Rema Padman
The Model Says Walk: How Surface Heuristics Override Implicit Constraints in LLM Reasoning
Mar 30, 2026Abstract:Large language models systematically fail when a salient surface cue conflicts with an unstated feasibility constraint. We study this through a diagnose-measure-bridge-treat framework. Causal-behavioral analysis of the ``car wash problem'' across six models reveals approximately context-independent sigmoid heuristics: the distance cue exerts 8.7 to 38 times more influence than the goal, and token-level attribution shows patterns more consistent with keyword associations than compositional inference. The Heuristic Override Benchmark (HOB) -- 500 instances spanning 4 heuristic by 5 constraint families with minimal pairs and explicitness gradients -- demonstrates generality across 14 models: under strict evaluation (10/10 correct), no model exceeds 75%, and presence constraints are hardest (44%). A minimal hint (e.g., emphasizing the key object) recovers +15 pp on average, suggesting the failure lies in constraint inference rather than missing knowledge; 12/14 models perform worse when the constraint is removed (up to -39 pp), revealing conservative bias. Parametric probes confirm that the sigmoid pattern generalizes to cost, efficiency, and semantic-similarity heuristics; goal-decomposition prompting recovers +6 to 9 pp by forcing models to enumerate preconditions before answering. Together, these results characterize heuristic override as a systematic reasoning vulnerability and provide a benchmark for measuring progress toward resolving it.
When Documents Disagree: Measuring Institutional Variation in Transplant Guidance with Retrieval-Augmented Language Models
Mar 23, 2026Abstract:Patient education materials for solid-organ transplantation vary substantially across U.S. centers, yet no systematic method exists to quantify this heterogeneity at scale. We introduce a framework that grounds the same patient questions in different centers' handbooks using retrieval-augmented language models and compares the resulting answers using a five-label consistency taxonomy. Applied to 102 handbooks from 23 centers and 1,115 benchmark questions, the framework quantifies heterogeneity across four dimensions: question, topic, organ, and center. We find that 20.8% of non-absent pairwise comparisons exhibit clinically meaningful divergence, concentrated in condition monitoring and lifestyle topics. Coverage gaps are even more prominent: 96.2% of question-handbook pairs miss relevant content, with reproductive health at 95.1% absence. Center-level divergence profiles are stable and interpretable, where heterogeneity reflects systematic institutional differences, likely due to patient diversity. These findings expose an information gap in transplant patient education materials, with document-grounded medical question answering highlighting opportunities for content improvement.
Consistency of Large Reasoning Models Under Multi-Turn Attacks
Feb 16, 2026Abstract:Large reasoning models with reasoning capabilities achieve state-of-the-art performance on complex tasks, but their robustness under multi-turn adversarial pressure remains underexplored. We evaluate nine frontier reasoning models under adversarial attacks. Our findings reveal that reasoning confers meaningful but incomplete robustness: most reasoning models studied significantly outperform instruction-tuned baselines, yet all exhibit distinct vulnerability profiles, with misleading suggestions universally effective and social pressure showing model-specific efficacy. Through trajectory analysis, we identify five failure modes (Self-Doubt, Social Conformity, Suggestion Hijacking, Emotional Susceptibility, and Reasoning Fatigue) with the first two accounting for 50% of failures. We further demonstrate that Confidence-Aware Response Generation (CARG), effective for standard LLMs, fails for reasoning models due to overconfidence induced by extended reasoning traces; counterintuitively, random confidence embedding outperforms targeted extraction. Our results highlight that reasoning capabilities do not automatically confer adversarial robustness and that confidence-based defenses require fundamental redesign for reasoning models.
Closing Reasoning Gaps in Clinical Agents with Differential Reasoning Learning
Feb 10, 2026Abstract:Clinical decision support requires not only correct answers but also clinically valid reasoning. We propose Differential Reasoning Learning (DRL), a framework that improves clinical agents by learning from reasoning discrepancies. From reference reasoning rationales (e.g., physician-authored clinical rationale, clinical guidelines, or outputs from more capable models) and the agent's free-form chain-of-thought (CoT), DRL extracts reasoning graphs as directed acyclic graphs (DAGs) and performs a clinically weighted graph edit distance (GED)-based discrepancy analysis. An LLM-as-a-judge aligns semantically equivalent nodes and diagnoses discrepancies between graphs. These graph-level discrepancy diagnostics are converted into natural-language instructions and stored in a Differential Reasoning Knowledge Base (DR-KB). At inference, we retrieve top-$k$ instructions via Retrieval-Augmented Generation (RAG) to augment the agent prompt and patch likely logic gaps. Evaluation on open medical question answering (QA) benchmarks and a Return Visit Admissions (RVA) prediction task from internal clinical data demonstrates gains over baselines, improving both final-answer accuracy and reasoning fidelity. Ablation studies confirm gains from infusing reference reasoning rationales and the top-$k$ retrieval strategy. Clinicians' review of the output provides further assurance of the approach. Together, results suggest that DRL supports more reliable clinical decision-making in complex reasoning scenarios and offers a practical mechanism for deployment under limited token budgets.
Benchmarking Waitlist Mortality Prediction in Heart Transplantation Through Time-to-Event Modeling using New Longitudinal UNOS Dataset
Jul 09, 2025Abstract:Decisions about managing patients on the heart transplant waitlist are currently made by committees of doctors who consider multiple factors, but the process remains largely ad-hoc. With the growing volume of longitudinal patient, donor, and organ data collected by the United Network for Organ Sharing (UNOS) since 2018, there is increasing interest in analytical approaches to support clinical decision-making at the time of organ availability. In this study, we benchmark machine learning models that leverage longitudinal waitlist history data for time-dependent, time-to-event modeling of waitlist mortality. We train on 23,807 patient records with 77 variables and evaluate both survival prediction and discrimination at a 1-year horizon. Our best model achieves a C-Index of 0.94 and AUROC of 0.89, significantly outperforming previous models. Key predictors align with known risk factors while also revealing novel associations. Our findings can support urgency assessment and policy refinement in heart transplant decision making.
Beyond Single-Turn: A Survey on Multi-Turn Interactions with Large Language Models
Apr 08, 2025Abstract:Recent advancements in large language models (LLMs) have revolutionized their ability to handle single-turn tasks, yet real-world applications demand sophisticated multi-turn interactions. This survey provides a comprehensive review of recent advancements in evaluating and enhancing multi-turn interactions in LLMs. Focusing on task-specific scenarios, from instruction following in diverse domains such as math and coding to complex conversational engagements in roleplay, healthcare, education, and even adversarial jailbreak settings, we systematically examine the challenges of maintaining context, coherence, fairness, and responsiveness over prolonged dialogues. The paper organizes current benchmarks and datasets into coherent categories that reflect the evolving landscape of multi-turn dialogue evaluation. In addition, we review a range of enhancement methodologies under multi-turn settings, including model-centric strategies (contextual learning, supervised fine-tuning, reinforcement learning, and new architectures), external integration approaches (memory-augmented, retrieval-based methods, and knowledge graph), and agent-based techniques for collaborative interactions. Finally, we discuss open challenges and propose future directions for research to further advance the robustness and effectiveness of multi-turn interactions in LLMs. Related resources and papers are available at https://github.com/yubol-cmu/Awesome-Multi-Turn-LLMs.
Can LLMs Support Medical Knowledge Imputation? An Evaluation-Based Perspective
Mar 29, 2025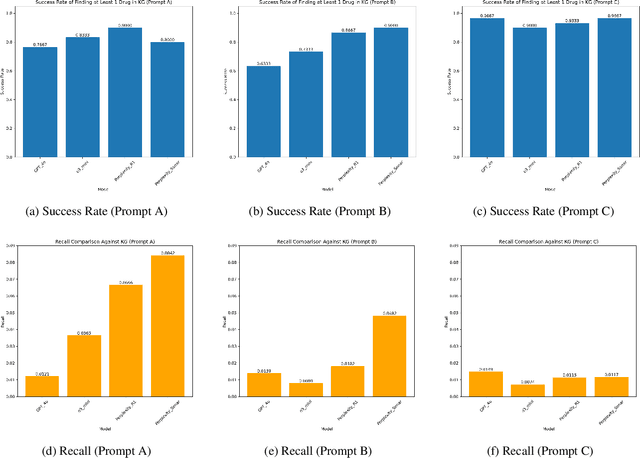
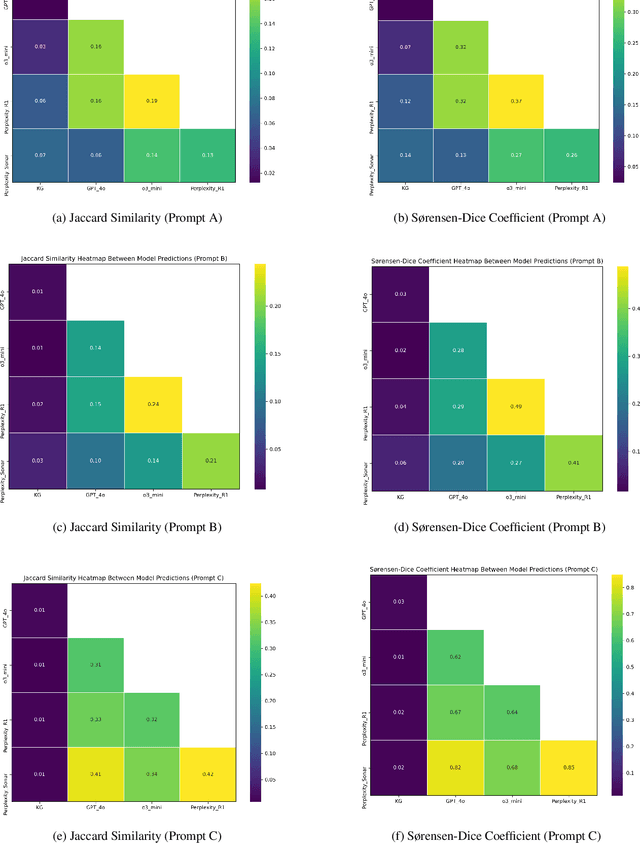
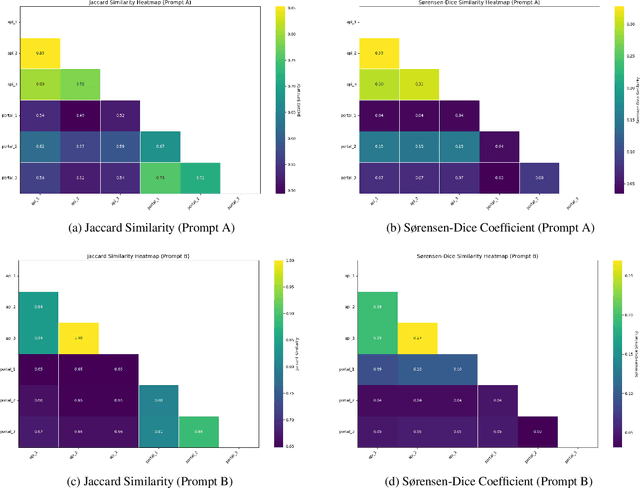
Abstract:Medical knowledge graphs (KGs) are essential for clinical decision support and biomedical research, yet they often exhibit incompleteness due to knowledge gaps and structural limitations in medical coding systems. This issue is particularly evident in treatment mapping, where coding systems such as ICD, Mondo, and ATC lack comprehensive coverage, resulting in missing or inconsistent associations between diseases and their potential treatments. To address this issue, we have explored the use of Large Language Models (LLMs) for imputing missing treatment relationships. Although LLMs offer promising capabilities in knowledge augmentation, their application in medical knowledge imputation presents significant risks, including factual inaccuracies, hallucinated associations, and instability between and within LLMs. In this study, we systematically evaluate LLM-driven treatment mapping, assessing its reliability through benchmark comparisons. Our findings highlight critical limitations, including inconsistencies with established clinical guidelines and potential risks to patient safety. This study serves as a cautionary guide for researchers and practitioners, underscoring the importance of critical evaluation and hybrid approaches when leveraging LLMs to enhance treatment mappings on medical knowledge graphs.
Firm or Fickle? Evaluating Large Language Models Consistency in Sequential Interactions
Mar 28, 2025



Abstract:Large Language Models (LLMs) have shown remarkable capabilities across various tasks, but their deployment in high-stake domains requires consistent performance across multiple interaction rounds. This paper introduces a comprehensive framework for evaluating and improving LLM response consistency, making three key contributions. First, we propose a novel Position-Weighted Consistency (PWC) score that captures both the importance of early-stage stability and recovery patterns in multi-turn interactions. Second, we present a carefully curated benchmark dataset spanning diverse domains and difficulty levels, specifically designed to evaluate LLM consistency under various challenging follow-up scenarios. Third, we introduce Confidence-Aware Response Generation (CARG), a framework that significantly improves response stability by incorporating model confidence signals into the generation process. Empirical results demonstrate that CARG significantly improves response stability without sacrificing accuracy, underscoring its potential for reliable LLM deployment in critical applications.
No Black Box Anymore: Demystifying Clinical Predictive Modeling with Temporal-Feature Cross Attention Mechanism
Mar 26, 2025



Abstract:Despite the outstanding performance of deep learning models in clinical prediction tasks, explainability remains a significant challenge. Inspired by transformer architectures, we introduce the Temporal-Feature Cross Attention Mechanism (TFCAM), a novel deep learning framework designed to capture dynamic interactions among clinical features across time, enhancing both predictive accuracy and interpretability. In an experiment with 1,422 patients with Chronic Kidney Disease, predicting progression to End-Stage Renal Disease, TFCAM outperformed LSTM and RETAIN baselines, achieving an AUROC of 0.95 and an F1-score of 0.69. Beyond performance gains, TFCAM provides multi-level explainability by identifying critical temporal periods, ranking feature importance, and quantifying how features influence each other across time before affecting predictions. Our approach addresses the "black box" limitations of deep learning in healthcare, offering clinicians transparent insights into disease progression mechanisms while maintaining state-of-the-art predictive performance.
Enhancing End Stage Renal Disease Outcome Prediction: A Multi-Sourced Data-Driven Approach
Oct 02, 2024Abstract:Objective: To improve prediction of Chronic Kidney Disease (CKD) progression to End Stage Renal Disease (ESRD) using machine learning (ML) and deep learning (DL) models applied to an integrated clinical and claims dataset of varying observation windows, supported by explainable AI (XAI) to enhance interpretability and reduce bias. Materials and Methods: We utilized data about 10,326 CKD patients, combining their clinical and claims information from 2009 to 2018. Following data preprocessing, cohort identification, and feature engineering, we evaluated multiple statistical, ML and DL models using data extracted from five distinct observation windows. Feature importance and Shapley value analysis were employed to understand key predictors. Models were tested for robustness, clinical relevance, misclassification errors and bias issues. Results: Integrated data models outperformed those using single data sources, with the Long Short-Term Memory (LSTM) model achieving the highest AUC (0.93) and F1 score (0.65). A 24-month observation window was identified as optimal for balancing early detection and prediction accuracy. The 2021 eGFR equation improved prediction accuracy and reduced racial bias, notably for African American patients. Discussion: Improved ESRD prediction accuracy, results interpretability and bias mitigation strategies presented in this study have the potential to significantly enhance CKD and ESRD management, support targeted early interventions and reduce healthcare disparities. Conclusion: This study presents a robust framework for predicting ESRD outcomes in CKD patients, improving clinical decision-making and patient care through multi-sourced, integrated data and AI/ML methods. Future research will expand data integration and explore the application of this framework to other chronic diseases.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge