Zhipeng Ding
Aladdin: Joint Atlas Building and Diffeomorphic Registration Learning with Pairwise Alignment
Feb 07, 2022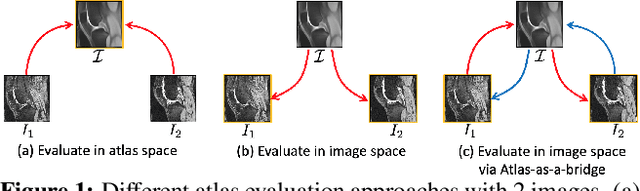
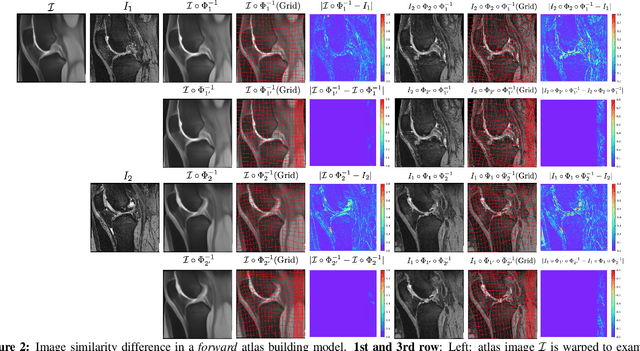
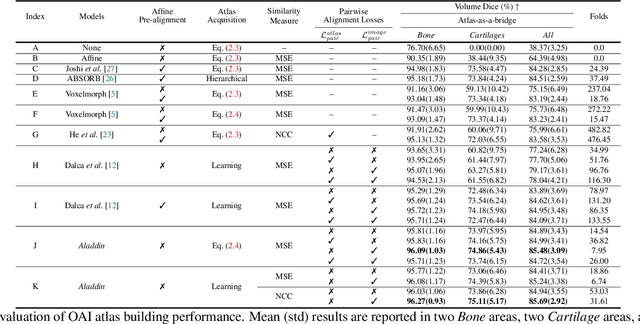
Abstract:Atlas building and image registration are important tasks for medical image analysis. Once one or multiple atlases from an image population have been constructed, commonly (1) images are warped into an atlas space to study intra-subject or inter-subject variations or (2) a possibly probabilistic atlas is warped into image space to assign anatomical labels. Atlas estimation and nonparametric transformations are computationally expensive as they usually require numerical optimization. Additionally, previous approaches for atlas building often define similarity measures between a fuzzy atlas and each individual image, which may cause alignment difficulties because a fuzzy atlas does not exhibit clear anatomical structures in contrast to the individual images. This work explores using a convolutional neural network (CNN) to jointly predict the atlas and a stationary velocity field (SVF) parameterization for diffeomorphic image registration with respect to the atlas. Our approach does not require affine pre-registrations and utilizes pairwise image alignment losses to increase registration accuracy. We evaluate our model on 3D knee magnetic resonance images (MRI) from the OAI-ZIB dataset. Our results show that the proposed framework achieves better performance than other state-of-the-art image registration algorithms, allows for end-to-end training, and for fast inference at test time.
VoteNet++: Registration Refinement for Multi-Atlas Segmentation
Oct 26, 2020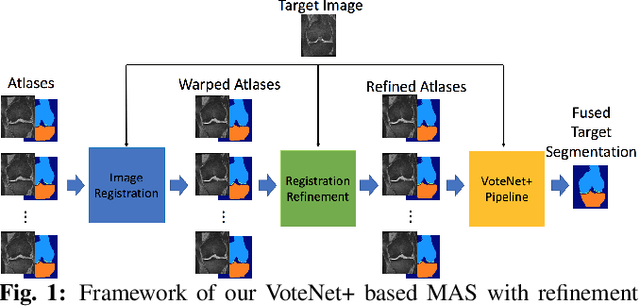
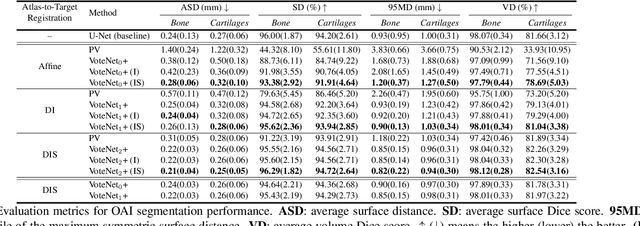
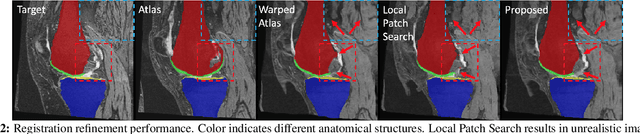
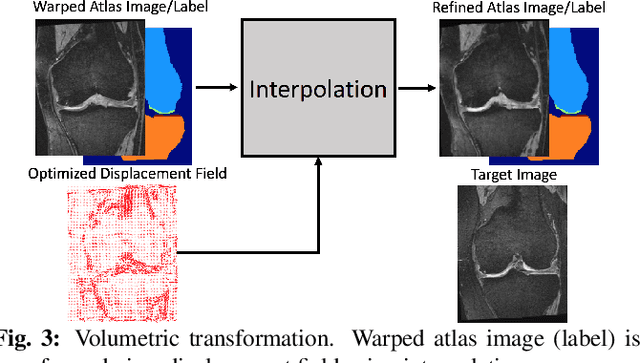
Abstract:Multi-atlas segmentation (MAS) is a popular image segmentation technique for medical images. In this work, we improve the performance of MAS by correcting registration errors before label fusion. Specifically, we use a volumetric displacement field to refine registrations based on image anatomical appearance and predicted labels. We show the influence of the initial spatial alignment as well as the beneficial effect of using label information for MAS performance. Experiments demonstrate that the proposed refinement approach improves MAS performance on a 3D magnetic resonance dataset of the knee.
Local Temperature Scaling for Probability Calibration
Aug 12, 2020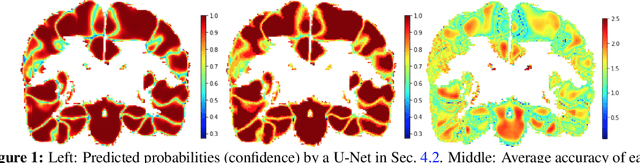
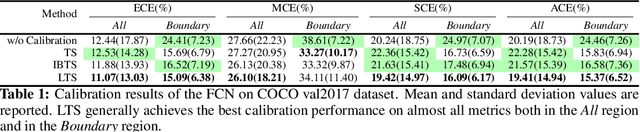
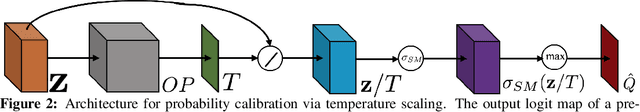
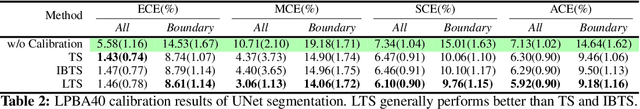
Abstract:For semantic segmentation, label probabilities are often uncalibrated as they are typically only the by-product of a segmentation task. Intersection over Union (IoU) and Dice score are often used as criteria for segmentation success, while metrics related to label probabilities are rarely explored. On the other hand, probability calibration approaches have been studied, which aim at matching probability outputs with experimentally observed errors, but they mainly focus on classification tasks, not on semantic segmentation. Thus, we propose a learning-based calibration method that focuses on multi-label semantic segmentation. Specifically, we adopt a tree-like convolution neural network to predict local temperature values for probability calibration. One advantage of our approach is that it does not change prediction accuracy, hence allowing for calibration as a post-processing step. Experiments on the COCO and LPBA40 datasets demonstrate improved calibration performance over different metrics. We also demonstrate the performance of our method for multi-atlas brain segmentation from magnetic resonance images.
VoteNet+ : An Improved Deep Learning Label Fusion Method for Multi-atlas Segmentation
Nov 01, 2019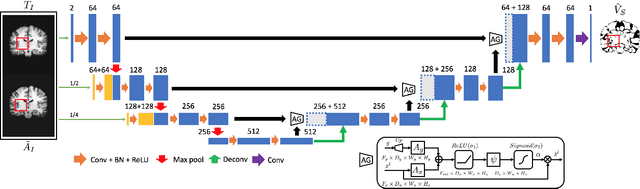
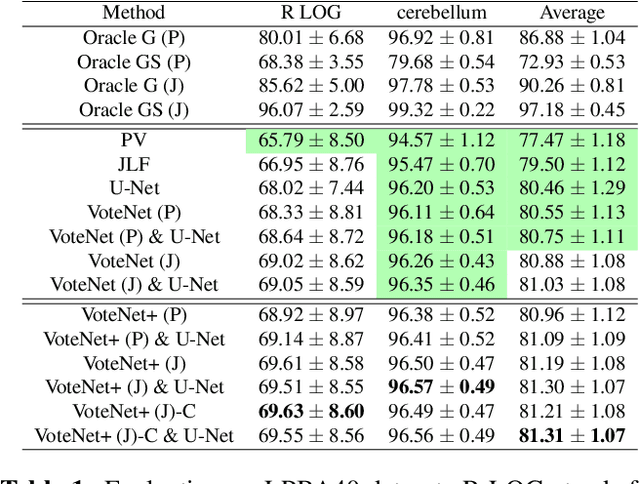
Abstract:In this work, we improve the performance of multi-atlas segmentation (MAS) by integrating the recently proposed VoteNet model with the joint label fusion (JLF) approach. Specifically, we first illustrate that using a deep convolutional neural network to predict atlas probabilities can better distinguish correct atlas labels from incorrect ones than relying on image intensity difference as is typical in JLF. Motivated by this finding, we propose VoteNet+, an improved deep network to locally predict the probability of an atlas label to differs from the label of the target image. Furthermore, we show that JLF is more suitable for the VoteNet framework as a label fusion method than plurality voting. Lastly, we use Platt scaling to calibrate the probabilities of our new model. Results on LPBA40 3D MR brain images show that our proposed method can achieve better performance than VoteNet.
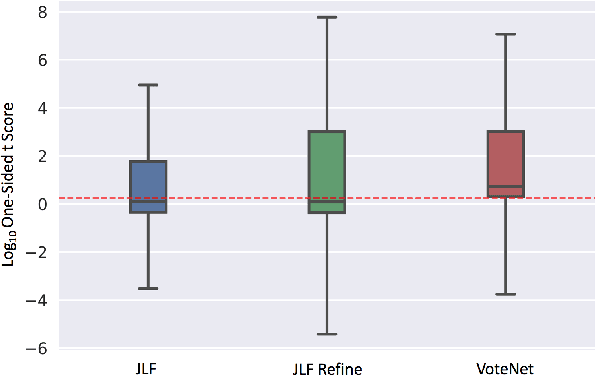
VoteNet: A Deep Learning Label Fusion Method for Multi-Atlas Segmentation
May 31, 2019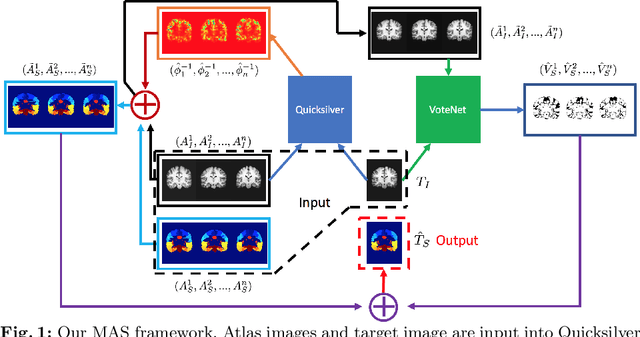
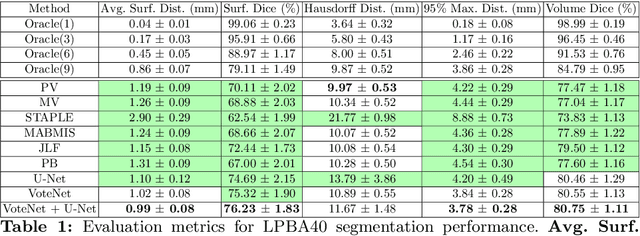
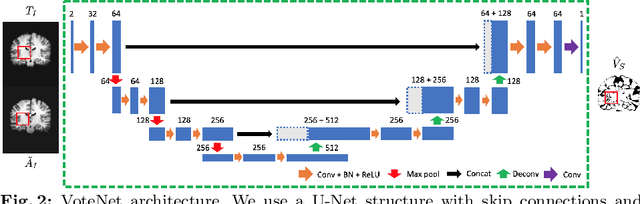
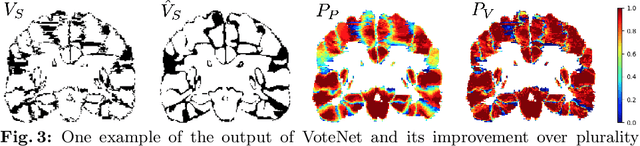
Abstract:Deep learning (DL) approaches are state-of-the-art for many medical image segmentation tasks. They offer a number of advantages: they can be trained for specific tasks, computations are fast at test time, and segmentation quality is typically high. In contrast, previously popular multi-atlas segmentation (MAS) methods are relatively slow (as they rely on costly registrations) and even though sophisticated label fusion strategies have been proposed, DL approaches generally outperform MAS. In this work, we propose a DL-based label fusion strategy (VoteNet) which locally selects a set of reliable atlases whose labels are then fused via plurality voting. Experiments on 3D brain MRI data show that by selecting a good initial atlas set MAS with VoteNet significantly outperforms a number of other label fusion strategies as well as a direct DL segmentation approach. We also provide an experimental analysis of the upper performance bound achievable by our method. While unlikely achievable in practice, this bound suggests room for further performance improvements. Lastly, to address the runtime disadvantage of standard MAS, all our results make use of a fast DL registration approach.
Fast Predictive Simple Geodesic Regression
Nov 15, 2017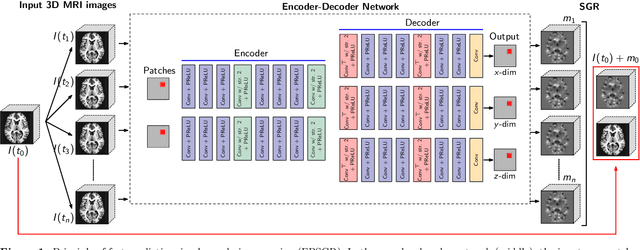
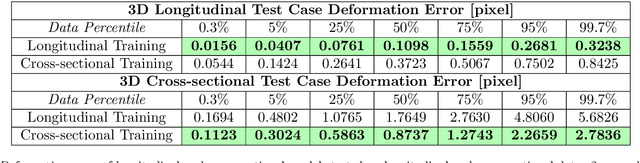
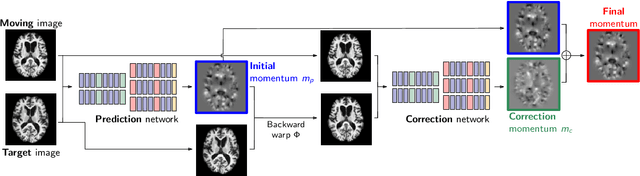
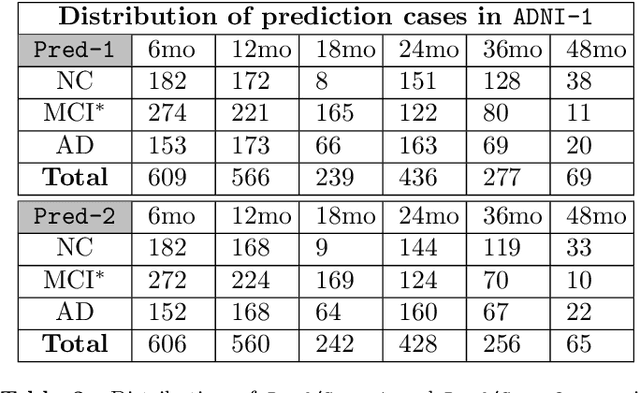
Abstract:Deformable image registration and regression are important tasks in medical image analysis. However, they are computationally expensive, especially when analyzing large-scale datasets that contain thousands of images. Hence, cluster computing is typically used, making the approaches dependent on such computational infrastructure. Even larger computational resources are required as study sizes increase. This limits the use of deformable image registration and regression for clinical applications and as component algorithms for other image analysis approaches. We therefore propose using a fast predictive approach to perform image registrations. In particular, we employ these fast registration predictions to approximate a simplified geodesic regression model to capture longitudinal brain changes. The resulting method is orders of magnitude faster than the standard optimization-based regression model and hence facilitates large-scale analysis on a single graphics processing unit (GPU). We evaluate our results on 3D brain magnetic resonance images (MRI) from the ADNI datasets.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge