Ryosuke Araki
Reference-Free Image Quality Assessment for Virtual Try-On via Human Feedback
Mar 13, 2026Abstract:Given a person image and a garment image, image-based Virtual Try-ON (VTON) synthesizes a try-on image of the person wearing the target garment. As VTON systems become increasingly important in practical applications such as fashion e-commerce, reliable evaluation of their outputs has emerged as a critical challenge. In real-world scenarios, ground-truth images of the same person wearing the target garment are typically unavailable, making reference-based evaluation impractical. Moreover, widely used distribution-level metrics such as Fréchet Inception Distance and Kernel Inception Distance measure dataset-level similarity and fail to reflect the perceptual quality of individual generated images. To address these limitations, we propose Image Quality Assessment for Virtual Try-On (VTON-IQA), a reference-free framework for human-aligned, image-level quality assessment without requiring ground-truth images. To model human perceptual judgments, we construct VTON-QBench, a large-scale human-annotated benchmark comprising 62,688 try-on images generated by 14 representative VTON models and 431,800 quality annotations collected from 13,838 qualified annotators. To the best of our knowledge, this is the largest dataset to date for human subjective evaluation in virtual try-on. Evaluating virtual try-on quality requires verifying both garment fidelity and the preservation of person-specific details. To explicitly model such interactions, we introduce an Interleaved Cross-Attention module that extends standard transformer blocks by inserting a cross-attention layer between self-attention and MLP in the latter blocks. Extensive experiments show that VTON-IQA achieves reliable human-aligned image-level quality prediction. Moreover, we conduct a comprehensive benchmark evaluation of 14 representative VTON models using VTON-IQA.
Combining Noise-to-Image and Image-to-Image GANs: Brain MR Image Augmentation for Tumor Detection
May 31, 2019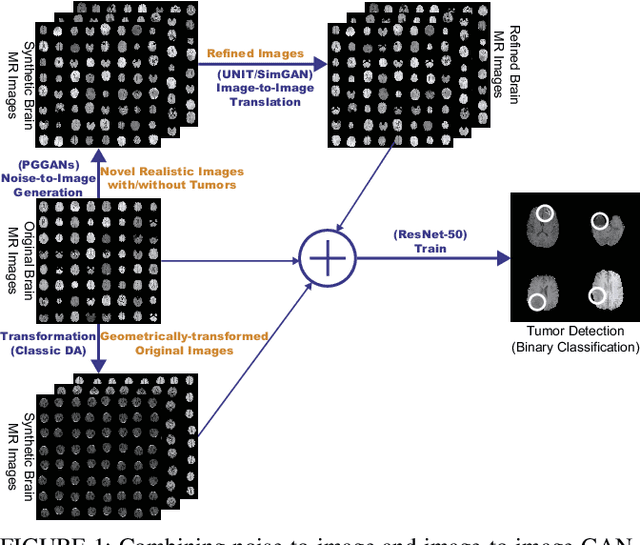
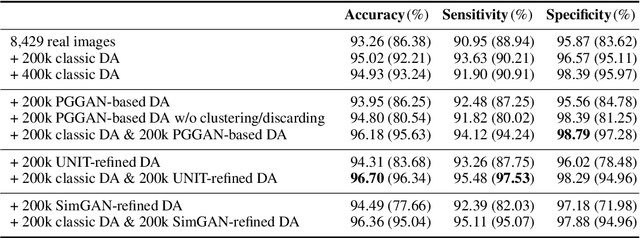
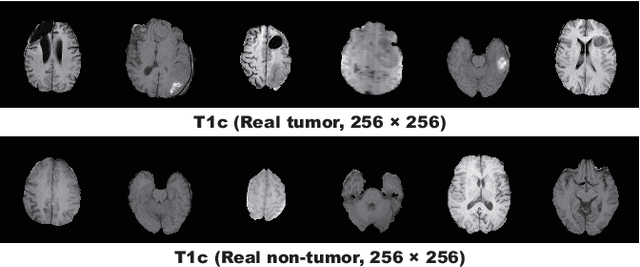
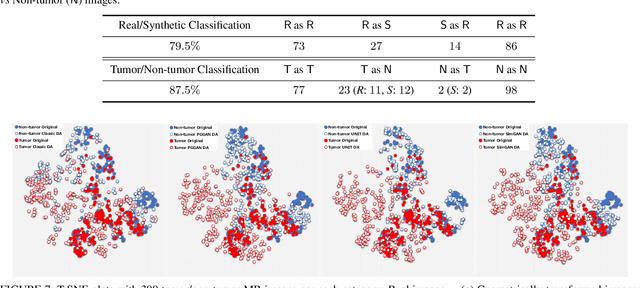
Abstract:Convolutional Neural Networks (CNNs) can achieve excellent computer-assisted diagnosis performance, relying on sufficient annotated training data. Unfortunately, most medical imaging datasets, often collected from various scanners, are small and fragmented. In this context, as a Data Augmentation (DA) technique, Generative Adversarial Networks (GANs) can synthesize realistic/diverse additional training images to fill the data lack in the real image distribution; researchers have improved classification by augmenting images with noise-to-image (e.g., random noise samples to diverse pathological images) or image-to-image GANs (e.g., a benign image to a malignant one). Yet, no research has reported results combining (i) noise-to-image GANs and image-to-image GANs or (ii) GANs and other deep generative models, for further performance boost. Therefore, to maximize the DA effect with the GAN combinations, we propose a two-step GAN-based DA that generates and refines brain MR images with/without tumors separately: (i) Progressive Growing of GANs (PGGANs), multi-stage noise-to-image GAN for high-resolution image generation, first generates realistic/diverse 256 x 256 images--even a physician cannot accurately distinguish them from real ones via Visual Turing Test; (ii) UNsupervised Image-to-image Translation or SimGAN, image-to-image GAN combining GANs/Variational AutoEncoders or using a GAN loss for DA, further refines the texture/shape of the PGGAN-generated images similarly to the real ones. We thoroughly investigate CNN-based tumor classification results, also considering the influence of pre-training on ImageNet and discarding weird-looking GAN-generated images. The results show that, when combined with classic DA, our two-step GAN-based DA can significantly outperform the classic DA alone, in tumor detection (i.e., boosting sensitivity from 93.63% to 97.53%) and also in other tasks.
Infinite Brain MR Images: PGGAN-based Data Augmentation for Tumor Detection
Mar 29, 2019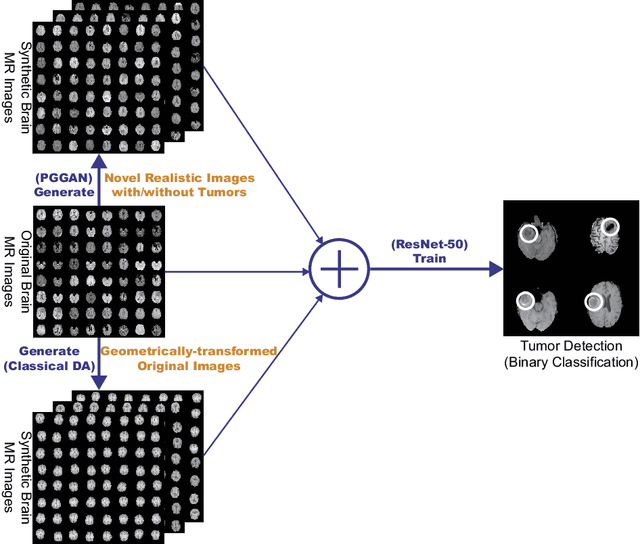

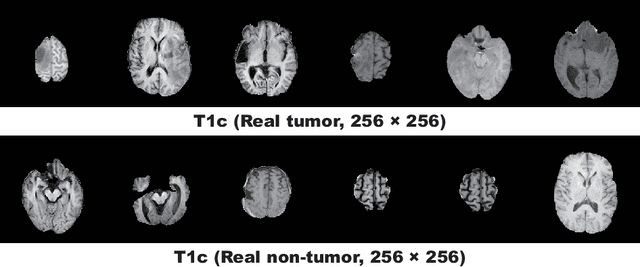
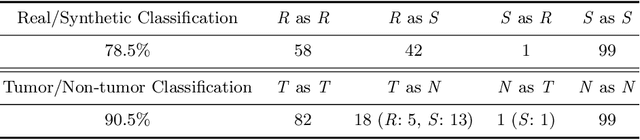
Abstract:Due to the lack of available annotated medical images, accurate computer-assisted diagnosis requires intensive Data Augmentation (DA) techniques, such as geometric/intensity transformations of original images; however, those transformed images intrinsically have a similar distribution to the original ones, leading to limited performance improvement. To fill the data lack in the real image distribution, we synthesize brain contrast-enhanced Magnetic Resonance (MR) images---realistic but completely different from the original ones---using Generative Adversarial Networks (GANs). This study exploits Progressive Growing of GANs (PGGANs), a multi-stage generative training method, to generate original-sized 256 X 256 MR images for Convolutional Neural Network-based brain tumor detection, which is challenging via conventional GANs; difficulties arise due to unstable GAN training with high resolution and a variety of tumors in size, location, shape, and contrast. Our preliminary results show that this novel PGGAN-based DA method can achieve promising performance improvement, when combined with classical DA, in tumor detection and also in other medical imaging tasks.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge