model trained on the whole OCT B-scan presented the best performance (AUROC = 0.983, accuracy = 0.927, sensitivity = 0.862, specificity = 0.913). The models trained on the ROIs obtained with the cropping method led to significantly higher outcomes than those obtained with masking, with the exception of the retinal tissue, where no statistically significant difference was observed between cropping and masking (p = 0.47). This study demonstrated that while using the complete OCT B-scan provided the highest accuracy in classifying intermediate AMD, models trained on specific ROIs such as the RPE-BM complex or the choroid can still achieve high performance.
Computer-aided diagnosis and prediction in brain disorders
Jun 29, 2022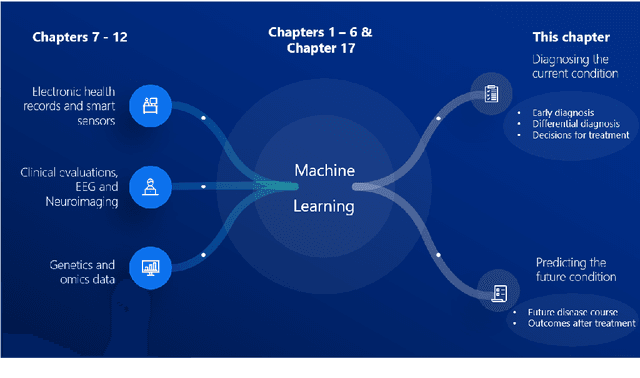
Abstract:Computer-aided methods have shown added value for diagnosing and predicting brain disorders and can thus support decision making in clinical care and treatment planning. This chapter will provide insight into the type of methods, their working, their input data - such as cognitive tests, imaging and genetic data - and the types of output they provide. We will focus on specific use cases for diagnosis, i.e. estimating the current 'condition' of the patient, such as early detection and diagnosis of dementia, differential diagnosis of brain tumours, and decision making in stroke. Regarding prediction, i.e. estimation of the future 'condition' of the patient, we will zoom in on use cases such as predicting the disease course in multiple sclerosis and predicting patient outcomes after treatment in brain cancer. Furthermore, based on these use cases, we will assess the current state-of-the-art methodology and highlight current efforts on benchmarking of these methods and the importance of open science therein. Finally, we assess the current clinical impact of computer-aided methods and discuss the required next steps to increase clinical impact.
Federated Learning Enables Big Data for Rare Cancer Boundary Detection
Apr 25, 2022Abstract:Although machine learning (ML) has shown promise in numerous domains, there are concerns about generalizability to out-of-sample data. This is currently addressed by centrally sharing ample, and importantly diverse, data from multiple sites. However, such centralization is challenging to scale (or even not feasible) due to various limitations. Federated ML (FL) provides an alternative to train accurate and generalizable ML models, by only sharing numerical model updates. Here we present findings from the largest FL study to-date, involving data from 71 healthcare institutions across 6 continents, to generate an automatic tumor boundary detector for the rare disease of glioblastoma, utilizing the largest dataset of such patients ever used in the literature (25,256 MRI scans from 6,314 patients). We demonstrate a 33% improvement over a publicly trained model to delineate the surgically targetable tumor, and 23% improvement over the tumor's entire extent. We anticipate our study to: 1) enable more studies in healthcare informed by large and diverse data, ensuring meaningful results for rare diseases and underrepresented populations, 2) facilitate further quantitative analyses for glioblastoma via performance optimization of our consensus model for eventual public release, and 3) demonstrate the effectiveness of FL at such scale and task complexity as a paradigm shift for multi-site collaborations, alleviating the need for data sharing.
Ten years of image analysis and machine learning competitions in dementia
Dec 15, 2021
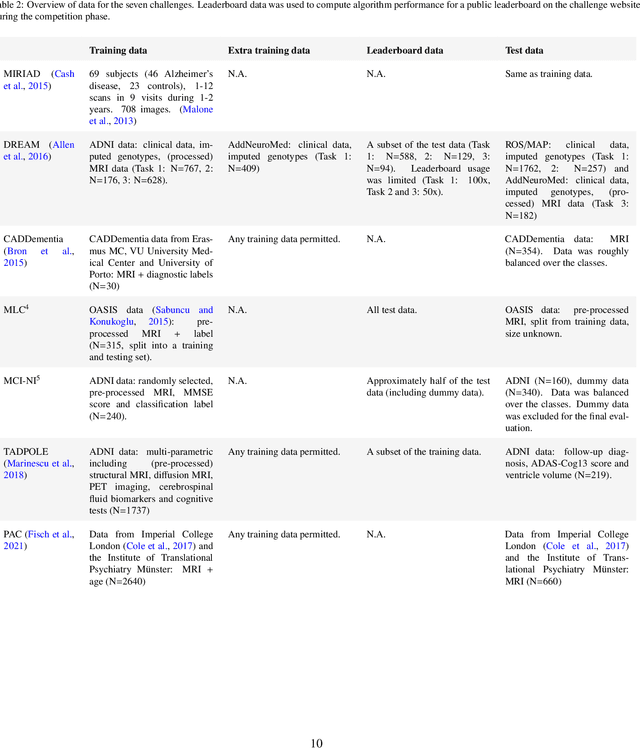
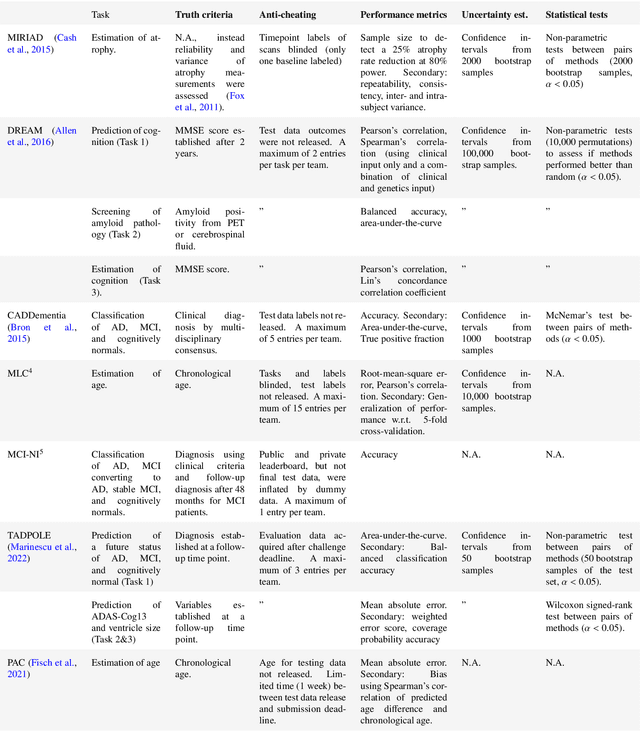
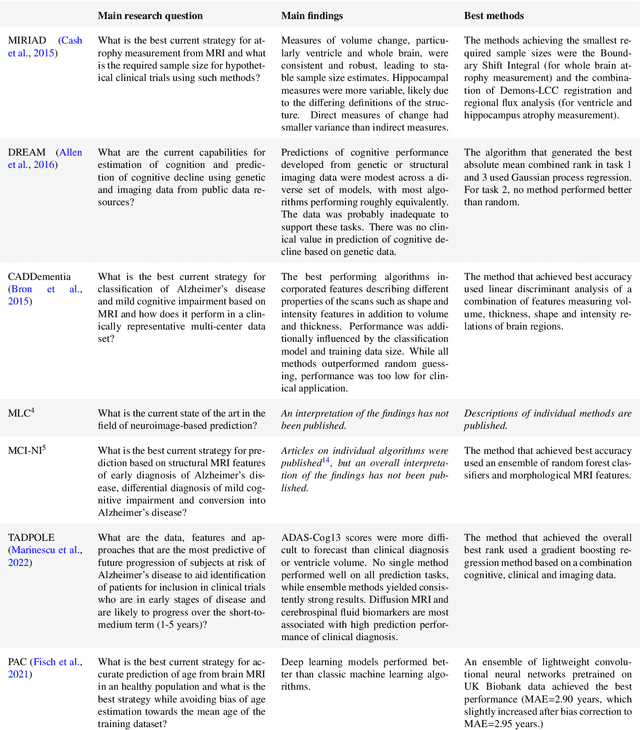
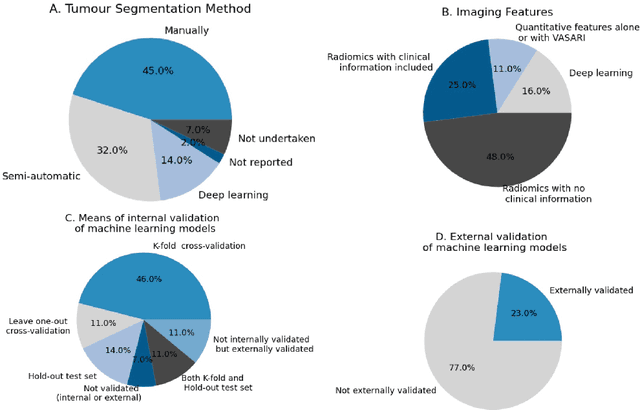
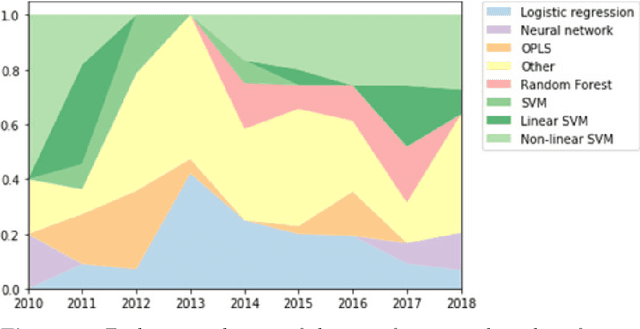
Abstract:Machine learning methods exploiting multi-parametric biomarkers, especially based on neuroimaging, have huge potential to improve early diagnosis of dementia and to predict which individuals are at-risk of developing dementia. To benchmark algorithms in the field of machine learning and neuroimaging in dementia and assess their potential for use in clinical practice and clinical trials, seven grand challenges have been organized in the last decade: MIRIAD, Alzheimer's Disease Big Data DREAM, CADDementia, Machine Learning Challenge, MCI Neuroimaging, TADPOLE, and the Predictive Analytics Competition. Based on two challenge evaluation frameworks, we analyzed how these grand challenges are complementing each other regarding research questions, datasets, validation approaches, results and impact. The seven grand challenges addressed questions related to screening, diagnosis, prediction and monitoring in (pre-clinical) dementia. There was little overlap in clinical questions, tasks and performance metrics. Whereas this has the advantage of providing insight on a broad range of questions, it also limits the validation of results across challenges. In general, winning algorithms performed rigorous data pre-processing and combined a wide range of input features. Despite high state-of-the-art performances, most of the methods evaluated by the challenges are not clinically used. To increase impact, future challenges could pay more attention to statistical analysis of which factors (i.e., features, models) relate to higher performance, to clinical questions beyond Alzheimer's disease, and to using testing data beyond the Alzheimer's Disease Neuroimaging Initiative. Given the potential and lessons learned in the past ten years, we are excited by the prospects of grand challenges in machine learning and neuroimaging for the next ten years and beyond.
Automatic Segmentation of the Optic Nerve Head Region in Optical Coherence Tomography: A Methodological Review
Sep 06, 2021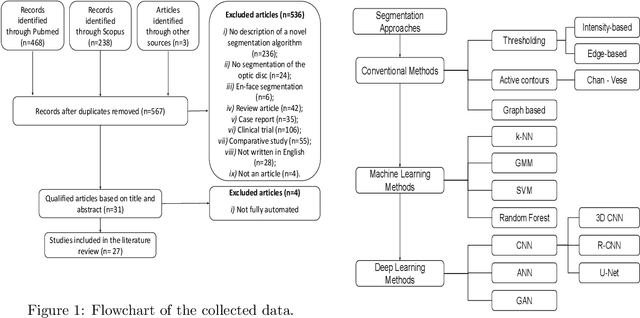

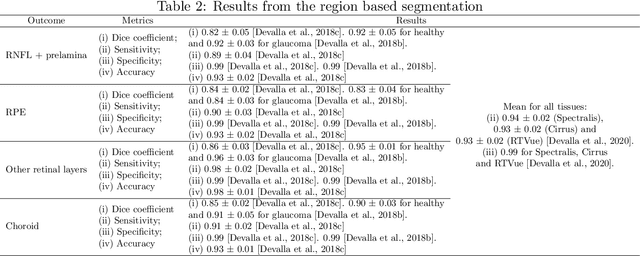
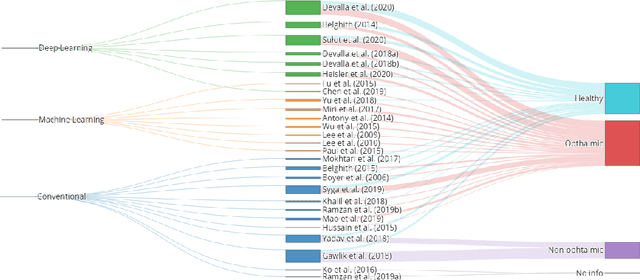
Abstract:The optic nerve head represents the intraocular section of the optic nerve (ONH), which is prone to damage by intraocular pressure. The advent of optical coherence tomography (OCT) has enabled the evaluation of novel optic nerve head parameters, namely the depth and curvature of the lamina cribrosa (LC). Together with the Bruch's membrane opening minimum-rim-width, these seem to be promising optic nerve head parameters for diagnosis and monitoring of retinal diseases such as glaucoma. Nonetheless, these optical coherence tomography derived biomarkers are mostly extracted through manual segmentation, which is time-consuming and prone to bias, thus limiting their usability in clinical practice. The automatic segmentation of optic nerve head in OCT scans could further improve the current clinical management of glaucoma and other diseases. This review summarizes the current state-of-the-art in automatic segmentation of the ONH in OCT. PubMed and Scopus were used to perform a systematic review. Additional works from other databases (IEEE, Google Scholar and ARVO IOVS) were also included, resulting in a total of 27 reviewed studies. For each algorithm, the methods, the size and type of dataset used for validation, and the respective results were carefully analyzed. The results show that deep learning-based algorithms provide the highest accuracy, sensitivity and specificity for segmenting the different structures of the ONH including the LC. However, a lack of consensus regarding the definition of segmented regions, extracted parameters and validation approaches has been observed, highlighting the importance and need of standardized methodologies for ONH segmentation.
Signal-carrying speckle in Optical Coherence Tomography: a methodological review on biomedical applications
Aug 30, 2021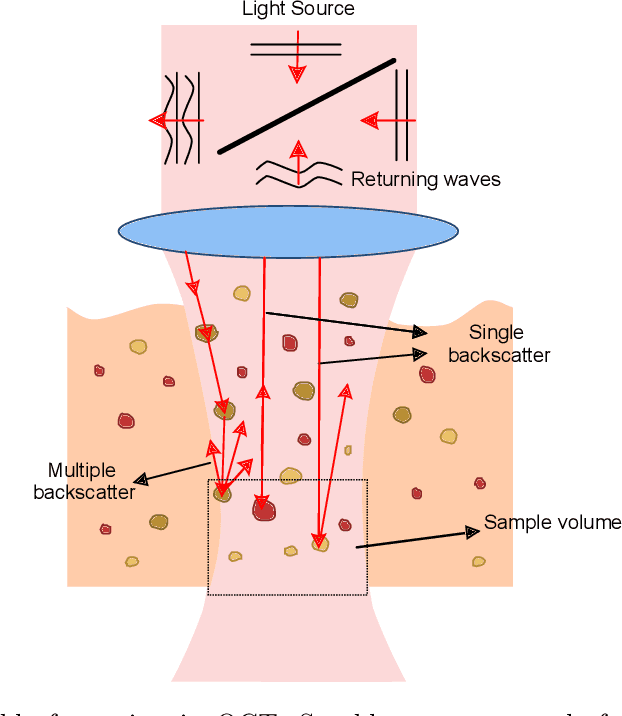
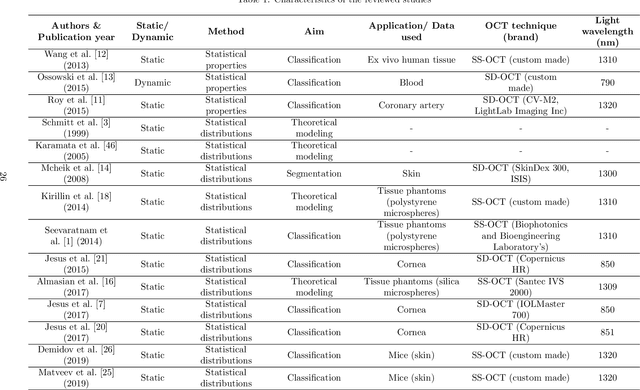
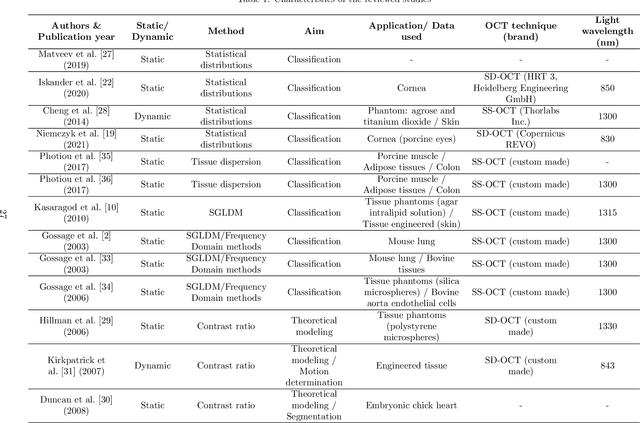
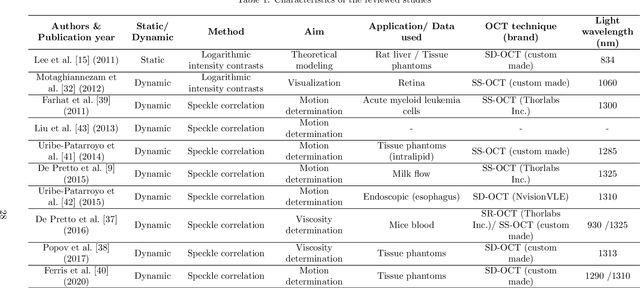
Abstract:Significance: Speckle has historically been considered a source of noise in coherent light imaging. However, a number of works in optical coherence tomography (OCT) imaging have shown that speckle patterns may contain relevant information regarding sub-resolution and structural properties of the tissues from which it is originated. Aim: The objective of this work is to provide a comprehensive overview of the methods developed for retrieving speckle information in biomedical OCT applications. Approach: PubMed and Scopus databases were used to perform a systematic review on studies published until April 2021. From 134-screened studies, 37 were eligible for this review. Results: The studies have been clustered according to the nature of their analysis, namely static or dynamic, and all features were described and analysed. The results show that features retrieved from speckle can be used successfully in different applications, such as classification and segmentation. However, the results also show that speckle analysis is highly application-dependant, and the best approach varies between applications. Conclusions: Several of the reviewed analysis were only performed in a theoretical context or using phantoms, showing that signal-carrying speckle analysis in OCT imaging is still in its early stage, and further work is needed to validate its applicability and reproducibility in a clinical context.
Reproducible radiomics through automated machine learning validated on twelve clinical applications
Aug 19, 2021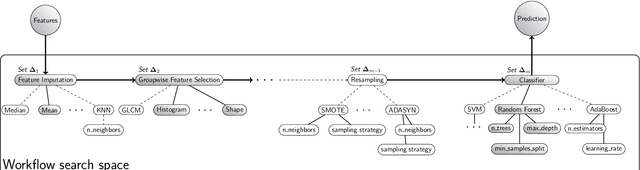
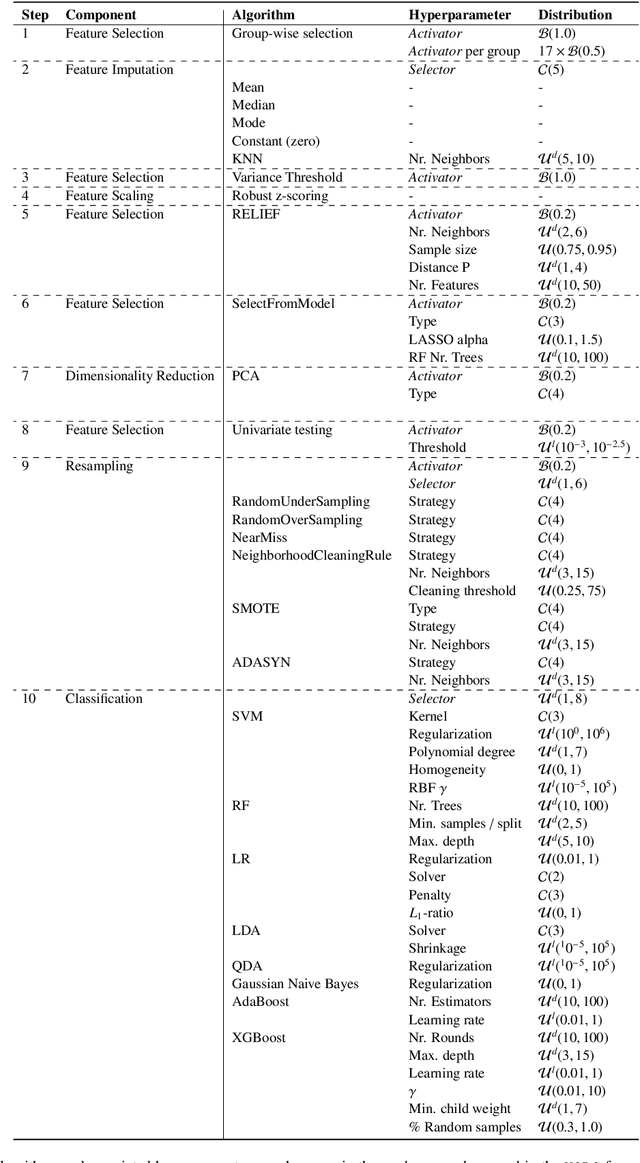
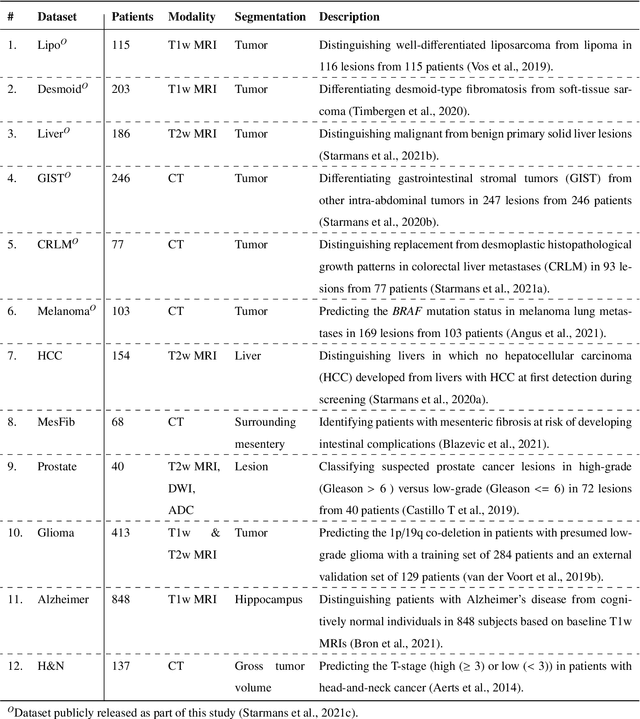
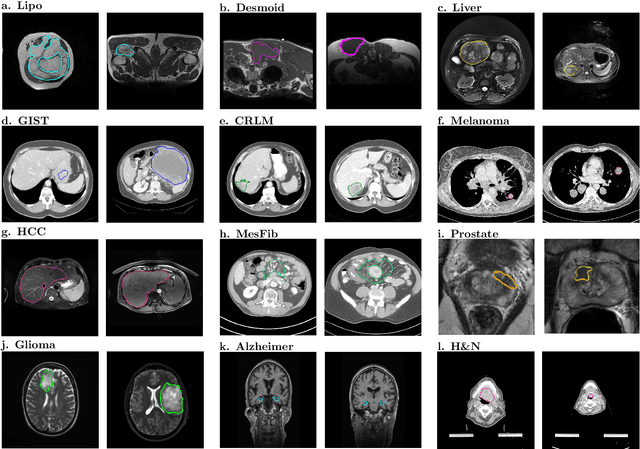
Abstract:Radiomics uses quantitative medical imaging features to predict clinical outcomes. While many radiomics methods have been described in the literature, these are generally designed for a single application. The aim of this study is to generalize radiomics across applications by proposing a framework to automatically construct and optimize the radiomics workflow per application. To this end, we formulate radiomics as a modular workflow, consisting of several components: image and segmentation preprocessing, feature extraction, feature and sample preprocessing, and machine learning. For each component, a collection of common algorithms is included. To optimize the workflow per application, we employ automated machine learning using a random search and ensembling. We evaluate our method in twelve different clinical applications, resulting in the following area under the curves: 1) liposarcoma (0.83); 2) desmoid-type fibromatosis (0.82); 3) primary liver tumors (0.81); 4) gastrointestinal stromal tumors (0.77); 5) colorectal liver metastases (0.68); 6) melanoma metastases (0.51); 7) hepatocellular carcinoma (0.75); 8) mesenteric fibrosis (0.81); 9) prostate cancer (0.72); 10) glioma (0.70); 11) Alzheimer's disease (0.87); and 12) head and neck cancer (0.84). Concluding, our method fully automatically constructs and optimizes the radiomics workflow, thereby streamlining the search for radiomics biomarkers in new applications. To facilitate reproducibility and future research, we publicly release six datasets, the software implementation of our framework (open-source), and the code to reproduce this study.
A Review of Generative Adversarial Networks in Cancer Imaging: New Applications, New Solutions
Jul 20, 2021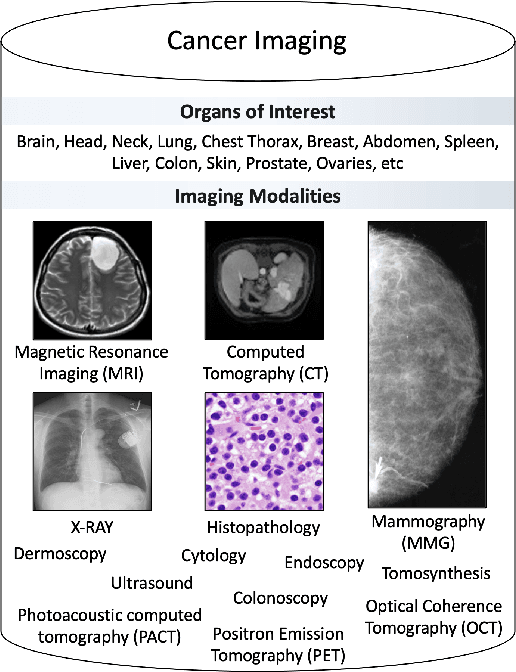
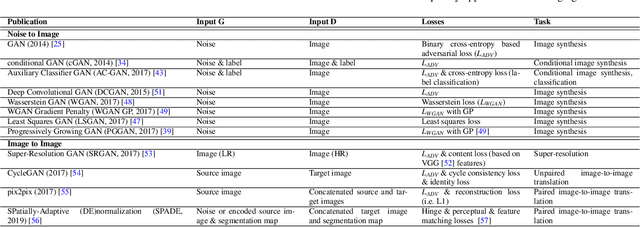
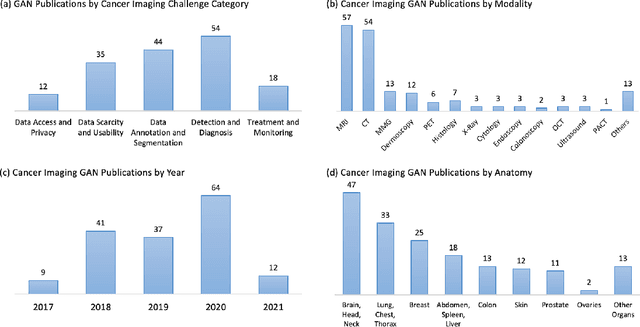
Abstract:Despite technological and medical advances, the detection, interpretation, and treatment of cancer based on imaging data continue to pose significant challenges. These include high inter-observer variability, difficulty of small-sized lesion detection, nodule interpretation and malignancy determination, inter- and intra-tumour heterogeneity, class imbalance, segmentation inaccuracies, and treatment effect uncertainty. The recent advancements in Generative Adversarial Networks (GANs) in computer vision as well as in medical imaging may provide a basis for enhanced capabilities in cancer detection and analysis. In this review, we assess the potential of GANs to address a number of key challenges of cancer imaging, including data scarcity and imbalance, domain and dataset shifts, data access and privacy, data annotation and quantification, as well as cancer detection, tumour profiling and treatment planning. We provide a critical appraisal of the existing literature of GANs applied to cancer imagery, together with suggestions on future research directions to address these challenges. We analyse and discuss 163 papers that apply adversarial training techniques in the context of cancer imaging and elaborate their methodologies, advantages and limitations. With this work, we strive to bridge the gap between the needs of the clinical cancer imaging community and the current and prospective research on GANs in the artificial intelligence community.
Evaluating glioma growth predictions as a forward ranking problem
Mar 22, 2021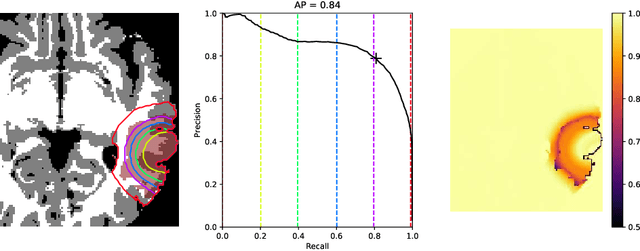
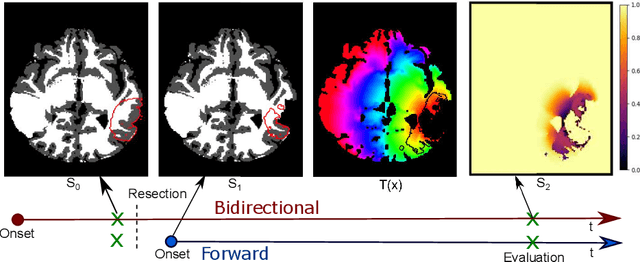
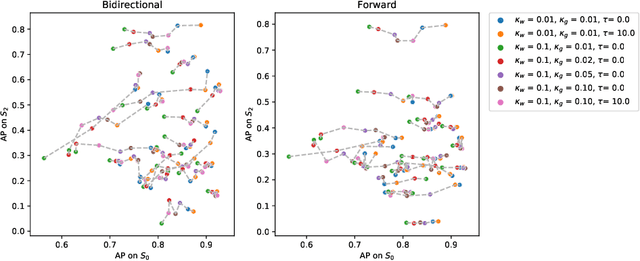
Abstract:The problem of tumor growth prediction is challenging, but promising results have been achieved with both model-driven and statistical methods. In this work, we present a framework for the evaluation of growth predictions that focuses on the spatial infiltration patterns, and specifically evaluating a prediction of future growth. We propose to frame the problem as a ranking problem rather than a segmentation problem. Using the average precision as a metric, we can evaluate the results with segmentations while using the full spatiotemporal prediction. Furthermore, by separating the model goodness-of-fit from future predictive performance, we show that in some cases, a better fit of model parameters does not guarantee a better the predictive power.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge