Jiaxiang Jiang
Synthetic Data Generation for Long-Tail Medical Image Classification: A Case Study in Skin Lesions
May 04, 2026Abstract:Long-tailed class distributions are pervasive in multi-class medical datasets and pose significant challenges for deep learning models which typically underperform on tail classes with limited samples. This limitation is particularly problematic in medical applications, where rare classes often correspond to severe or high-risk diseases and therefore require high diagnostic accuracy. Existing solutions-including specialized architectures, rebalanced loss functions, and handcrafted data augmentation-offer only marginal improvements and struggle to scale due to their limited and largely deterministic variability. To address these challenges, we introduce a diffusion-model-driven synthetic data augmentation pipeline tailored for medical long-tailed classification. Our approach features a novel inpainting diffusion model combined with an Out-of-Distribution (OOD) post-selection mechanism to ensure diverse, realistic, and clinically meaningful synthetic samples. Evaluated on the ISIC2019 skin lesion classification dataset, one of the largest and most imbalanced medical imaging benchmarks, our method yields substantial improvements in overall performance, with particularly pronounced gains on tail classes with more than $28\%$ improvement on the class with the fewest samples. These results demonstrate the effectiveness of diffusion-based augmentation in mitigating long-tail imbalance and enhancing medical classification robustness.
3D Neuron Morphology Analysis
Dec 14, 2022



Abstract:We consider the problem of finding an accurate representation of neuron shapes, extracting sub-cellular features, and classifying neurons based on neuron shapes. In neuroscience research, the skeleton representation is often used as a compact and abstract representation of neuron shapes. However, existing methods are limited to getting and analyzing "curve" skeletons which can only be applied for tubular shapes. This paper presents a 3D neuron morphology analysis method for more general and complex neuron shapes. First, we introduce the concept of skeleton mesh to represent general neuron shapes and propose a novel method for computing mesh representations from 3D surface point clouds. A skeleton graph is then obtained from skeleton mesh and is used to extract sub-cellular features. Finally, an unsupervised learning method is used to embed the skeleton graph for neuron classification. Extensive experiment results are provided and demonstrate the robustness of our method to analyze neuron morphology.
Deep Learning Enabled Time-Lapse 3D Cell Analysis
Aug 17, 2022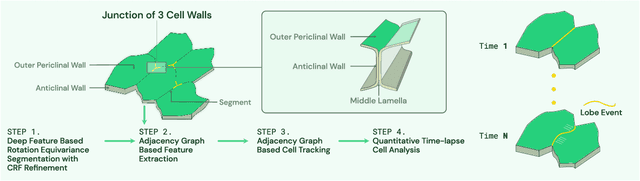
Abstract:This paper presents a method for time-lapse 3D cell analysis. Specifically, we consider the problem of accurately localizing and quantitatively analyzing sub-cellular features, and for tracking individual cells from time-lapse 3D confocal cell image stacks. The heterogeneity of cells and the volume of multi-dimensional images presents a major challenge for fully automated analysis of morphogenesis and development of cells. This paper is motivated by the pavement cell growth process, and building a quantitative morphogenesis model. We propose a deep feature based segmentation method to accurately detect and label each cell region. An adjacency graph based method is used to extract sub-cellular features of the segmented cells. Finally, the robust graph based tracking algorithm using multiple cell features is proposed for associating cells at different time instances. Extensive experiment results are provided and demonstrate the robustness of the proposed method. The code is available on Github and the method is available as a service through the BisQue portal.
Semi supervised segmentation and graph-based tracking of 3D nuclei in time-lapse microscopy
Oct 26, 2020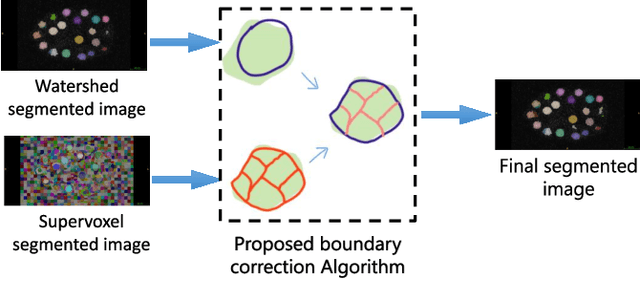


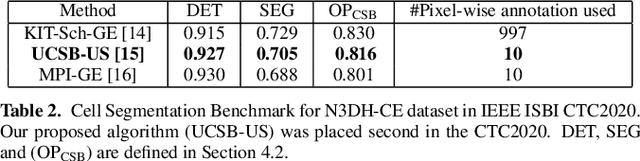
Abstract:We propose a novel weakly supervised method to improve the boundary of the 3D segmented nuclei utilizing an over-segmented image. This is motivated by the observation that current state-of-the-art deep learning methods do not result in accurate boundaries when the training data is weakly annotated. Towards this, a 3D U-Net is trained to get the centroid of the nuclei and integrated with a simple linear iterative clustering (SLIC) supervoxel algorithm that provides better adherence to cluster boundaries. To track these segmented nuclei, our algorithm utilizes the relative nuclei location depicting the processes of nuclei division and apoptosis. The proposed algorithmic pipeline achieves better segmentation performance compared to the state-of-the-art method in Cell Tracking Challenge (CTC) 2019 and comparable performance to state-of-the-art methods in IEEE ISBI CTC2020 while utilizing very few pixel-wise annotated data. Detailed experimental results are provided, and the source code is available on GitHub.
Accurate 3D Cell Segmentation using Deep Feature and CRF Refinement
Feb 13, 2019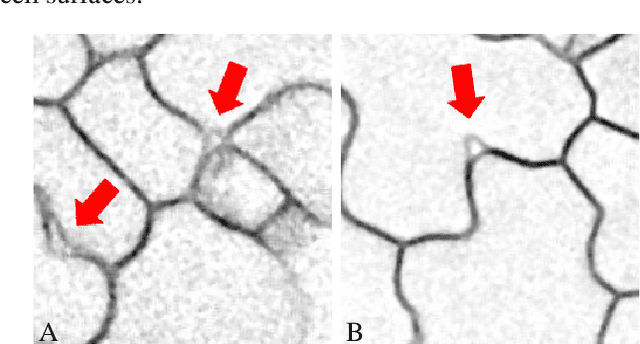
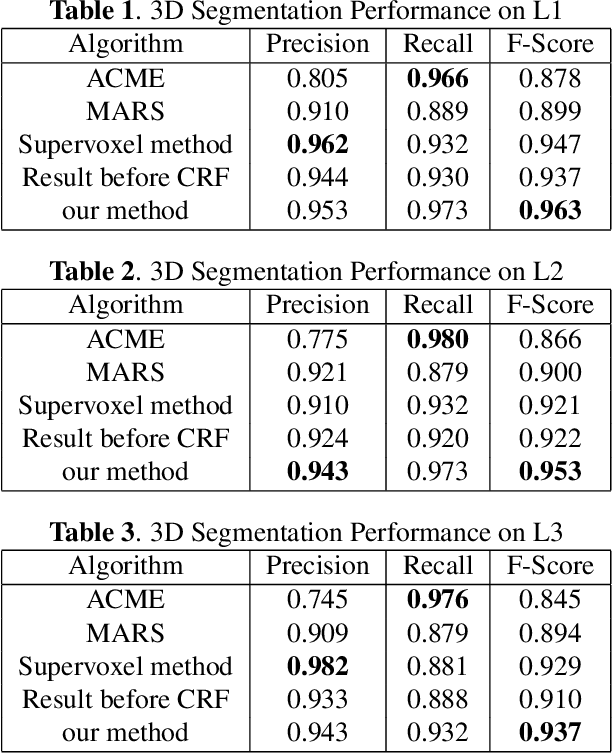

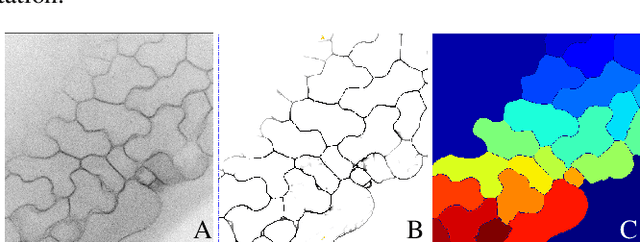
Abstract:We consider the problem of accurately identifying cell boundaries and labeling individual cells in confocal microscopy images, specifically, 3D image stacks of cells with tagged cell membranes. Precise identification of cell boundaries, their shapes, and quantifying inter-cellular space leads to a better understanding of cell morphogenesis. Towards this, we outline a cell segmentation method that uses a deep neural network architecture to extract a confidence map of cell boundaries, followed by a 3D watershed algorithm and a final refinement using a conditional random field. In addition to improving the accuracy of segmentation compared to other state-of-the-art methods, the proposed approach also generalizes well to different datasets without the need to retrain the network for each dataset. Detailed experimental results are provided, and the source code is available on GitHub.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge