Samir Mitha
MLP-SRGAN: A Single-Dimension Super Resolution GAN using MLP-Mixer
Mar 11, 2023



Abstract:We propose a novel architecture called MLP-SRGAN, which is a single-dimension Super Resolution Generative Adversarial Network (SRGAN) that utilizes Multi-Layer Perceptron Mixers (MLP-Mixers) along with convolutional layers to upsample in the slice direction. MLP-SRGAN is trained and validated using high resolution (HR) FLAIR MRI from the MSSEG2 challenge dataset. The method was applied to three multicentre FLAIR datasets (CAIN, ADNI, CCNA) of images with low spatial resolution in the slice dimension to examine performance on held-out (unseen) clinical data. Upsampled results are compared to several state-of-the-art SR networks. For images with high resolution (HR) ground truths, peak-signal-to-noise-ratio (PSNR) and structural similarity index (SSIM) are used to measure upsampling performance. Several new structural, no-reference image quality metrics were proposed to quantify sharpness (edge strength), noise (entropy), and blurriness (low frequency information) in the absence of ground truths. Results show MLP-SRGAN results in sharper edges, less blurring, preserves more texture and fine-anatomical detail, with fewer parameters, faster training/evaluation time, and smaller model size than existing methods. Code for MLP-SRGAN training and inference, data generators, models and no-reference image quality metrics will be available at https://github.com/IAMLAB-Ryerson/MLP-SRGAN.
Biomedical image analysis competitions: The state of current participation practice
Dec 16, 2022Abstract:The number of international benchmarking competitions is steadily increasing in various fields of machine learning (ML) research and practice. So far, however, little is known about the common practice as well as bottlenecks faced by the community in tackling the research questions posed. To shed light on the status quo of algorithm development in the specific field of biomedical imaging analysis, we designed an international survey that was issued to all participants of challenges conducted in conjunction with the IEEE ISBI 2021 and MICCAI 2021 conferences (80 competitions in total). The survey covered participants' expertise and working environments, their chosen strategies, as well as algorithm characteristics. A median of 72% challenge participants took part in the survey. According to our results, knowledge exchange was the primary incentive (70%) for participation, while the reception of prize money played only a minor role (16%). While a median of 80 working hours was spent on method development, a large portion of participants stated that they did not have enough time for method development (32%). 25% perceived the infrastructure to be a bottleneck. Overall, 94% of all solutions were deep learning-based. Of these, 84% were based on standard architectures. 43% of the respondents reported that the data samples (e.g., images) were too large to be processed at once. This was most commonly addressed by patch-based training (69%), downsampling (37%), and solving 3D analysis tasks as a series of 2D tasks. K-fold cross-validation on the training set was performed by only 37% of the participants and only 50% of the participants performed ensembling based on multiple identical models (61%) or heterogeneous models (39%). 48% of the respondents applied postprocessing steps.
Mitosis Detection for Breast Cancer Pathology Images using UV-Net
Sep 21, 2021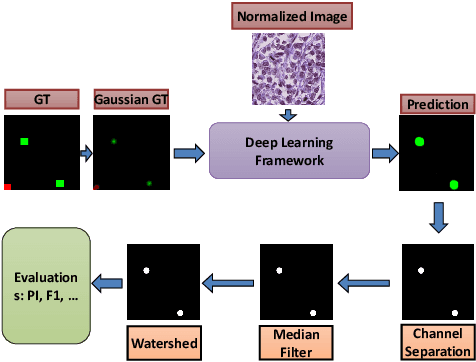
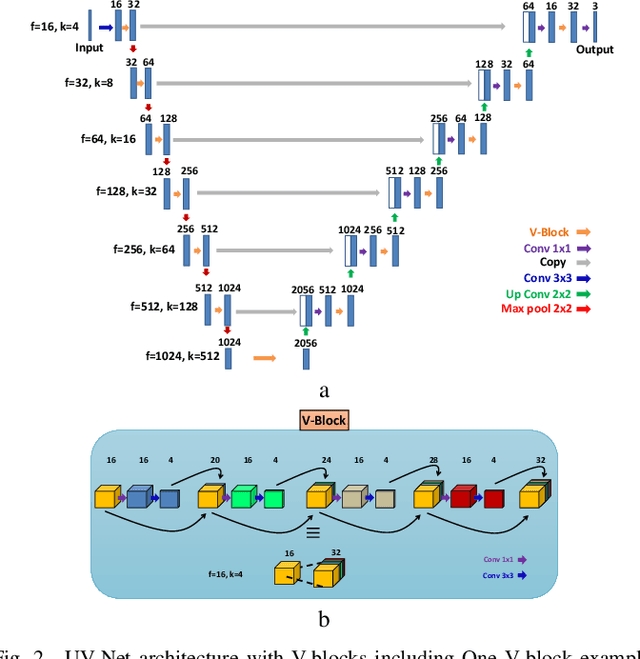
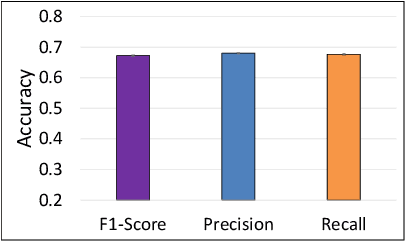
Abstract:The difficulty of detecting mitosis and its similarity to non-mitosis objects has remained a challenge in computational pathology. The lack of publicly available data has added more complexity. Deep learning algorithms have shown potentials in mitosis detection tasks. However, they face challenges when applied to pathology images with dense medium and diverse dataset. This paper introduces an optimized UV-Net architecture, developed to focus on mitosis details with high-resolution through feature preservation. Stain normalization methods are used to generalize the trained network. An F1 score of 0.6721 is achieved using this network.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge