Jiajun Cheng
Bridging Restoration and Diagnosis: A Comprehensive Benchmark for Retinal Fundus Enhancement
Apr 04, 2026Abstract:Over the past decade, generative models have demonstrated success in enhancing fundus images. However, the evaluation of these models remains a challenge. A benchmark for fundus image enhancement is needed for three main reasons:(1) Conventional denoising metrics such as PSNR and SSIM fail to capture clinically relevant features, such as lesion preservation and vessel morphology consistency, limiting their applicability in real-world settings; (2) There is a lack of unified evaluation protocols that address both paired and unpaired enhancement methods, particularly those guided by clinical expertise; and (3) An evaluation framework should provide actionable insights to guide future advancements in clinically aligned enhancement models. To address these gaps, we introduce EyeBench-V2, a benchmark designed to bridge the gap between enhancement model performance and clinical utility. Our work offers three key contributions:(1) Multi-dimensional clinical-alignment through downstream evaluations: Beyond standard enhancement metrics, we assess performance across clinically meaningful tasks including vessel segmentation, diabetic retinopathy (DR) grading, generalization to unseen noise patterns, and lesion segmentation. (2) Expert-guided evaluation design: We curate a novel dataset enabling fair comparisons between paired and unpaired enhancement methods, accompanied by a structured manual assessment protocol by medical experts, which evaluates clinically critical aspects such as lesion structure alterations, background color shifts, and the introduction of artificial structures. (3) Actionable insights: Our benchmark provides a rigorous, task-oriented analysis of existing generative models, equipping clinical researchers with the evidence needed to make informed decisions, while also identifying limitations in current methods to inform the design of next-generation enhancement models.
Benchmarking performance, explainability, and evaluation strategies of vision-language models for surgery: Challenges and opportunities
May 16, 2025


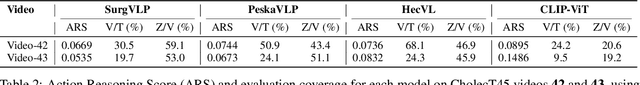
Abstract:Minimally invasive surgery (MIS) presents significant visual and technical challenges, including surgical instrument classification and understanding surgical action involving instruments, verbs, and anatomical targets. While many machine learning-based methods have been developed for surgical understanding, they typically rely on procedure- and task-specific models trained on small, manually annotated datasets. In contrast, the recent success of vision-language models (VLMs) trained on large volumes of raw image-text pairs has demonstrated strong adaptability to diverse visual data and a range of downstream tasks. This opens meaningful research questions: how well do these general-purpose VLMs perform in the surgical domain? In this work, we explore those questions by benchmarking several VLMs across diverse surgical datasets, including general laparoscopic procedures and endoscopic submucosal dissection, to assess their current capabilities and limitations. Our benchmark reveals key gaps in the models' ability to consistently link language to the correct regions in surgical scenes.
Learning Networks from Wide-Sense Stationary Stochastic Processes
Dec 04, 2024Abstract:Complex networked systems driven by latent inputs are common in fields like neuroscience, finance, and engineering. A key inference problem here is to learn edge connectivity from node outputs (potentials). We focus on systems governed by steady-state linear conservation laws: $X_t = {L^{\ast}}Y_{t}$, where $X_t, Y_t \in \mathbb{R}^p$ denote inputs and potentials, respectively, and the sparsity pattern of the $p \times p$ Laplacian $L^{\ast}$ encodes the edge structure. Assuming $X_t$ to be a wide-sense stationary stochastic process with a known spectral density matrix, we learn the support of $L^{\ast}$ from temporally correlated samples of $Y_t$ via an $\ell_1$-regularized Whittle's maximum likelihood estimator (MLE). The regularization is particularly useful for learning large-scale networks in the high-dimensional setting where the network size $p$ significantly exceeds the number of samples $n$. We show that the MLE problem is strictly convex, admitting a unique solution. Under a novel mutual incoherence condition and certain sufficient conditions on $(n, p, d)$, we show that the ML estimate recovers the sparsity pattern of $L^\ast$ with high probability, where $d$ is the maximum degree of the graph underlying $L^{\ast}$. We provide recovery guarantees for $L^\ast$ in element-wise maximum, Frobenius, and operator norms. Finally, we complement our theoretical results with several simulation studies on synthetic and benchmark datasets, including engineered systems (power and water networks), and real-world datasets from neural systems (such as the human brain).
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge