Kazunori Yamaguchi
Continuous quantification of viral plaque dynamics using ultra-large-area label-free imaging enables rapid antiviral susceptibility testing
May 03, 2026Abstract:The plaque reduction assay (PRA) remains the gold standard for antiviral susceptibility testing, evaluating drug potency by measuring reductions in plaque-forming units (PFUs). However, the traditional PRA is time-consuming, labor-intensive, prone to manual counting errors, and offers limited scalability. Moreover, its reliance on destructive fixation and chemical staining reduces the assay to a static, endpoint observation, obscuring the dynamic, time-resolved kinetics of dose-dependent viral inhibition. Here, we introduce a label-free, time-resolved PRA platform that transforms the conventional assay into a continuous, high-dimensional measurement of viral infection dynamics. Our system integrates a compact lens-free imaging setup with a custom-designed ultra-large-area (100 cm^2) thin-film transistor (TFT) image sensor and deep learning-based algorithms to autonomously quantify PFU dynamics within an incubator. Validated using herpes simplex virus type-1 (HSV-1) treated with acyclovir, the platform matched chemically-stained ground truth measurements with zero false positives while accelerating readout by ~26 hours. Crucially, our system revealed that increasing drug concentrations induce temporally distinct delays and suppress new PFU formation, enabling conclusive drug efficacy evaluations within ~60 hours post-infection. This scalable, label-free framework redefines antiviral susceptibility testing as a rapid, time-resolved and information-rich measurement framework, providing a generalizable platform for virology research, high-throughput drug screening, and clinical diagnostics.
Deep Learning-enabled Detection and Classification of Bacterial Colonies using a Thin Film Transistor (TFT) Image Sensor
May 07, 2022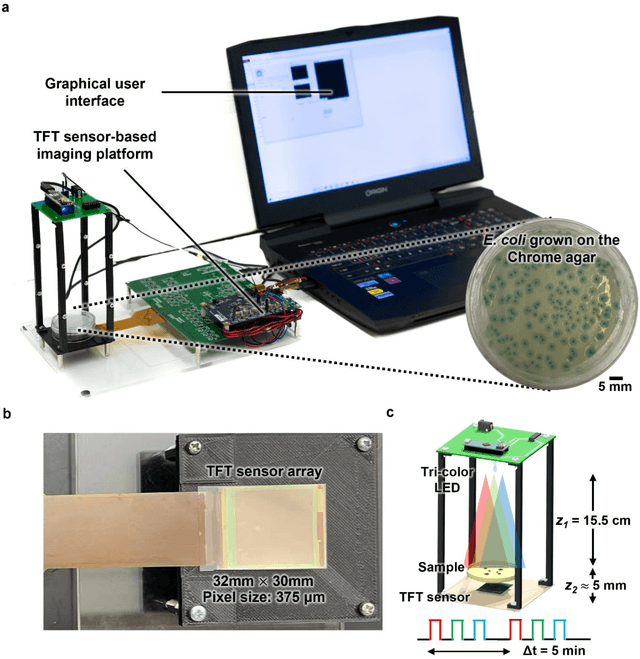
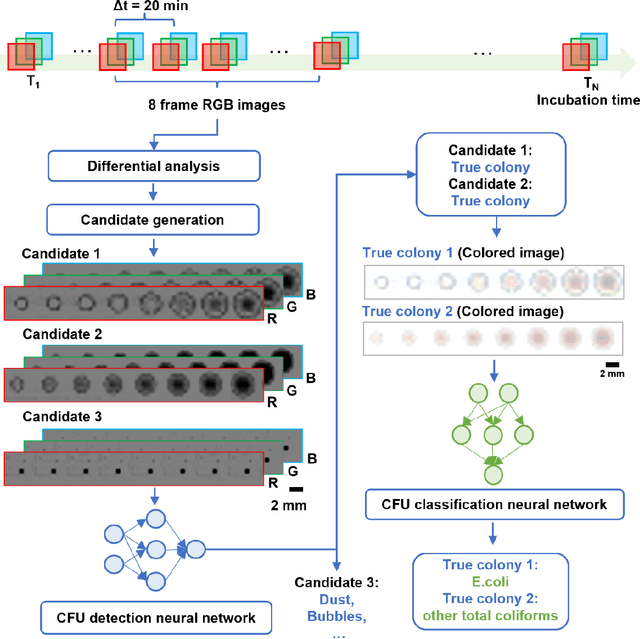

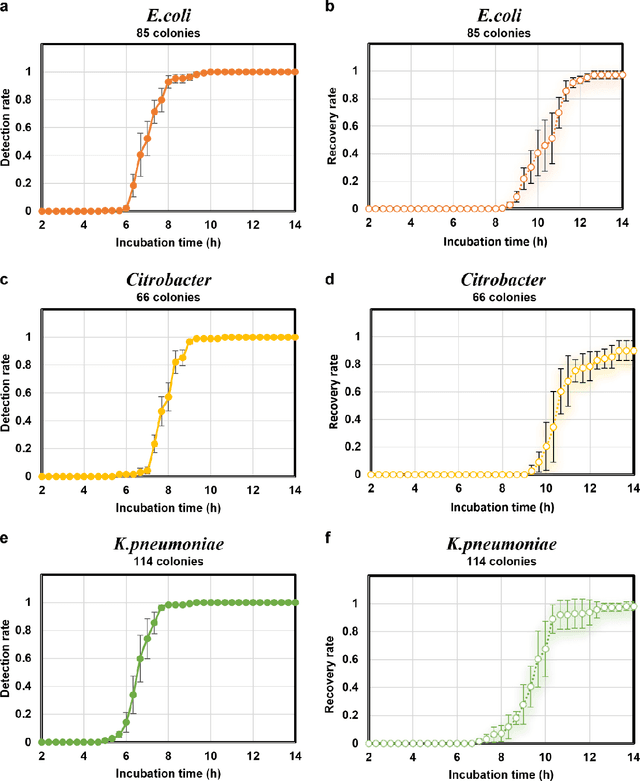
Abstract:Early detection and identification of pathogenic bacteria such as Escherichia coli (E. coli) is an essential task for public health. The conventional culture-based methods for bacterial colony detection usually take >24 hours to get the final read-out. Here, we demonstrate a bacterial colony-forming-unit (CFU) detection system exploiting a thin-film-transistor (TFT)-based image sensor array that saves ~12 hours compared to the Environmental Protection Agency (EPA)-approved methods. To demonstrate the efficacy of this CFU detection system, a lensfree imaging modality was built using the TFT image sensor with a sample field-of-view of ~10 cm^2. Time-lapse images of bacterial colonies cultured on chromogenic agar plates were automatically collected at 5-minute intervals. Two deep neural networks were used to detect and count the growing colonies and identify their species. When blindly tested with 265 colonies of E. coli and other coliform bacteria (i.e., Citrobacter and Klebsiella pneumoniae), our system reached an average CFU detection rate of 97.3% at 9 hours of incubation and an average recovery rate of 91.6% at ~12 hours. This TFT-based sensor can be applied to various microbiological detection methods. Due to the large scalability, ultra-large field-of-view, and low cost of the TFT-based image sensors, this platform can be integrated with each agar plate to be tested and disposed of after the automated CFU count. The imaging field-of-view of this platform can be cost-effectively increased to >100 cm^2 to provide a massive throughput for CFU detection using, e.g., roll-to-roll manufacturing of TFTs as used in the flexible display industry.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge