Fei Shan
Large-Scale Screening of COVID-19 from Community Acquired Pneumonia using Infection Size-Aware Classification
Mar 22, 2020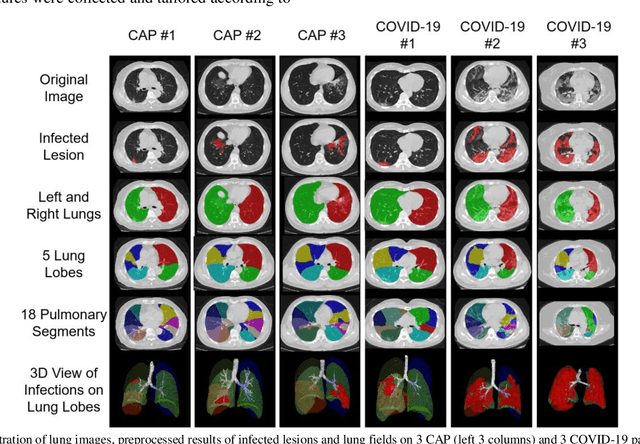
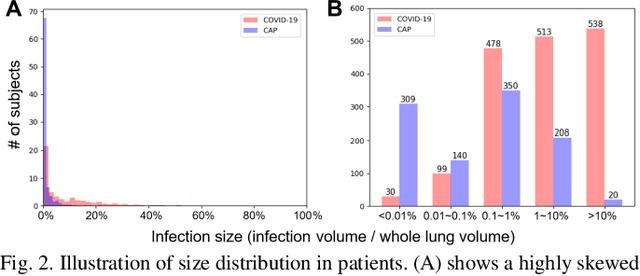
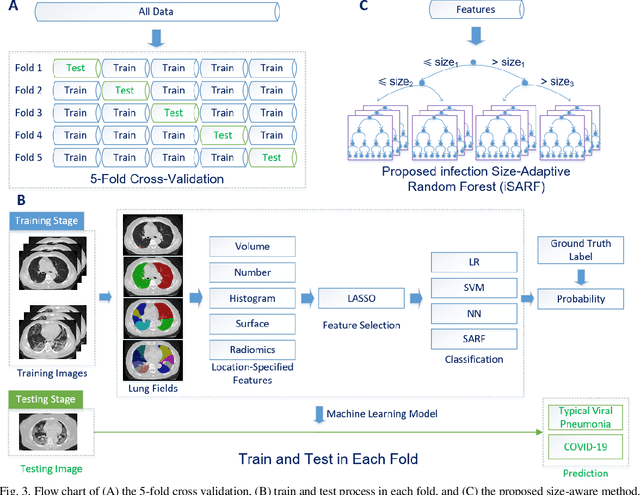
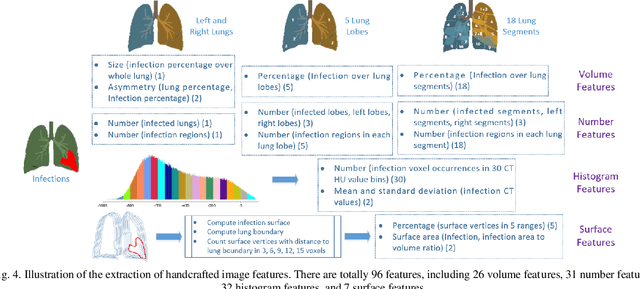
Abstract:The worldwide spread of coronavirus disease (COVID-19) has become a threatening risk for global public health. It is of great importance to rapidly and accurately screen patients with COVID-19 from community acquired pneumonia (CAP). In this study, a total of 1658 patients with COVID-19 and 1027 patients of CAP underwent thin-section CT. All images were preprocessed to obtain the segmentations of both infections and lung fields, which were used to extract location-specific features. An infection Size Aware Random Forest method (iSARF) was proposed, in which subjects were automated categorized into groups with different ranges of infected lesion sizes, followed by random forests in each group for classification. Experimental results show that the proposed method yielded sensitivity of 0.907, specificity of 0.833, and accuracy of 0.879 under five-fold cross-validation. Large performance margins against comparison methods were achieved especially for the cases with infection size in the medium range, from 0.01% to 10%. The further inclusion of Radiomics features show slightly improvement. It is anticipated that our proposed framework could assist clinical decision making.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge