Florian Dubost
for the ALFA study
Hydranet: Data Augmentation for Regression Neural Networks
Jul 12, 2018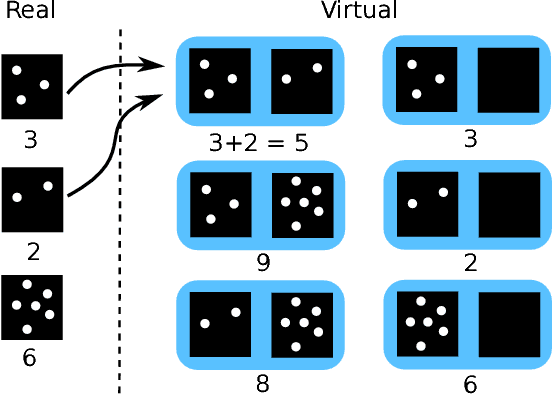
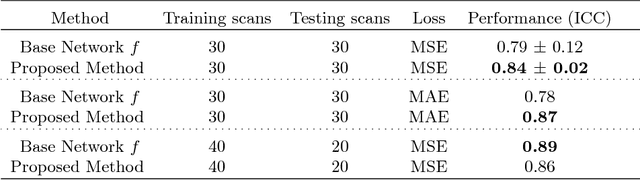
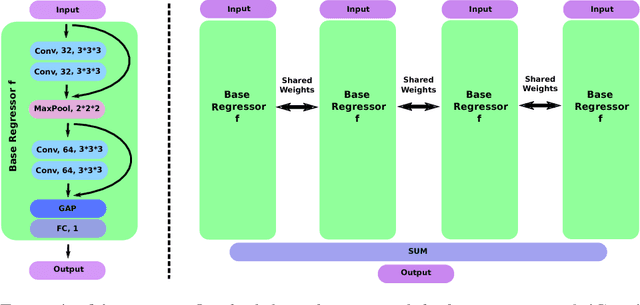
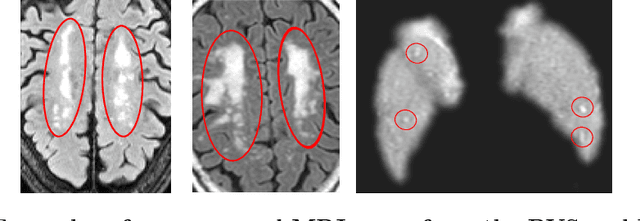
Abstract:Despite recent efforts, deep learning techniques remain often heavily dependent on a large quantity of labeled data. This problem is even more challenging in medical image analysis where the annotator expertise is often scarce. In this paper we propose a novel data-augmentation method to regularize neural network regressors, learning from a single global label per image. The principle of the method is to create new samples by recombining existing ones. We demonstrate the performance of our algorithm on two tasks: the regression of number of enlarged perivascular spaces in the basal ganglia; and the regression of white matter hyperintensities volume. We show that the proposed method improves the performance even when more basic data augmentation is used. Furthermore we reached an intraclass correlation coefficient between ground truth and network predictions of 0.73 on the first task and 0.86 on the second task, only using between 25 and 30 scans with a single global label per scan for training. To achieve a similar correlation on the first task, state-of-the-art methods needed more than 1000 training scans.
Quantification of Lung Abnormalities in Cystic Fibrosis using Deep Networks
Mar 21, 2018Abstract:Cystic fibrosis is a genetic disease which may appear in early life with structural abnormalities in lung tissues. We propose to detect these abnormalities using a texture classification approach. Our method is a cascade of two convolutional neural networks. The first network detects the presence of abnormal tissues. The second network identifies the type of the structural abnormalities: bronchiectasis, atelectasis or mucus plugging.We also propose a network computing pixel-wise heatmaps of abnormality presence learning only from the patch-wise annotations. Our database consists of CT scans of 194 subjects. We use 154 subjects to train our algorithms and the 40 remaining ones as a test set. We compare our method with random forest and a single neural network approach. The first network reaches an accuracy of 0,94 for disease detection, 0,18 higher than the random forest classifier and 0,37 higher than the single neural network. Our cascade approach yields a final class-averaged F1-score of 0,33, outperforming the baseline method and the single network by 0,10 and 0,12.
* SPIE - Medical Imaging 2018: Image Processing
GP-Unet: Lesion Detection from Weak Labels with a 3D Regression Network
Oct 30, 2017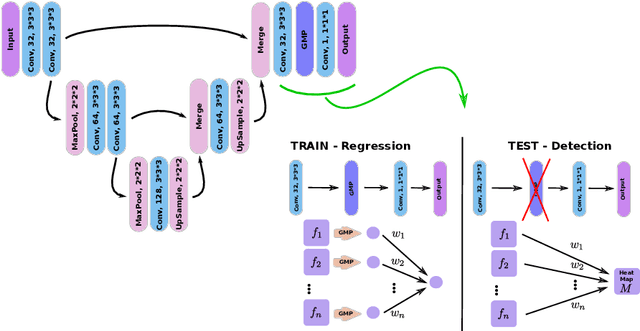
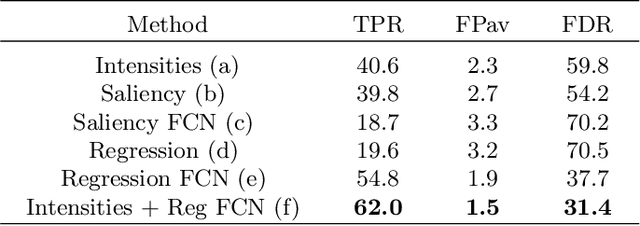
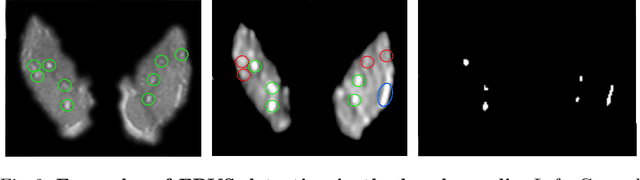
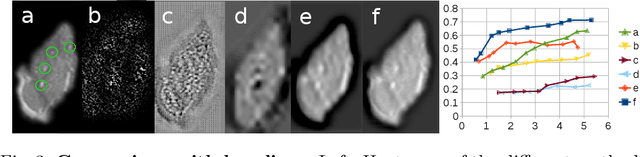
Abstract:We propose a novel convolutional neural network for lesion detection from weak labels. Only a single, global label per image - the lesion count - is needed for training. We train a regression network with a fully convolutional architecture combined with a global pooling layer to aggregate the 3D output into a scalar indicating the lesion count. When testing on unseen images, we first run the network to estimate the number of lesions. Then we remove the global pooling layer to compute localization maps of the size of the input image. We evaluate the proposed network on the detection of enlarged perivascular spaces in the basal ganglia in MRI. Our method achieves a sensitivity of 62% with on average 1.5 false positives per image. Compared with four other approaches based on intensity thresholding, saliency and class maps, our method has a 20% higher sensitivity.
Segmentation of Intracranial Arterial Calcification with Deeply Supervised Residual Dropout Networks
Jun 04, 2017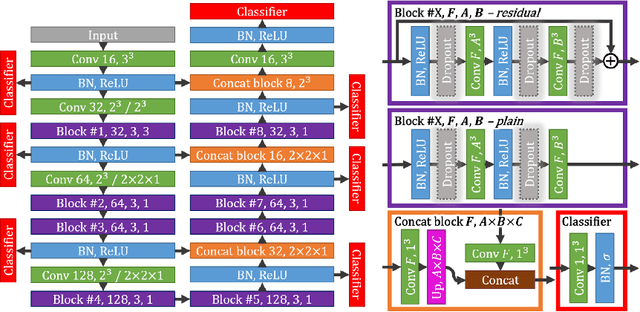
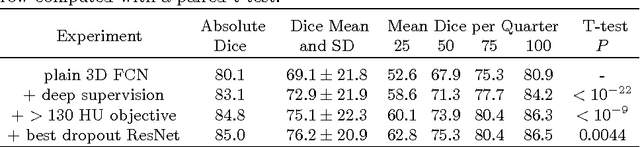
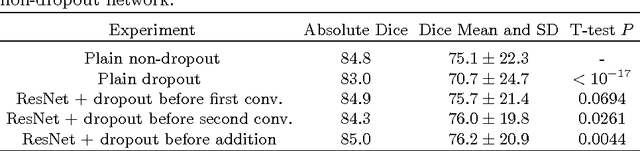
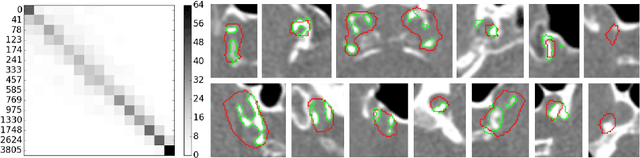
Abstract:Intracranial carotid artery calcification (ICAC) is a major risk factor for stroke, and might contribute to dementia and cognitive decline. Reliance on time-consuming manual annotation of ICAC hampers much demanded further research into the relationship between ICAC and neurological diseases. Automation of ICAC segmentation is therefore highly desirable, but difficult due to the proximity of the lesions to bony structures with a similar attenuation coefficient. In this paper, we propose a method for automatic segmentation of ICAC; the first to our knowledge. Our method is based on a 3D fully convolutional neural network that we extend with two regularization techniques. Firstly, we use deep supervision (hidden layers supervision) to encourage discriminative features in the hidden layers. Secondly, we augment the network with skip connections, as in the recently developed ResNet, and dropout layers, inserted in a way that skip connections circumvent them. We investigate the effect of skip connections and dropout. In addition, we propose a simple problem-specific modification of the network objective function that restricts the focus to the most important image regions and simplifies the optimization. We train and validate our model using 882 CT scans and test on 1,000. Our regularization techniques and objective improve the average Dice score by 7.1%, yielding an average Dice of 76.2% and 97.7% correlation between predicted ICAC volumes and manual annotations.
Hands-Free Segmentation of Medical Volumes via Binary Inputs
Sep 20, 2016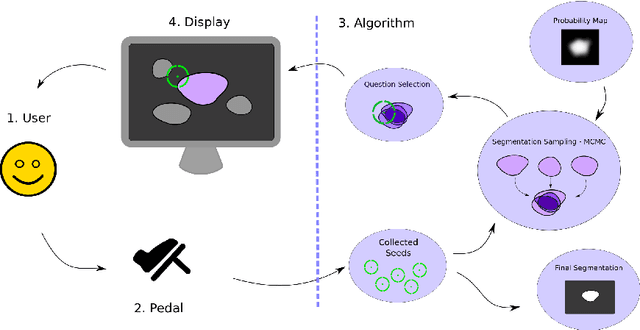
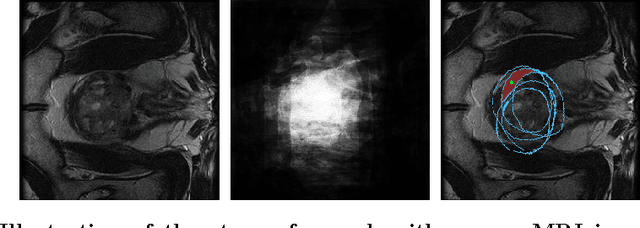
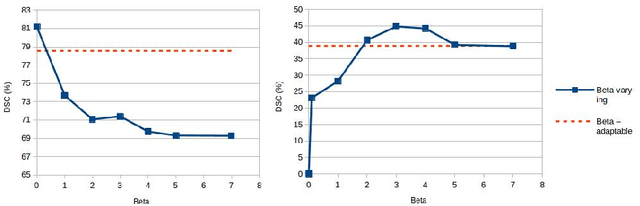
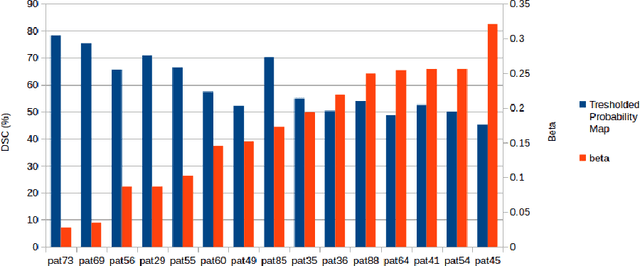
Abstract:We propose a novel hands-free method to interactively segment 3D medical volumes. In our scenario, a human user progressively segments an organ by answering a series of questions of the form "Is this voxel inside the object to segment?". At each iteration, the chosen question is defined as the one halving a set of candidate segmentations given the answered questions. For a quick and efficient exploration, these segmentations are sampled according to the Metropolis-Hastings algorithm. Our sampling technique relies on a combination of relaxed shape prior, learnt probability map and consistency with previous answers. We demonstrate the potential of our strategy on a prostate segmentation MRI dataset. Through the study of failure cases with synthetic examples, we demonstrate the adaptation potential of our method. We also show that our method outperforms two intuitive baselines: one based on random questions, the other one being the thresholded probability map.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge