Sergio Uribe
Unsupervised 4D Flow MRI Velocity Enhancement and Unwrapping Using Divergence-Free Neural Networks
Mar 31, 2026Abstract:This work introduces an unsupervised Divergence and Aliasing-Free neural network (DAF-FlowNet) for 4D Flow Magnetic Resonance Imaging (4D Flow MRI) that jointly enhances noisy velocity fields and corrects phase wrapping artifacts. DAF-FlowNet parameterizes velocities as the curl of a vector potential, enforcing mass conservation by construction and avoiding explicit divergence-penalty tuning. A cosine data-consistency loss enables simultaneous denoising and unwrapping from wrapped phase images. On synthetic aortic 4D Flow MRI generated from computational fluid dynamics, DAF-FlowNet achieved lower errors than existing techniques (up to 11% lower velocity normalized root mean square error, 11% lower directional error, and 44% lower divergence relative to the best-performing alternative across noise levels), with robustness to moderate segmentation perturbations. For unwrapping, at peak velocity/velocity-encoding ratios of 1.4 and 2.1, DAF-FlowNet achieved 0.18% and 5.2% residual wrapped voxels, representing reductions of 72% and 18% relative to the best alternative method, respectively. In scenarios with both noise and aliasing, the proposed single-stage formulation outperformed a state-of-the-art sequential pipeline (up to 15% lower velocity normalized root mean square error, 11% lower directional error, and 28% lower divergence). Across 10 hypertrophic cardiomyopathy patient datasets, DAF-FlowNet preserved fine-scale flow features, corrected aliased regions, and improved internal flow consistency, as indicated by reduced inter-plane flow bias in aortic and pulmonary mass-conservation analyses recommended by the 4D Flow MRI consensus guidelines. These results support DAF-FlowNet as a framework that unifies velocity enhancement and phase unwrapping to improve the reliability of cardiovascular 4D Flow MRI.
A Physics-based Generative Model to Synthesize Training Datasets for MRI-based Fat Quantification
Dec 11, 2024



Abstract:Deep learning-based techniques have potential to optimize scan and post-processing times required for MRI-based fat quantification, but they are constrained by the lack of large training datasets. Generative models are a promising tool to perform data augmentation by synthesizing realistic datasets. However no previous methods have been specifically designed to generate datasets for quantitative MRI (q-MRI) tasks, where reference quantitative maps and large variability in scanning protocols are usually required. We propose a Physics-Informed Latent Diffusion Model (PI-LDM) to synthesize quantitative parameter maps jointly with customizable MR images by incorporating the signal generation model. We assessed the quality of PI-LDM's synthesized data using metrics such as the Fr\'echet Inception Distance (FID), obtaining comparable scores to state-of-the-art generative methods (FID: 0.0459). We also trained a U-Net for the MRI-based fat quantification task incorporating synthetic datasets. When we used a few real (10 subjects, $~200$ slices) and numerous synthetic samples ($>3000$), fat fraction at specific liver ROIs showed a low bias on data obtained using the same protocol than training data ($0.10\%$ at $\hbox{ROI}_1$, $0.12\%$ at $\hbox{ROI}_2$) and on data acquired with an alternative protocol ($0.14\%$ at $\hbox{ROI}_1$, $0.62\%$ at $\hbox{ROI}_2$). Future work will be to extend PI-LDM to other q-MRI applications.
Physics-informed neural networks for blood flow inverse problems
Aug 02, 2023Abstract:Physics-informed neural networks (PINNs) have emerged as a powerful tool for solving inverse problems, especially in cases where no complete information about the system is known and scatter measurements are available. This is especially useful in hemodynamics since the boundary information is often difficult to model, and high-quality blood flow measurements are generally hard to obtain. In this work, we use the PINNs methodology for estimating reduced-order model parameters and the full velocity field from scatter 2D noisy measurements in the ascending aorta. The results show stable and accurate parameter estimations when using the method with simulated data, while the velocity reconstruction shows dependence on the measurement quality and the flow pattern complexity. The method allows for solving clinical-relevant inverse problems in hemodynamics and complex coupled physical systems.
WarpPINN: Cine-MR image registration with physics-informed neural networks
Nov 22, 2022Abstract:Heart failure is typically diagnosed with a global function assessment, such as ejection fraction. However, these metrics have low discriminate power, failing to distinguish different types of this disease. Quantifying local deformations in the form of cardiac strain can provide helpful information, but it remains a challenge. In this work, we introduce WarpPINN, a physics-informed neural network to perform image registration to obtain local metrics of the heart deformation. We apply this method to cine magnetic resonance images to estimate the motion during the cardiac cycle. We inform our neural network of near-incompressibility of cardiac tissue by penalizing the jacobian of the deformation field. The loss function has two components: an intensity-based similarity term between the reference and the warped template images, and a regularizer that represents the hyperelastic behavior of the tissue. The architecture of the neural network allows us to easily compute the strain via automatic differentiation to assess cardiac activity. We use Fourier feature mappings to overcome the spectral bias of neural networks, allowing us to capture discontinuities in the strain field. We test our algorithm on a synthetic example and on a cine-MRI benchmark of 15 healthy volunteers. We outperform current methodologies both landmark tracking and strain estimation. We expect that WarpPINN will enable more precise diagnostics of heart failure based on local deformation information. Source code is available at https://github.com/fsahli/WarpPINN.
Inspecting state of the art performance and NLP metrics in image-based medical report generation
Nov 21, 2020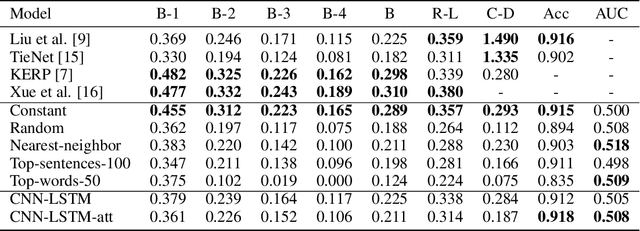
Abstract:Several deep learning architectures have been proposed over the last years to deal with the problem of generating a written report given an imaging exam as input. Most works evaluate the generated reports using standard Natural Language Processing (NLP) metrics (e.g. BLEU, ROUGE), reporting significant progress. In this article, we contrast this progress by comparing state of the art (SOTA) models against weak baselines. We show that simple and even naive approaches yield near SOTA performance on most traditional NLP metrics. We conclude that evaluation methods in this task should be further studied towards correctly measuring clinical accuracy, ideally involving physicians to contribute to this end.
A Survey on Deep Learning and Explainability for Automatic Image-based Medical Report Generation
Oct 20, 2020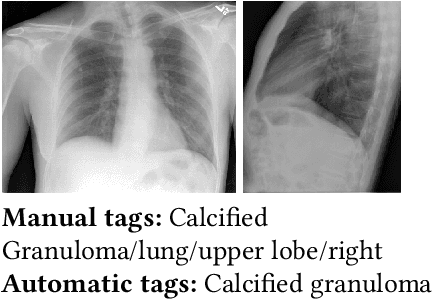
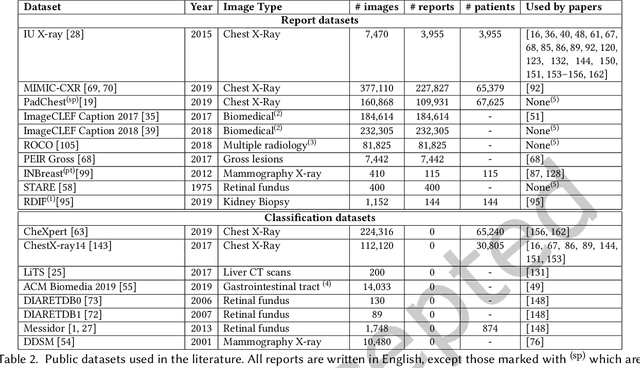
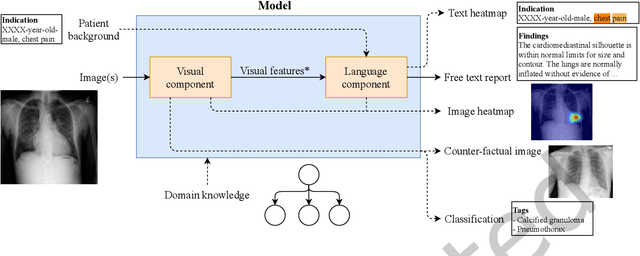
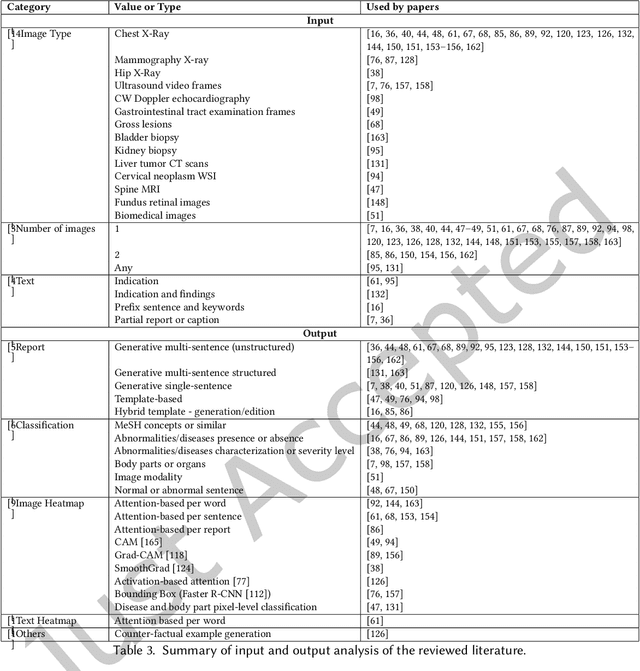
Abstract:Every year physicians face an increasing demand of image-based diagnosis from patients, a problem that can be addressed with recent artificial intelligence methods. In this context, we survey works in the area of automatic report generation from medical images, with emphasis on methods using deep neural networks, with respect to: (1) Datasets, (2) Architecture Design, (3) Explainability and (4) Evaluation Metrics. Our survey identifies interesting developments, but also remaining challenges. Among them, the current evaluation of generated reports is especially weak, since it mostly relies on traditional Natural Language Processing (NLP) metrics, which do not accurately capture medical correctness.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge