Emery N. Brown
Enhanced Portable Ultra Low-Field Diffusion Tensor Imaging with Bayesian Artifact Correction and Deep Learning-Based Super-Resolution
Feb 11, 2026Abstract:Portable, ultra-low-field (ULF) magnetic resonance imaging has the potential to expand access to neuroimaging but currently suffers from coarse spatial and angular resolutions and low signal-to-noise ratios. Diffusion tensor imaging (DTI), a sequence tailored to detect and reconstruct white matter tracts within the brain, is particularly prone to such imaging degradation due to inherent sequence design coupled with prolonged scan times. In addition, ULF DTI scans exhibit artifacting that spans both the space and angular domains, requiring a custom modelling algorithm for subsequent correction. We introduce a nine-direction, single-shell ULF DTI sequence, as well as a companion Bayesian bias field correction algorithm that possesses angular dependence and convolutional neural network-based superresolution algorithm that is generalizable across DTI datasets and does not require re-training (''DiffSR''). We show through a synthetic downsampling experiment and white matter assessment in real, matched ULF and high-field DTI scans that these algorithms can recover microstructural and volumetric white matter information at ULF. We also show that DiffSR can be directly applied to white matter-based Alzheimers disease classification in synthetically degraded scans, with notable improvements in agreement between DTI metrics, as compared to un-degraded scans. We freely disseminate the Bayesian bias correction algorithm and DiffSR with the goal of furthering progress on both ULF reconstruction methods and general DTI sequence harmonization. We release all code related to DiffSR for $\href{https://github.com/markolchanyi/DiffSR}{public \space use}$.
Controlling Level of Unconsciousness by Titrating Propofol with Deep Reinforcement Learning
Sep 09, 2020


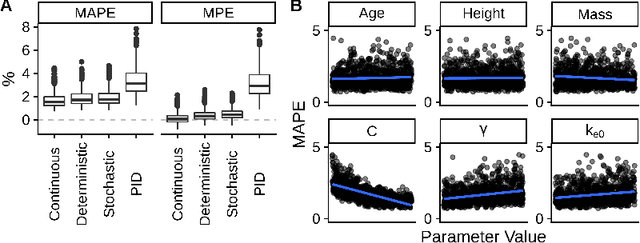
Abstract:Reinforcement Learning (RL) can be used to fit a mapping from patient state to a medication regimen. Prior studies have used deterministic and value-based tabular learning to learn a propofol dose from an observed anesthetic state. Deep RL replaces the table with a deep neural network and has been used to learn medication regimens from registry databases. Here we perform the first application of deep RL to closed-loop control of anesthetic dosing in a simulated environment. We use the cross-entropy method to train a deep neural network to map an observed anesthetic state to a probability of infusing a fixed propofol dosage. During testing, we implement a deterministic policy that transforms the probability of infusion to a continuous infusion rate. The model is trained and tested on simulated pharmacokinetic/pharmacodynamic models with randomized parameters to ensure robustness to patient variability. The deep RL agent significantly outperformed a proportional-integral-derivative controller (median absolute performance error 1.7% +/- 0.6 and 3.4% +/- 1.2). Modeling continuous input variables instead of a table affords more robust pattern recognition and utilizes our prior domain knowledge. Deep RL learned a smooth policy with a natural interpretation to data scientists and anesthesia care providers alike.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge