Benjamin A. Goldstein
Multimodal Training to Unimodal Deployment: Leveraging Unstructured Data During Training to Optimize Structured Data Only Deployment
Mar 23, 2026Abstract:Unstructured Electronic Health Record (EHR) data, such as clinical notes, contain clinical contextual observations that are not directly reflected in structured data fields. This additional information can substantially improve model learning. However, due to their unstructured nature, these data are often unavailable or impractical to use when deploying a model. We introduce a multimodal learning framework that leverages unstructured EHR data during training while producing a model that can be deployed using only structured EHR data. Using a cohort of 3,466 children evaluated for late talking, we generated note embeddings with BioClinicalBERT and encoded structured embeddings from demographics and medical codes. A note-based teacher model and a structured-only student model were jointly trained using contrastive learning and contrastive knowledge distillation loss, producing a strong classifier (AUROC = 0.985). Our proposed model reached an AUROC of 0.705, outperforming the structured-only baseline of 0.656. These results demonstrate that incorporating unstructured data during training enhances the model's capacity to identify task-relevant information within structured EHR data, enabling a deployable structured-only phenotype model.
TRACER: Transfer Learning based Real-time Adaptation for Clinical Evolving Risk
Dec 14, 2025Abstract:Clinical decision support tools built on electronic health records often experience performance drift due to temporal population shifts, particularly when changes in the clinical environment initially affect only a subset of patients, resulting in a transition to mixed populations. Such case-mix changes commonly arise following system-level operational updates or the emergence of new diseases, such as COVID-19. We propose TRACER (Transfer Learning-based Real-time Adaptation for Clinical Evolving Risk), a framework that identifies encounter-level transition membership and adapts predictive models using transfer learning without full retraining. In simulation studies, TRACER outperformed static models trained on historical or contemporary data. In a real-world application predicting hospital admission following emergency department visits across the COVID-19 transition, TRACER improved both discrimination and calibration. TRACER provides a scalable approach for maintaining robust predictive performance under evolving and heterogeneous clinical conditions.
FairPOT: Balancing AUC Performance and Fairness with Proportional Optimal Transport
Aug 05, 2025Abstract:Fairness metrics utilizing the area under the receiver operator characteristic curve (AUC) have gained increasing attention in high-stakes domains such as healthcare, finance, and criminal justice. In these domains, fairness is often evaluated over risk scores rather than binary outcomes, and a common challenge is that enforcing strict fairness can significantly degrade AUC performance. To address this challenge, we propose Fair Proportional Optimal Transport (FairPOT), a novel, model-agnostic post-processing framework that strategically aligns risk score distributions across different groups using optimal transport, but does so selectively by transforming a controllable proportion, i.e., the top-lambda quantile, of scores within the disadvantaged group. By varying lambda, our method allows for a tunable trade-off between reducing AUC disparities and maintaining overall AUC performance. Furthermore, we extend FairPOT to the partial AUC setting, enabling fairness interventions to concentrate on the highest-risk regions. Extensive experiments on synthetic, public, and clinical datasets show that FairPOT consistently outperforms existing post-processing techniques in both global and partial AUC scenarios, often achieving improved fairness with slight AUC degradation or even positive gains in utility. The computational efficiency and practical adaptability of FairPOT make it a promising solution for real-world deployment.
An Outcome Model Approach to Translating a Randomized Controlled Trial Results to a Target Population
Jun 25, 2018
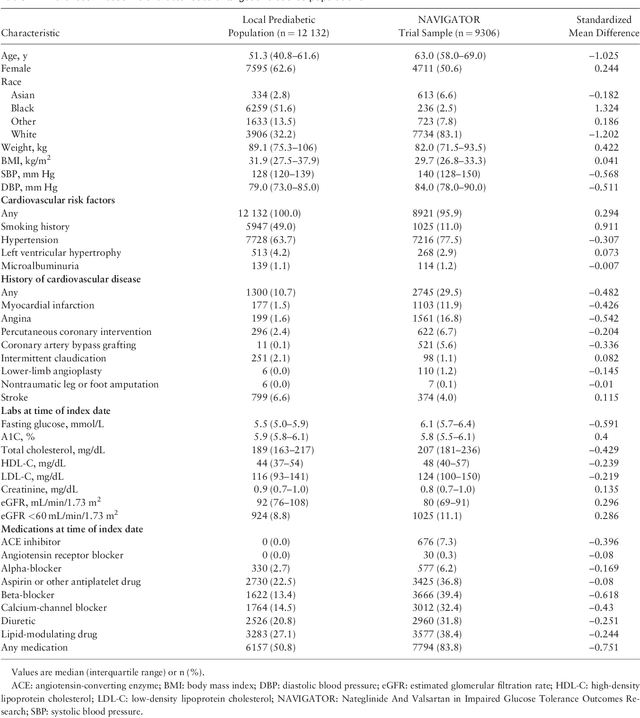


Abstract:Participants enrolled into randomized controlled trials (RCTs) often do not reflect real-world populations. Previous research in how best to translate RCT results to target populations has focused on weighting RCT data to look like the target data. Simulation work, however, has suggested that an outcome model approach may be preferable. Here we describe such an approach using source data from the 2x2 factorial NAVIGATOR trial which evaluated the impact of valsartan and nateglinide on cardiovascular outcomes and new-onset diabetes in a pre-diabetic population. Our target data consisted of people with pre-diabetes serviced at our institution. We used Random Survival Forests to develop separate outcome models for each of the 4 treatments, estimating the 5-year risk difference for progression to diabetes and estimated the treatment effect in our local patient populations, as well as sub-populations, and the results compared to the traditional weighting approach. Our models suggested that the treatment effect for valsartan in our patient population was the same as in the trial, whereas for nateglinide treatment effect was stronger than observed in the original trial. Our effect estimates were more efficient than the weighting approach.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge