Anca-Ligia Grosu
Deep-Learning Atlas Registration for Melanoma Brain Metastases: Preserving Pathology While Enabling Cohort-Level Analyses
Feb 13, 2026Abstract:Melanoma brain metastases (MBM) are common and spatially heterogeneous lesions, complicating cohort-level analyses due to anatomical variability and differing MRI protocols. We propose a fully differentiable, deep-learning-based deformable registration framework that aligns individual pathological brains to a common atlas while preserving metastatic tissue without requiring lesion masks or preprocessing. Missing anatomical correspondences caused by metastases are handled through a forward-model similarity metric based on distance-transformed anatomical labels, combined with a volume-preserving regularization term to ensure deformation plausibility. Registration performance was evaluated using Dice coefficient (DSC), Hausdorff distance (HD), average symmetric surface distance (ASSD), and Jacobian-based measures. The method was applied to 209 MBM patients from three centres, enabling standardized mapping of metastases to anatomical, arterial, and perfusion atlases. The framework achieved high registration accuracy across datasets (DSC 0.89-0.92, HD 6.79-7.60 mm, ASSD 0.63-0.77 mm) while preserving metastatic volumes. Spatial analysis demonstrated significant over-representation of MBM in the cerebral cortex and putamen, under-representation in white matter, and consistent localization near the gray-white matter junction. No arterial territory showed increased metastasis frequency after volume correction. This approach enables robust atlas registration of pathological brain MRI without lesion masks and supports reproducible multi-centre analyses. Applied to MBM, it confirms and refines known spatial predilections, particularly preferential seeding near the gray-white matter junction and cortical regions. The publicly available implementation facilitates reproducible research and extension to other brain tumours and neurological pathologies.
Segmentation of Prostate Tumour Volumes from PET Images is a Different Ball Game
Jul 15, 2024



Abstract:Accurate segmentation of prostate tumours from PET images presents a formidable challenge in medical image analysis. Despite considerable work and improvement in delineating organs from CT and MR modalities, the existing standards do not transfer well and produce quality results in PET related tasks. Particularly, contemporary methods fail to accurately consider the intensity-based scaling applied by the physicians during manual annotation of tumour contours. In this paper, we observe that the prostate-localised uptake threshold ranges are beneficial for suppressing outliers. Therefore, we utilize the intensity threshold values, to implement a new custom-feature-clipping normalisation technique. We evaluate multiple, established U-Net variants under different normalisation schemes, using the nnU-Net framework. All models were trained and tested on multiple datasets, obtained with two radioactive tracers: [68-Ga]Ga-PSMA-11 and [18-F]PSMA-1007. Our results show that the U-Net models achieve much better performance when the PET scans are preprocessed with our novel clipping technique.
Analysis of clinical, dosimetric and radiomic features for predicting local failure after stereotactic radiotherapy of brain metastases in malignant melanoma
May 31, 2024



Abstract:Background: The aim of this study was to investigate the role of clinical, dosimetric and pretherapeutic magnetic resonance imaging (MRI) features for lesion-specific outcome prediction of stereotactic radiotherapy (SRT) in patients with brain metastases from malignant melanoma (MBM). Methods: In this multicenter, retrospective analysis, we reviewed 517 MBM from 130 patients treated with SRT (single fraction or hypofractionated). For each gross tumor volume (GTV) 1576 radiomic features (RF) were calculated (788 each for the GTV and for a 3 mm margin around the GTV). Clinical parameters, radiation dose and RF from pretherapeutic contrast-enhanced T1-weighted MRI from different institutions were evaluated with a feature processing and elimination pipeline in a nested cross-validation scheme. Results: Seventy-two (72) of 517 lesions (13.9%) showed a local failure (LF) after SRT. The processing pipeline showed clinical, dosimetric and radiomic features providing information for LF prediction. The most prominent ones were the correlation of the gray level co-occurrence matrix of the margin (hazard ratio (HR): 0.37, confidence interval (CI): 0.23-0.58) and systemic therapy before SRT (HR: 0.55, CI: 0.42-0.70). The majority of RF associated with LF was calculated in the margin around the GTV. Conclusions: Pretherapeutic MRI based RF connected with lesion-specific outcome after SRT could be identified, despite multicentric data and minor differences in imaging protocols. Image data analysis of the surrounding metastatic environment may provide therapy-relevant information with the potential to further individualize radiotherapy strategies.
IB-U-Nets: Improving medical image segmentation tasks with 3D Inductive Biased kernels
Oct 28, 2022



Abstract:Despite the success of convolutional neural networks for 3D medical-image segmentation, the architectures currently used are still not robust enough to the protocols of different scanners, and the variety of image properties they produce. Moreover, access to large-scale datasets with annotated regions of interest is scarce, and obtaining good results is thus difficult. To overcome these challenges, we introduce IB-U-Nets, a novel architecture with inductive bias, inspired by the visual processing in vertebrates. With the 3D U-Net as the base, we add two 3D residual components to the second encoder blocks. They provide an inductive bias, helping U-Nets to segment anatomical structures from 3D images with increased robustness and accuracy. We compared IB-U-Nets with state-of-the-art 3D U-Nets on multiple modalities and organs, such as the prostate and spleen, using the same training and testing pipeline, including data processing, augmentation and cross-validation. Our results demonstrate the superior robustness and accuracy of IB-U-Nets, especially on small datasets, as is typically the case in medical-image analysis. IB-U-Nets source code and models are publicly available.
Measuring breathing induced oesophageal motion and its dosimetric impact
Oct 19, 2020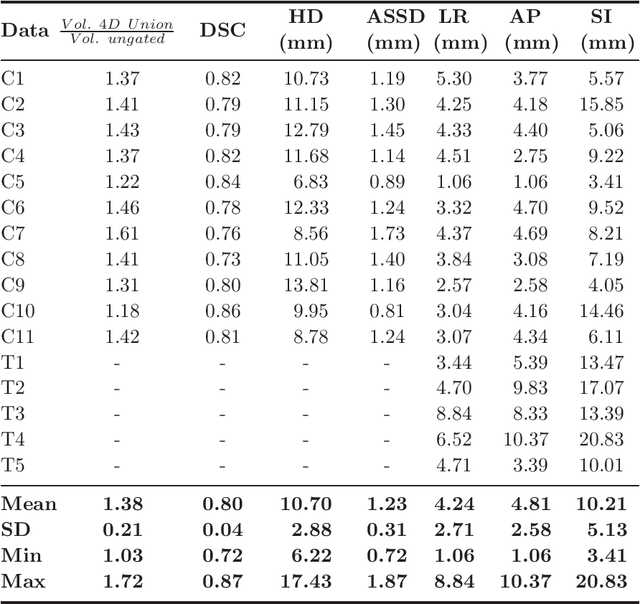
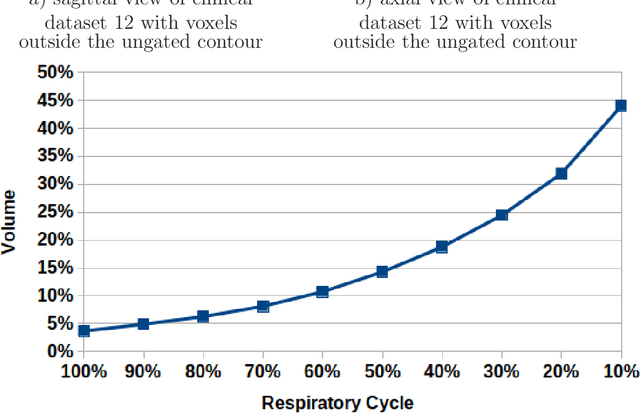
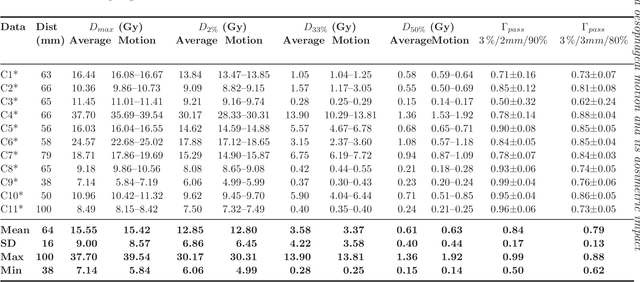
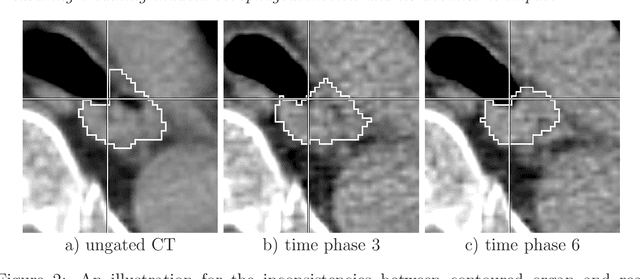
Abstract:Stereotactic body radiation therapy allows for a precise and accurate dose delivery. Organ motion during treatment bares the risk of undetected high dose healthy tissue exposure. An organ very susceptible to high dose is the oesophagus. Its low contrast on CT and the oblong shape renders motion estimation difficult. We tackle this issue by modern algorithms to measure the oesophageal motion voxel-wise and to estimate motion related dosimetric impact. Oesophageal motion was measured using deformable image registration and 4DCT of 11 internal and 5 public datasets. Current clinical practice of contouring the organ on 3DCT was compared to timely resolved 4DCT contours. The dosimetric impact of the motion was estimated by analysing the trajectory of each voxel in the 4D dose distribution. Finally an organ motion model was built, allowing for easier patient-wise comparisons. Motion analysis showed mean absolute maximal motion amplitudes of 4.24 +/- 2.71 mm left-right, 4.81 +/- 2.58 mm anterior-posterior and 10.21 +/- 5.13 mm superior-inferior. Motion between the cohorts differed significantly. In around 50 % of the cases the dosimetric passing criteria was violated. Contours created on 3DCT did not cover 14 % of the organ for 50 % of the respiratory cycle and the 3D contour is around 38 % smaller than the union of all 4D contours. The motion model revealed that the maximal motion is not limited to the lower part of the organ. Our results showed motion amplitudes higher than most reported values in the literature and that motion is very heterogeneous across patients. Therefore, individual motion information should be considered in contouring and planning.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge