Jens Rittscher
Conv2Warp: An unsupervised deformable image registration with continuous convolution and warping
Aug 16, 2019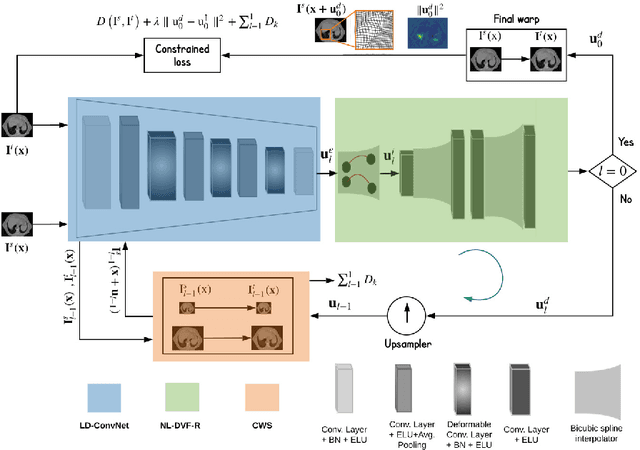
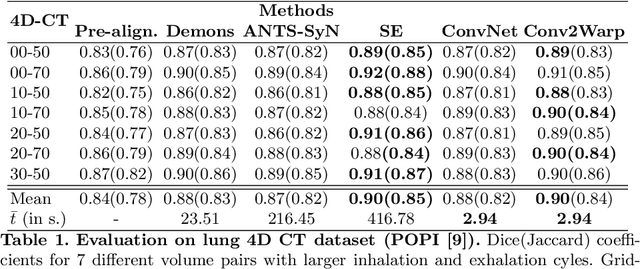
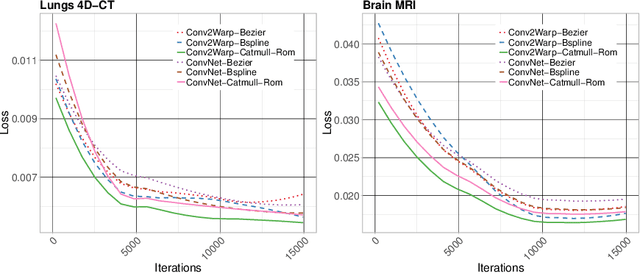
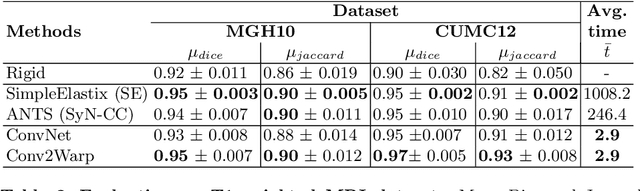
Abstract:Recent successes in deep learning based deformable image registration (DIR) methods have demonstrated that complex deformation can be learnt directly from data while reducing computation time when compared to traditional methods. However, the reliance on fully linear convolutional layers imposes a uniform sampling of pixel/voxel locations which ultimately limits their performance. To address this problem, we propose a novel approach of learning a continuous warp of the source image. Here, the required deformation vector fields are obtained from a concatenated linear and non-linear convolution layers and a learnable bicubic Catmull-Rom spline resampler. This allows to compute smooth deformation field and more accurate alignment compared to using only linear convolutions and linear resampling. In addition, the continuous warping technique penalizes disagreements that are due to topological changes. Our experiments demonstrate that this approach manages to capture large non-linear deformations and minimizes the propagation of interpolation errors. While improving accuracy the method is computationally efficient. We present comparative results on a range of public 4D CT lung (POPI) and brain datasets (CUMC12, MGH10).
Ink removal from histopathology whole slide images by combining classification, detection and image generation models
May 10, 2019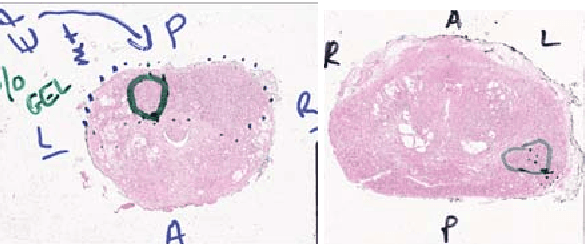
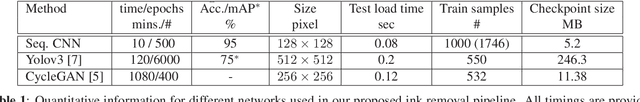

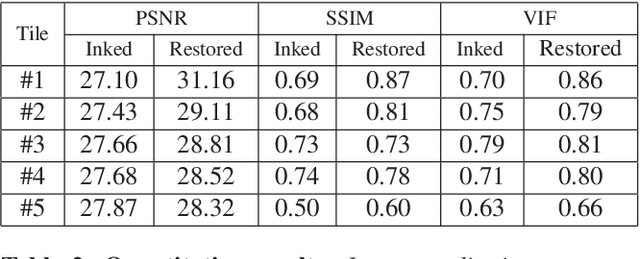
Abstract:Histopathology slides are routinely marked by pathologists using permanent ink markers that should not be removed as they form part of the medical record. Often tumour regions are marked up for the purpose of highlighting features or other downstream processing such an gene sequencing. Once digitised there is no established method for removing this information from the whole slide images limiting its usability in research and study. Removal of marker ink from these high-resolution whole slide images is non-trivial and complex problem as they contaminate different regions and in an inconsistent manner. We propose an efficient pipeline using convolution neural networks that results in ink-free images without compromising information and image resolution. Our pipeline includes a sequential classical convolution neural network for accurate classification of contaminated image tiles, a fast region detector and a domain adaptive cycle consistent adversarial generative model for restoration of foreground pixels. Both quantitative and qualitative results on four different whole slide images show that our approach yields visually coherent ink-free whole slide images.
Efficient video indexing for monitoring disease activity and progression in the upper gastrointestinal tract
May 10, 2019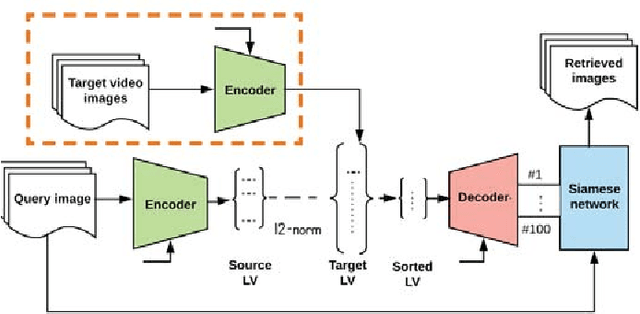
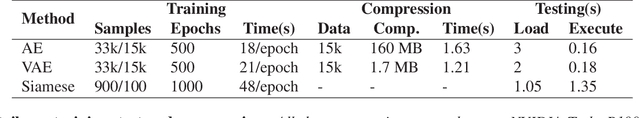
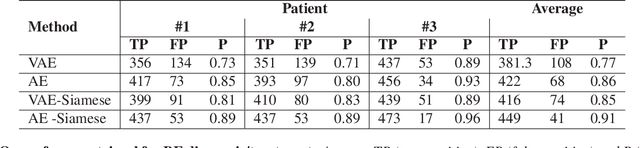
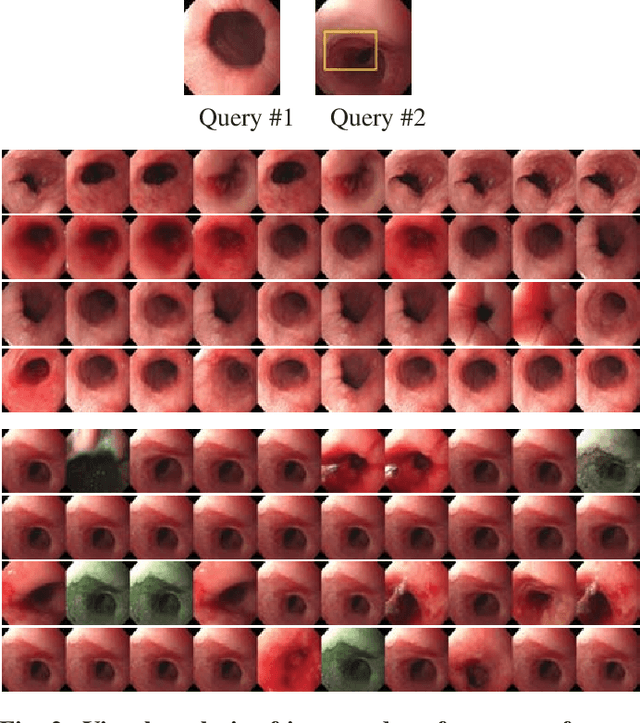
Abstract:Endoscopy is a routine imaging technique used for both diagnosis and minimally invasive surgical treatment. While the endoscopy video contains a wealth of information, tools to capture this information for the purpose of clinical reporting are rather poor. In date, endoscopists do not have any access to tools that enable them to browse the video data in an efficient and user friendly manner. Fast and reliable video retrieval methods could for example, allow them to review data from previous exams and therefore improve their ability to monitor disease progression. Deep learning provides new avenues of compressing and indexing video in an extremely efficient manner. In this study, we propose to use an autoencoder for efficient video compression and fast retrieval of video images. To boost the accuracy of video image retrieval and to address data variability like multi-modality and view-point changes, we propose the integration of a Siamese network. We demonstrate that our approach is competitive in retrieving images from 3 large scale videos of 3 different patients obtained against the query samples of their previous diagnosis. Quantitative validation shows that the combined approach yield an overall improvement of 5% and 8% over classical and variational autoencoders, respectively.
Endoscopy artifact detection (EAD 2019) challenge dataset
May 08, 2019
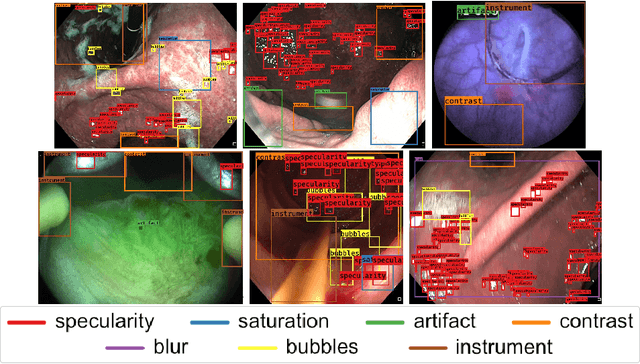
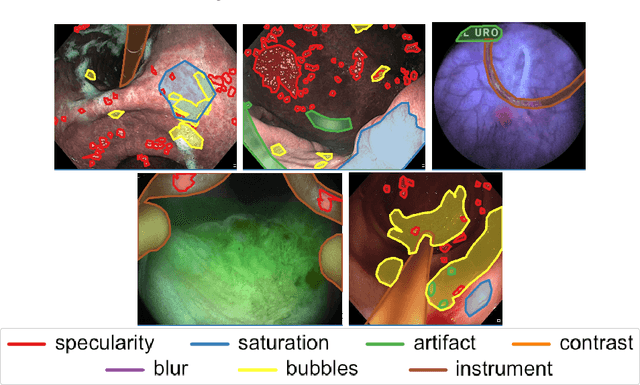
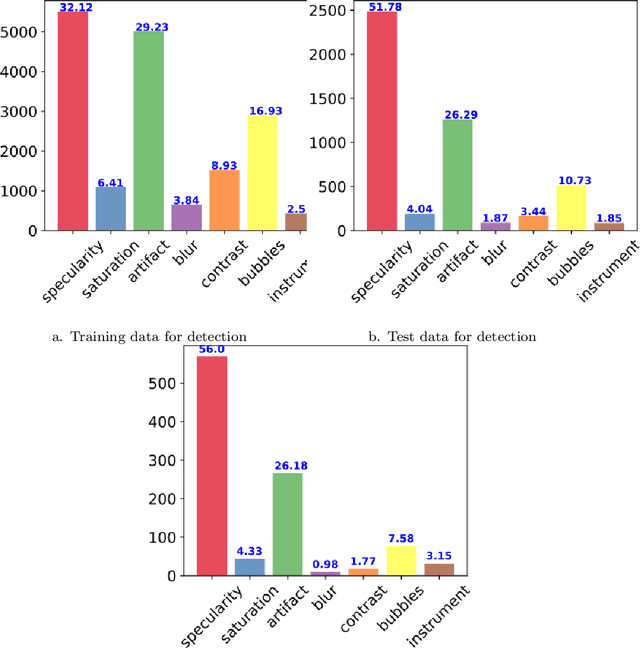
Abstract:Endoscopic artifacts are a core challenge in facilitating the diagnosis and treatment of diseases in hollow organs. Precise detection of specific artifacts like pixel saturations, motion blur, specular reflections, bubbles and debris is essential for high-quality frame restoration and is crucial for realizing reliable computer-assisted tools for improved patient care. At present most videos in endoscopy are currently not analyzed due to the abundant presence of multi-class artifacts in video frames. Through the endoscopic artifact detection (EAD 2019) challenge, we address this key bottleneck problem by solving the accurate identification and localization of endoscopic frame artifacts to enable further key quantitative analysis of unusable video frames such as mosaicking and 3D reconstruction which is crucial for delivering improved patient care. This paper summarizes the challenge tasks and describes the dataset and evaluation criteria established in the EAD 2019 challenge.
A deep learning framework for quality assessment and restoration in video endoscopy
Apr 15, 2019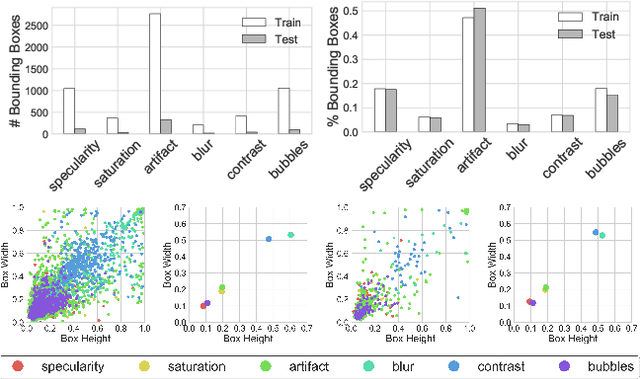

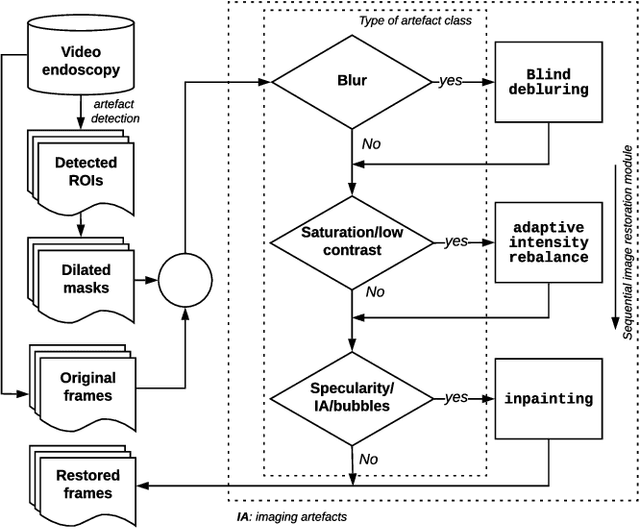

Abstract:Endoscopy is a routine imaging technique used for both diagnosis and minimally invasive surgical treatment. Artifacts such as motion blur, bubbles, specular reflections, floating objects and pixel saturation impede the visual interpretation and the automated analysis of endoscopy videos. Given the widespread use of endoscopy in different clinical applications, we contend that the robust and reliable identification of such artifacts and the automated restoration of corrupted video frames is a fundamental medical imaging problem. Existing state-of-the-art methods only deal with the detection and restoration of selected artifacts. However, typically endoscopy videos contain numerous artifacts which motivates to establish a comprehensive solution. We propose a fully automatic framework that can: 1) detect and classify six different primary artifacts, 2) provide a quality score for each frame and 3) restore mildly corrupted frames. To detect different artifacts our framework exploits fast multi-scale, single stage convolutional neural network detector. We introduce a quality metric to assess frame quality and predict image restoration success. Generative adversarial networks with carefully chosen regularization are finally used to restore corrupted frames. Our detector yields the highest mean average precision (mAP at 5% threshold) of 49.0 and the lowest computational time of 88 ms allowing for accurate real-time processing. Our restoration models for blind deblurring, saturation correction and inpainting demonstrate significant improvements over previous methods. On a set of 10 test videos we show that our approach preserves an average of 68.7% which is 25% more frames than that retained from the raw videos.
Improving Whole Slide Segmentation Through Visual Context - A Systematic Study
Jun 11, 2018
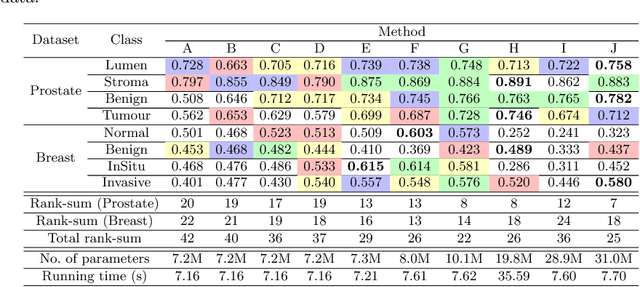
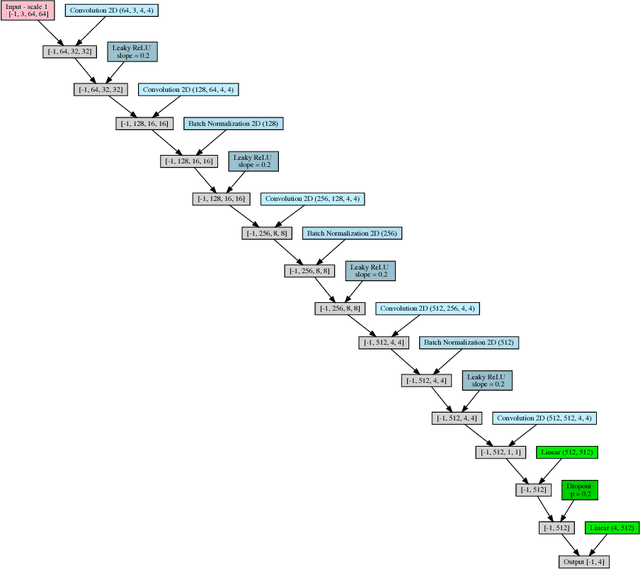
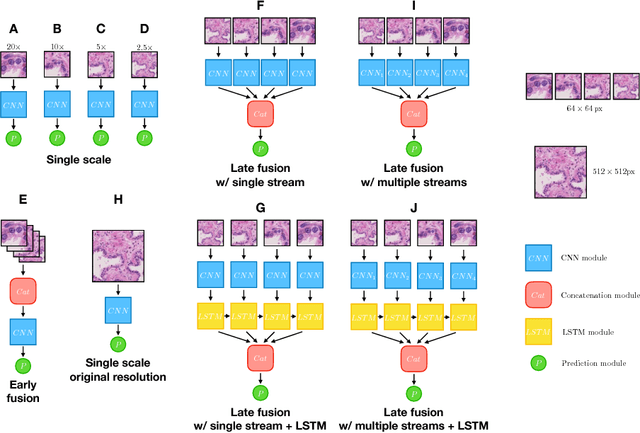
Abstract:While challenging, the dense segmentation of histology images is a necessary first step to assess changes in tissue architecture and cellular morphology. Although specific convolutional neural network architectures have been applied with great success to the problem, few effectively incorporate visual context information from multiple scales. With this paper, we present a systematic comparison of different architectures to assess how including multi-scale information affects segmentation performance. A publicly available breast cancer and a locally collected prostate cancer datasets are being utilised for this study. The results support our hypothesis that visual context and scale play a crucial role in histology image classification problems.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge