Constantin Pape
Synthetic patches, real images: screening for centrosome aberrations in EM images of human cancer cells
Aug 27, 2019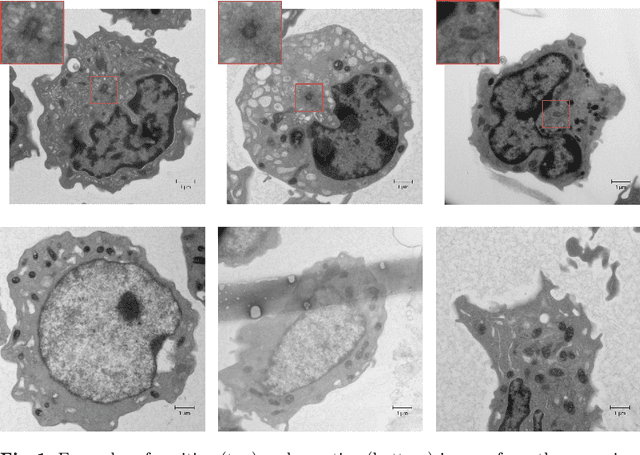
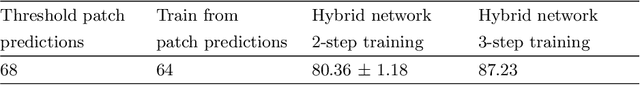
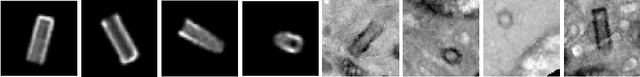
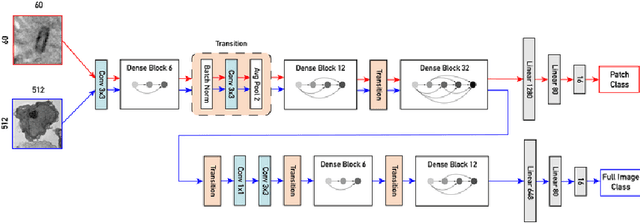
Abstract:Recent advances in high-throughput electron microscopy imaging enable detailed study of centrosome aberrations in cancer cells. While the image acquisition in such pipelines is automated, manual detection of centrioles is still necessary to select cells for re-imaging at higher magnification. In this contribution we propose an algorithm which performs this step automatically and with high accuracy. From the image labels produced by human experts and a 3D model of a centriole we construct an additional training set with patch-level labels. A two-level DenseNet is trained on the hybrid training data with synthetic patches and real images, achieving much better results on real patient data than training only at the image-level. The code can be found at https://github.com/kreshuklab/centriole_detection.
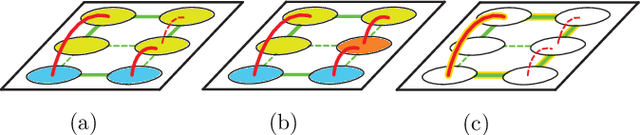
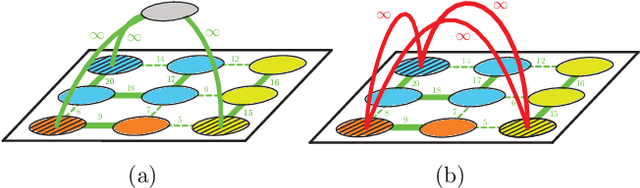
A Generalized Framework for Agglomerative Clustering of Signed Graphs applied to Instance Segmentation
Jun 27, 2019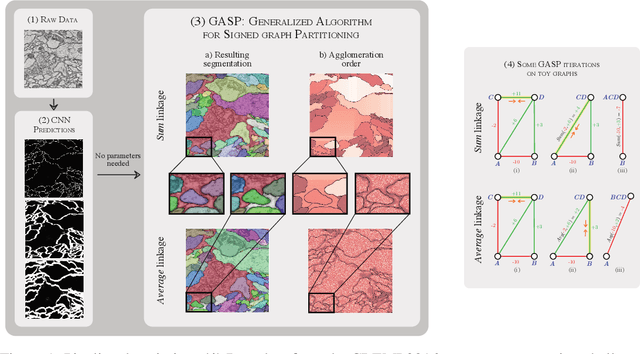
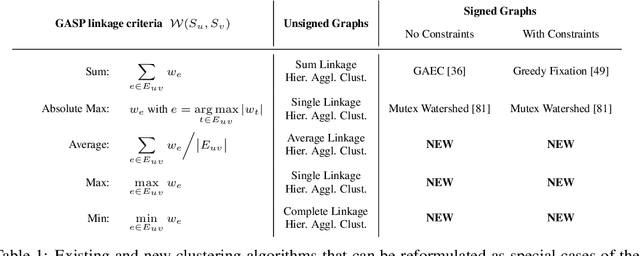
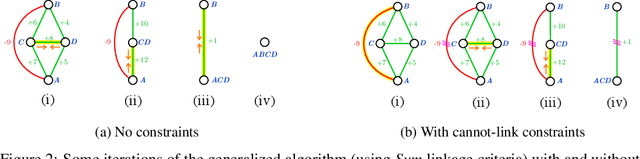
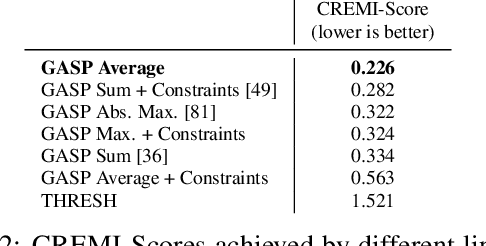
Abstract:We propose a novel theoretical framework that generalizes algorithms for hierarchical agglomerative clustering to weighted graphs with both attractive and repulsive interactions between the nodes. This framework defines GASP, a Generalized Algorithm for Signed graph Partitioning, and allows us to explore many combinations of different linkage criteria and cannot-link constraints. We prove the equivalence of existing clustering methods to some of those combinations, and introduce new algorithms for combinations which have not been studied. An extensive comparison is performed to evaluate properties of the clustering algorithms in the context of instance segmentation in images, including robustness to noise and efficiency. We show how one of the new algorithms proposed in our framework outperforms all previously known agglomerative methods for signed graphs, both on the competitive CREMI 2016 EM segmentation benchmark and on the CityScapes dataset.
Leveraging Domain Knowledge to improve EM image segmentation with Lifted Multicuts
May 25, 2019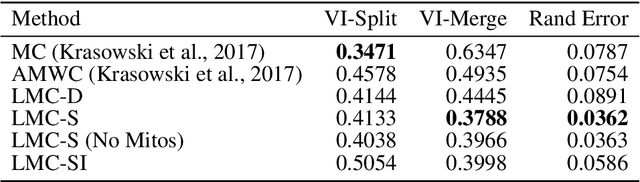
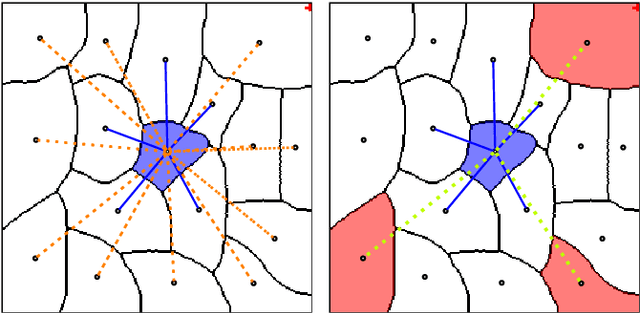
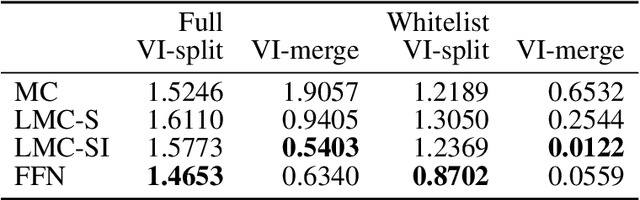
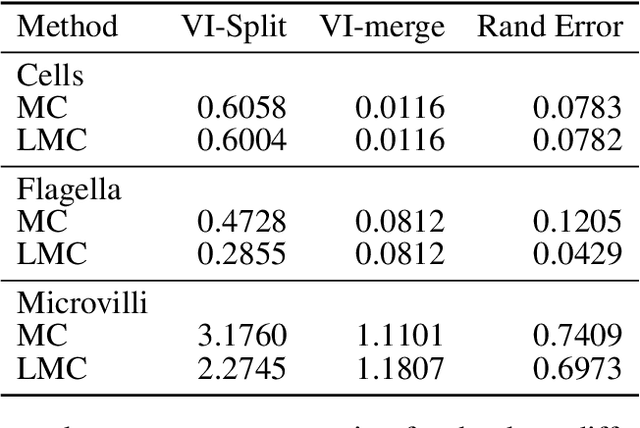
Abstract:The throughput of electron microscopes has increased significantly in recent years, enabling detailed analysis of cell morphology and ultrastructure. Analysis of neural circuits at single-synapse resolution remains the flagship target of this technique, but applications to cell and developmental biology are also starting to emerge at scale. The amount of data acquired in such studies makes manual instance segmentation, a fundamental step in many analysis pipelines, impossible. While automatic segmentation approaches have improved significantly thanks to the adoption of convolutional neural networks, their accuracy still lags behind human annotations and requires additional manual proof-reading. A major hindrance to further improvements is the limited field of view of the segmentation networks preventing them from exploiting the expected cell morphology or other prior biological knowledge which humans use to inform their segmentation decisions. In this contribution, we show how such domain-specific information can be leveraged by expressing it as long-range interactions in a graph partitioning problem known as the lifted multicut problem. Using this formulation, we demonstrate significant improvement in segmentation accuracy for three challenging EM segmentation problems from neuroscience and cell biology.
The Mutex Watershed and its Objective: Efficient, Parameter-Free Image Partitioning
Apr 25, 2019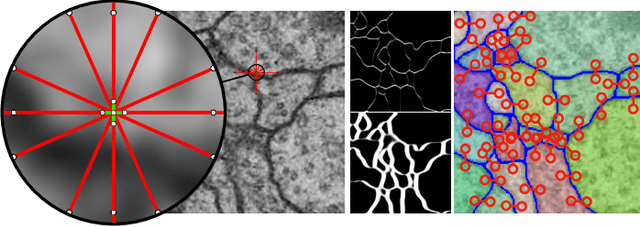
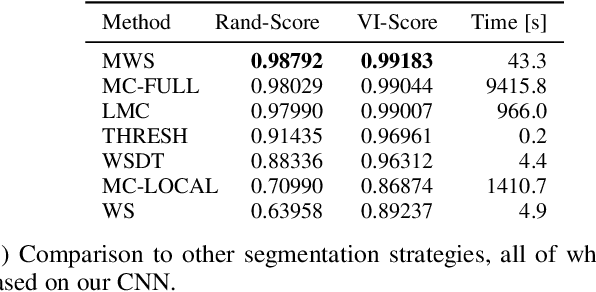
Abstract:Image partitioning, or segmentation without semantics, is the task of decomposing an image into distinct segments, or equivalently to detect closed contours. Most prior work either requires seeds, one per segment; or a threshold; or formulates the task as multicut / correlation clustering, an NP-hard problem. Here, we propose a greedy algorithm for signed graph partitioning, the "Mutex Watershed". Unlike seeded watershed, the algorithm can accommodate not only attractive but also repulsive cues, allowing it to find a previously unspecified number of segments without the need for explicit seeds or a tunable threshold. We also prove that this simple algorithm solves to global optimality an objective function that is intimately related to the multicut / correlation clustering integer linear programming formulation. The algorithm is deterministic, very simple to implement, and has empirically linearithmic complexity. When presented with short-range attractive and long-range repulsive cues from a deep neural network, the Mutex Watershed gives the best results currently known for the competitive ISBI 2012 EM segmentation benchmark.
Synaptic Cleft Segmentation in Non-Isotropic Volume Electron Microscopy of the Complete Drosophila Brain
May 07, 2018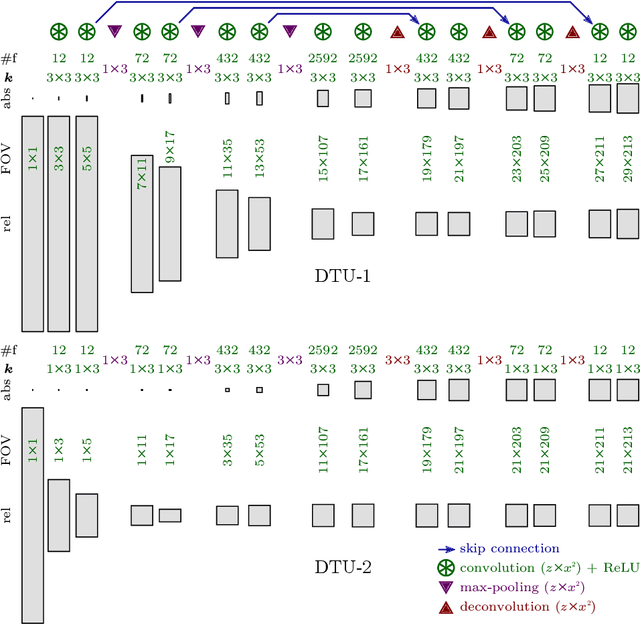
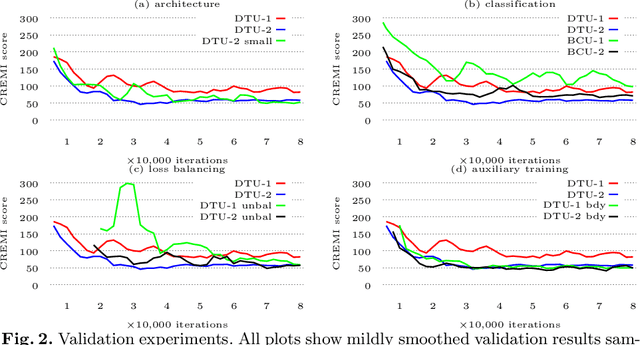
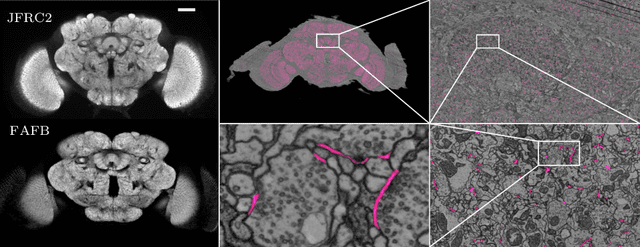
Abstract:Neural circuit reconstruction at single synapse resolution is increasingly recognized as crucially important to decipher the function of biological nervous systems. Volume electron microscopy in serial transmission or scanning mode has been demonstrated to provide the necessary resolution to segment or trace all neurites and to annotate all synaptic connections. Automatic annotation of synaptic connections has been done successfully in near isotropic electron microscopy of vertebrate model organisms. Results on non-isotropic data in insect models, however, are not yet on par with human annotation. We designed a new 3D-U-Net architecture to optimally represent isotropic fields of view in non-isotropic data. We used regression on a signed distance transform of manually annotated synaptic clefts of the CREMI challenge dataset to train this model and observed significant improvement over the state of the art. We developed open source software for optimized parallel prediction on very large volumetric datasets and applied our model to predict synaptic clefts in a 50 tera-voxels dataset of the complete Drosophila brain. Our model generalizes well to areas far away from where training data was available.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge